Lecture 1 Chemical Reaction Engineering CRE is the



































- Slides: 61

Lecture 1 Chemical Reaction Engineering (CRE) is the field that studies the rates and mechanisms of chemical reactions and the design of the reactors in which they take place. 1

Lecture 1 – Thursday �Introduction �Definitions �General Mole Balance Equation � Batch (BR) � Continuously Stirred Tank Reactor (CSTR) � Plug Flow Reactor (PFR) � Packed Bed Reactor (PBR) 2

Chemical Reaction Engineering �Chemical reaction engineering is at the heart of virtually every chemical process. It separates the chemical engineer from other engineers. Industries that Draw Heavily on Chemical Reaction Engineering (CRE) are: CPI (Chemical Process Industries) Examples like Dow, Du. Pont, Amoco, Chevron 3

4

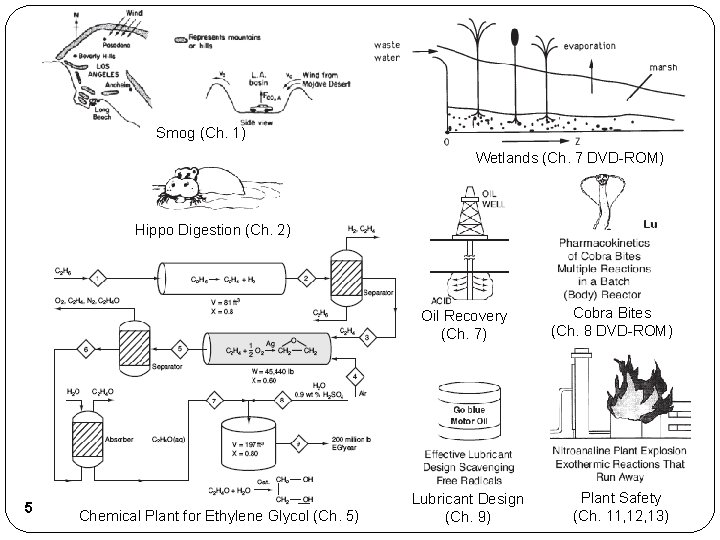
Smog (Ch. 1) Wetlands (Ch. 7 DVD-ROM) Hippo Digestion (Ch. 2) Oil Recovery (Ch. 7) 5 Chemical Plant for Ethylene Glycol (Ch. 5) Lubricant Design (Ch. 9) Cobra Bites (Ch. 8 DVD-ROM) Plant Safety (Ch. 11, 12, 13)

Materials on the Web and CD-ROM http: //www. umich. edu/~essen/ 6

Let’s Begin CRE �Chemical Reaction Engineering (CRE) is the field that studies the rates and mechanisms of chemical reactions and the design of the reactors in which they take place. 7

Chemical Identity �A chemical species is said to have reacted when it has lost its chemical identity. �The identity of a chemical species is determined by the kind, number, and configuration of that species’ atoms. 8

Chemical Identity �A chemical species is said to have reacted when it has lost its chemical identity. �There are three ways for a species to loose its identity: 1. Decomposition CH 3 H 2 + H 2 C=CH 2 2. Combination N 2 + O 2 2 NO 3. Isomerization C 2 H 5 CH=CH 2 CH 2=C(CH 3)2 9

Reaction Rate �The reaction rate is the rate at which a species looses its chemical identity per unit volume. �The rate of a reaction (mol/dm 3/s) can be expressed as either: �The rate of Disappearance of reactant: -r. A or as �The rate of Formation (Generation) of product: r. P 10

Reaction Rate Consider the isomerization A B r. A = the rate of formation of species A per unit volume -r. A = the rate of a disappearance of species A per unit volume r. B = the rate of formation of species B per unit volume 11

Reaction Rate EXAMPLE: A B If Species B is being formed at a rate of 0. 2 moles per decimeter cubed per second, i. e. , r. B = 0. 2 mole/dm 3/s Then A is disappearing at the same rate: -r. A= 0. 2 mole/dm 3/s The rate of formation (generation of A) is: r. A= -0. 2 mole/dm 3/s 12

Reaction Rate � For a catalytic reaction we refer to –r. A’ , which is the rate of disappearance of species A on a per mass of catalyst basis. (mol/gcat/s) NOTE: d. CA/dt is not the rate of reaction 13

