Lecture 1 chemistry measurement conversions Topic Brown Chapter

Lecture 1: chemistry, measurement, conversions Topic Brown Chapter 1 1. Science and Chemistry What is science? The scientific method What is chemistry? 1. 1 2. What is matter? 1. 2 States of matter Pure substances vs. mixtures Physical vs. chemical change 3. Measurement, metric units & prefixes Derived units and density Exact & inexact numbers Uncertainty, precision & accuracy Significant figures & use in calculation Scientific notation 4. Dimensional analysis = conversions With multiple conversion factors Conversions with unit prefixes With cubed unit volumes 1. 3 1. 4 1. 5 1. 6

Measurement is an essential part of chemistry. Quantitation uses the metric system. Chemistry uses scientific notation & unit prefixes. Amounts are usually very small.
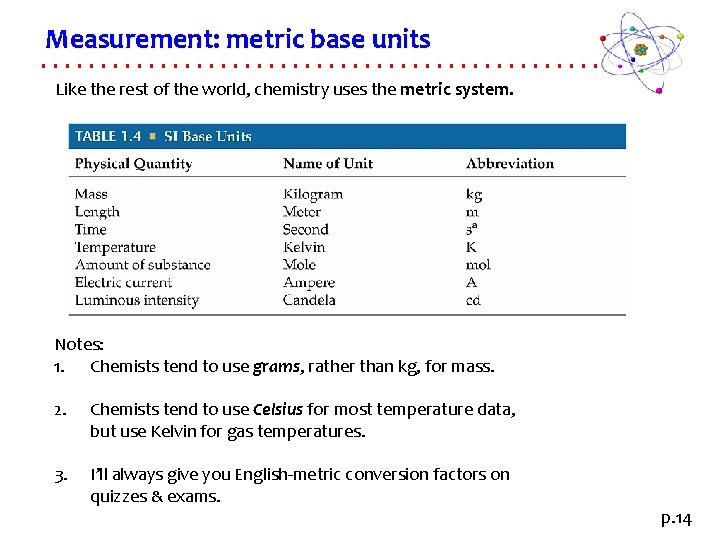
Measurement: metric base units Like the rest of the world, chemistry uses the metric system. Notes: 1. Chemists tend to use grams, rather than kg, for mass. 2. Chemists tend to use Celsius for most temperature data, but use Kelvin for gas temperatures. 3. I’ll always give you English-metric conversion factors on quizzes & exams. p. 14
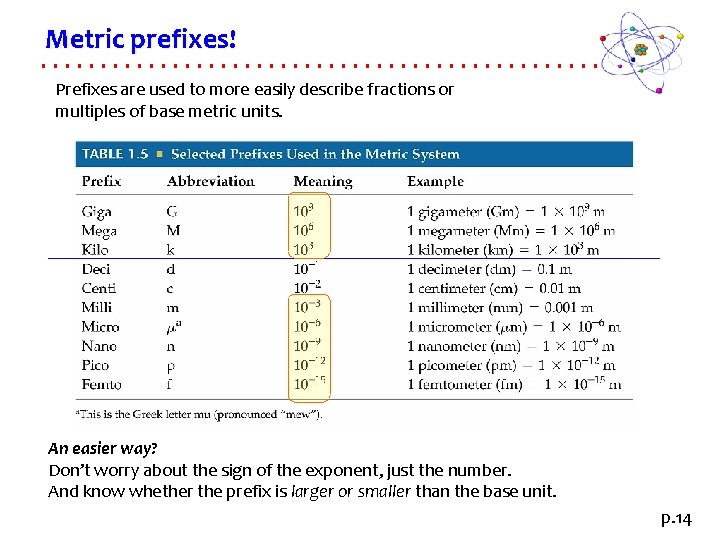
Metric prefixes! Prefixes are used to more easily describe fractions or multiples of base metric units. An easier way? Don’t worry about the sign of the exponent, just the number. And know whether the prefix is larger or smaller than the base unit. p. 14

What’s a derived SI unit? A derived unit is a “complex” unit created by multiplying or or dividing one unit by another. Speed has units of meters/second (m/s) Density has units of mass per volume (g/m. L = g/cm 3 = g/cc) Questions for you: 1. Is density an intensive or extensive property? 2. What’s the difference between density and mass? 3. How can you easily measure the density of an odd-shaped object? p. 17 & 20