Reaction Rate Consider species j: 1. rj is the rate of formation of species j per unit volume [e. g. mol/dm 3 s] 2. rj is a function of concentration, temperature, pressure, and the type of catalyst (if any) 3. rj is independent of the type of reaction system (batch, plug flow, etc. ) 4. rj is an algebraic equation, not a differential equation (e. g. -r. A = k. CA or -r. A = k. CA 2) 14

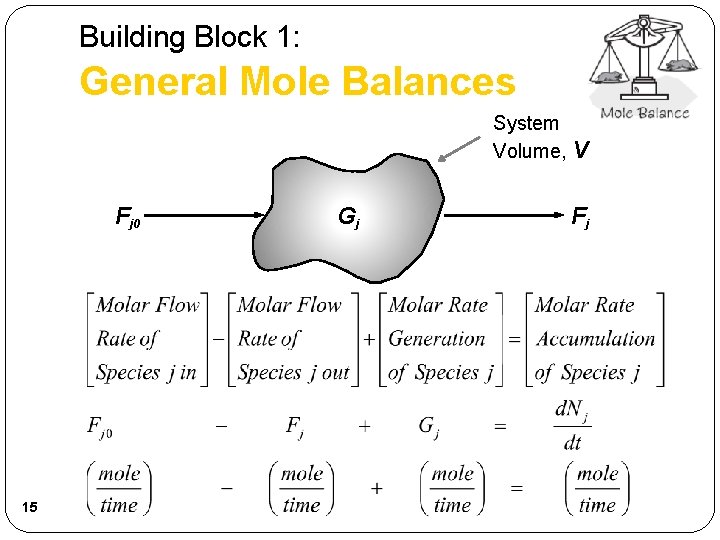
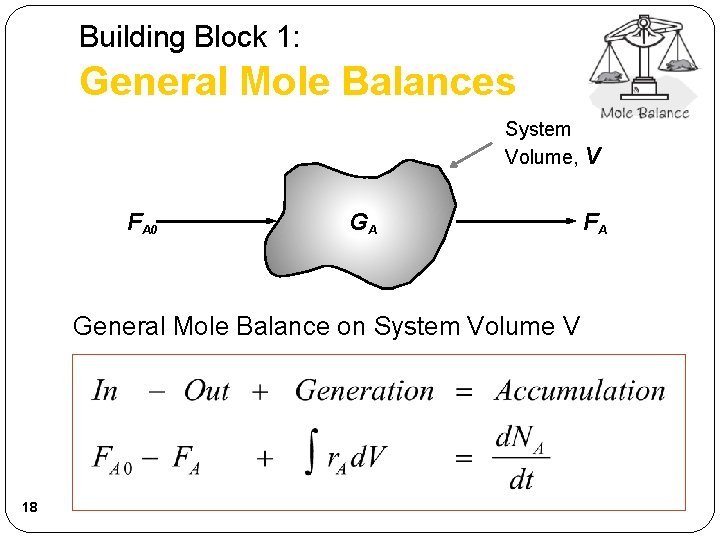
Building Block 1: General Mole Balances System Volume, V Fj 0 15 Gj Fj

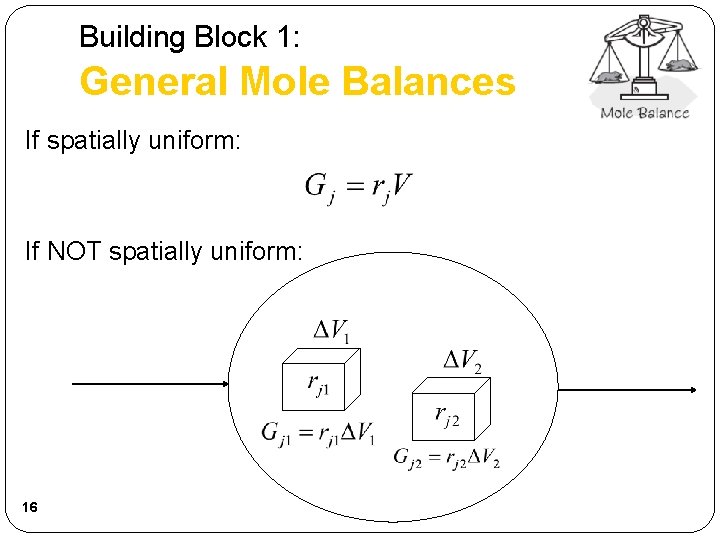
Building Block 1: General Mole Balances If spatially uniform: If NOT spatially uniform: 16

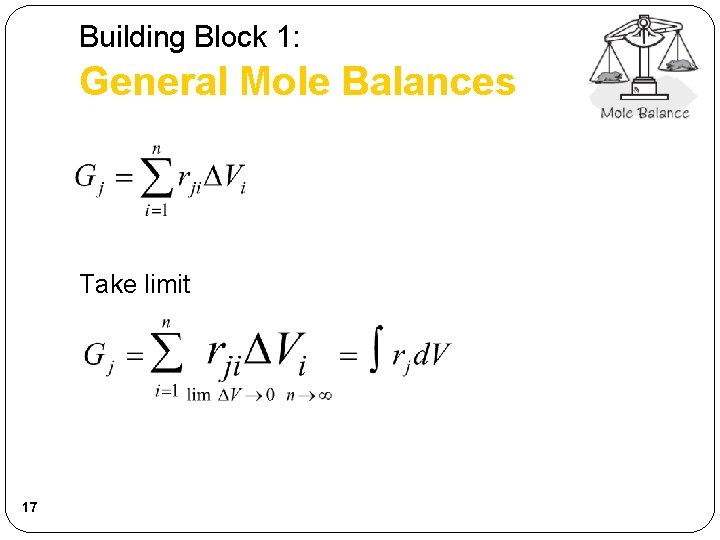
Building Block 1: General Mole Balances Take limit 17

Building Block 1: General Mole Balances System Volume, V FA 0 GA General Mole Balance on System Volume V 18 FA

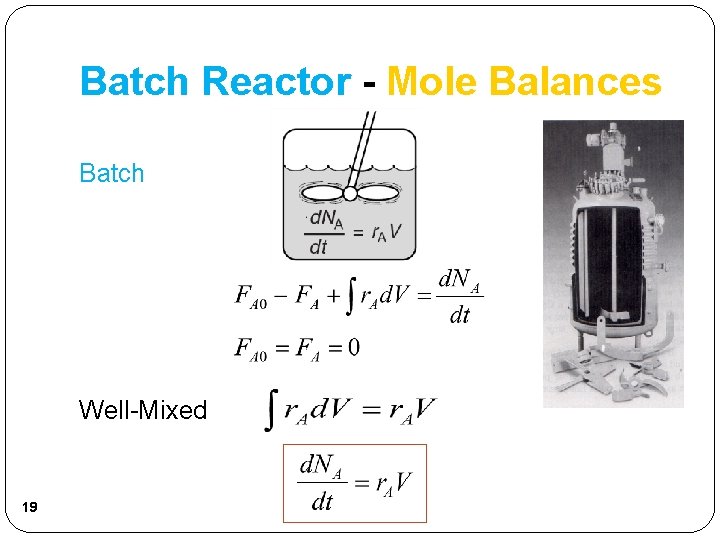
Batch Reactor - Mole Balances Batch Well-Mixed 19

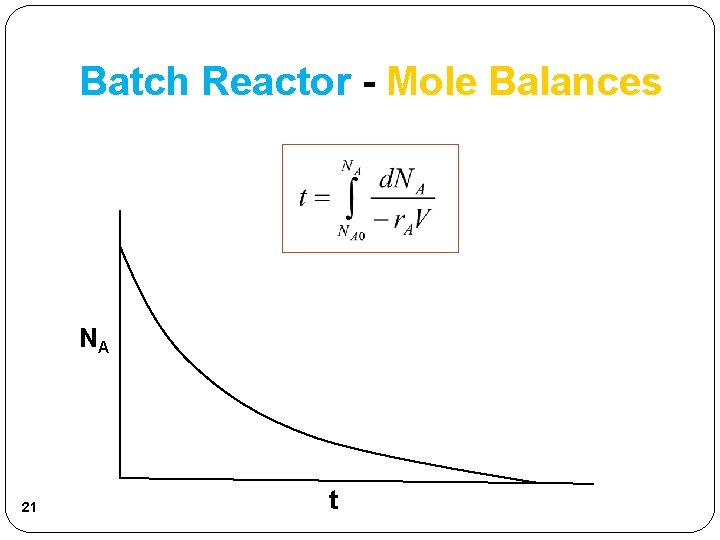
Batch Reactor - Mole Balances Integrating when Time necessary to reduce the number of moles of A from NA 0 to NA. 20