Calculating density It’s essential that you have the ability to rearrange equations in order to solve for the chosen variable. And ALWAYS show units! = m/v Calculate the density of beer if a keg has a mass of 7. 71 x 10 4 g & a volume of 58. 7 L. = m = 7. 71 x 104 g = 1. 31 g/m. L v 5. 87 x 104 m. L (Known value ~1. 050 g/m. L) What’s the mass of 3 m. L of gold if its density is 19. 3 g/cm 3? m= v --> 3 m. L 19. 3 g = 57. 9 g --> 60 g 1 m. L What volume is occupied by 2 kg of peanuts if their density is 0. 272 g/m. L? v=m --> 2 kg = 2000 g --> v = 2000 g 0. 272 g/m. L = 7352. 9 --> 7 L

Exact, inexact and uncertainty Numbers or values can be described as either exact or inexact: Exact Values are known precisely; or values given as references. Conversion factors are exact. Results of most counting are exact. Inexact Values measured by humans & their instruments; some uncertainty. Uncertainty Some level of uncertainty exists in all inexact numbers (or all measured values) The last digit is generally the most uncertain, since it represents the technical limits of the instrument used. Instruments are least accurate at the ends of their ranges. 2. 2405 0. 0001 g The ten thousandth place is the least certain as it’s the smallest calibration on the instrument. The notation also shows this by saying that 5 could easily be a 4 or a 6. p. 20 -1

Precision vs. accuracy Both terms are used to discuss how certain, or uncertain, data, results or other types of information are. But the terms are often confused. Precision How closely do individual measurements agree with each other? Accuracy How close is the measurement to a truth or known value? Questions for you? 1. Which is easier to achieve in the lab? 2. How can we know if we are precise? 3. How can we know if we’re accurate? p. 21

Significant figures - ZEROs All digits of a measured value are significant, thought the last value is usually least certain. So why is this such a pain in the #@&!? 1. Zeros 2. Calculations Zeros in a value may or may not be significant. Here are the rules: 1. Trapped zeros - between non-zero digits are ALWAYS significant 1005 kg, 1. 03 seconds 2. Leading zeros - before any non-zeros are NEVER significant 0. 0039 g, 0. 2 cm 3. Trailing zeros - may be significant IF WITH A DECIMAL 3500 NO, only 2 sf 3500. 0 YES, all five are sf p. 21 -2

Significant figures in calculations There are two simple rules that are OFTEN ignored… to the peril of chemistry grades. You want calculated answers to reflect the degree of calibration of the instruments used to gather the data. 1. Multiplication & division: least sf rules! The answer has only as many sf as the value with the lowest sf. (6. 221 cm)(5. 2 cm) = 32. 3492 cm 2 -> 32 cm 2 2. Addition & subtraction: limit number of sf after the decimal So the number of digits following the decimal is limited by the value in the calc with fewest sf after the decimal. Number before the decimal is not limited. 20. 4 + 1. 322 + 83 = 104. 722 -> 105 There are 1609. 344 m in a mile. How many meters are there in 1. 35 miles? 1. 35 miles 1609. 344 m = 2172. 6144 m --> 2. 17 x 103 m 1 mile 64. 2 + 7. 9 71. 2 = 0. 327 220. 3 p. 23

Scientific notation Allows rapid & clear expression of very large, or very small numbers with the correct number of significant figures. Exponent tells you how many zeros (or places) should follow the first digit, or how many places to move the decimal. + right; - left Translate from scientific nontation 101 = 2 X 101 = 10 20 102 = 100 2 X 102 = 103 = 1000 2. 5 X 102 = 250 104 = 10000 3. 9 X 106 = 3. 9 million 106 = 1 million 0. 8 X 1012 = 8 w/ 11 zeros 200 How many sig figs? 3 1. 03 X 104 1. 030 X 104 4 1. 0300 X 104 5 0. 0300 X 104 3 Calculate the volume of the box with width, length & height of 15. 5, 27. 3 x 5. 4 cm. volume of a cube = w x l x h = (15. 5 cm)(27. 3 cm)(5. 4 cm) = 2285. 01 cm 3 The height, 5. 4 cm, limits the number of sfs in the answer to 2. So, 2285. 01 cm 3 = 2. 3 x 103 cm 3
- Slides: 11