Batch Reactor - Mole Balances NA 21 t

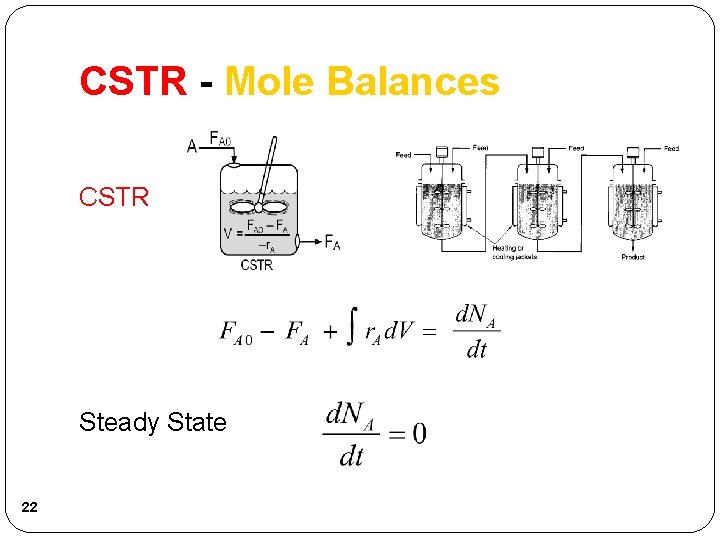
CSTR - Mole Balances CSTR Steady State 22

CSTR - Mole Balances Well Mixed CSTR volume necessary to reduce the molar flow rate from FA 0 to FA. 23

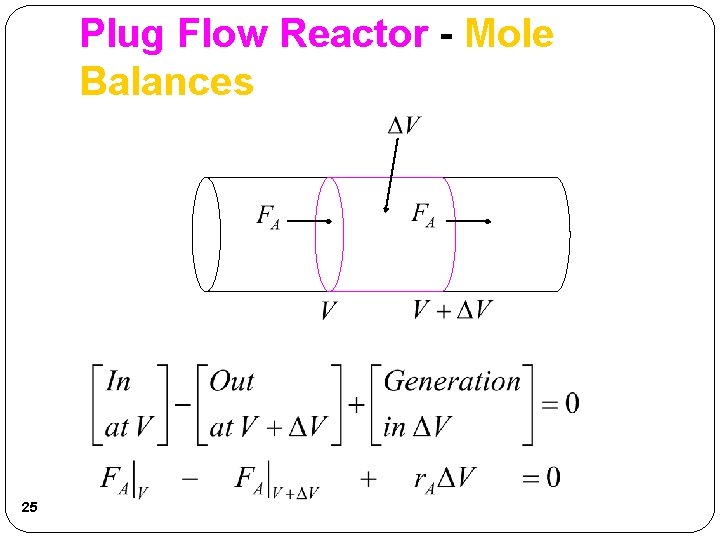
Plug Flow Reactor - Mole Balances 24

Plug Flow Reactor - Mole Balances 25

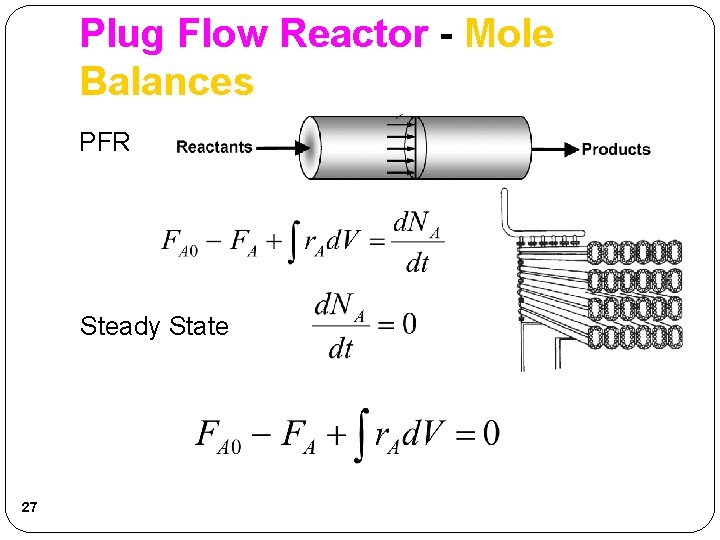
Plug Flow Reactor - Mole Balances Rearrange and take limit as ΔV 0 This is the volume necessary to reduce the entering molar flow rate (mol/s) from FA 0 to the exit molar flow rate of FA. 26

Plug Flow Reactor - Mole Balances PFR Steady State 27

Plug Flow Reactor Alternative Derivation - Mole Balances Differientiate with respect to V The integral form is: 28 This is the volume necessary to reduce the entering molar flow rate (mol/s) from FA 0 to the exit molar flow rate of FA.

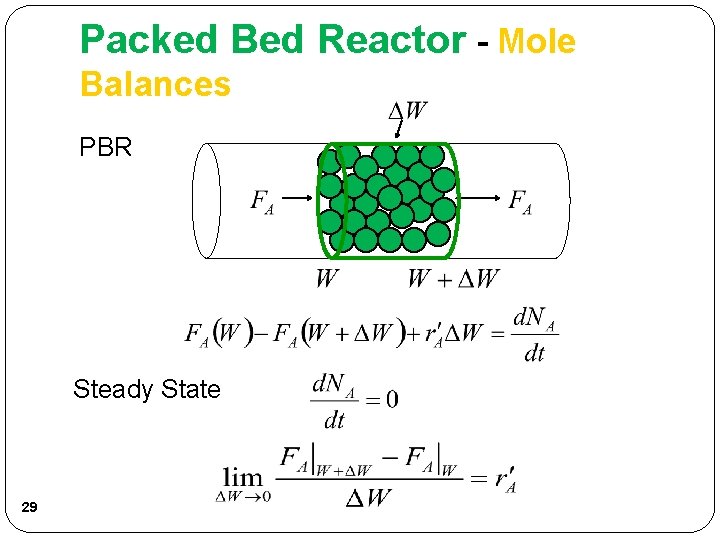
Packed Bed Reactor - Mole Balances PBR Steady State 29

Packed Bed Reactor - Mole Balances Rearrange: The integral form to find the catalyst weight is: PBR catalyst weight necessary to reduce the entering molar flow rate FA 0 to molar flow rate FA. 30

Reactor Mole Balances Summary The GMBE applied to the four major reactor types (and the general reaction A B) Reactor Differential Algebraic Integral NA Batch t CSTR FA PFR V PBR 31 FA W

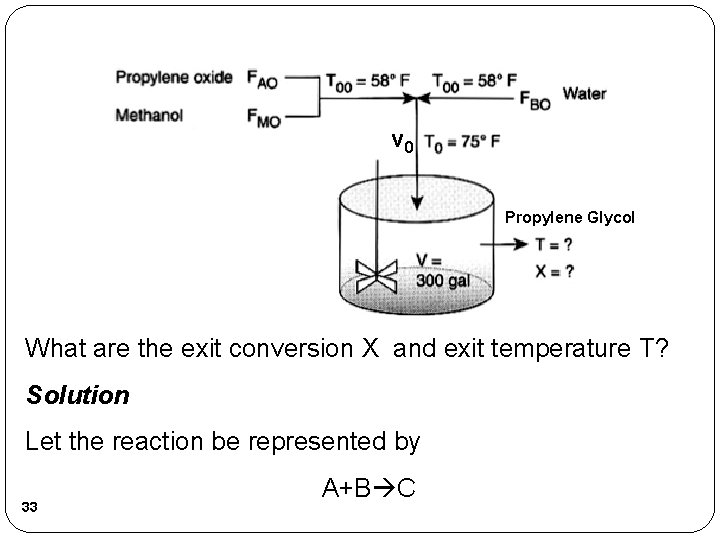
Reactors with Heat Effects �EXAMPLE: Production of Propylene Glycol in an Adiabatic CSTR �Propylene glycol is produced by the hydrolysis of propylene oxide: 32

v 0 Propylene Glycol What are the exit conversion X and exit temperature T? Solution Let the reaction be represented by 33 A+B C

34

35

36

37

38

Evaluate energy balance terms 39

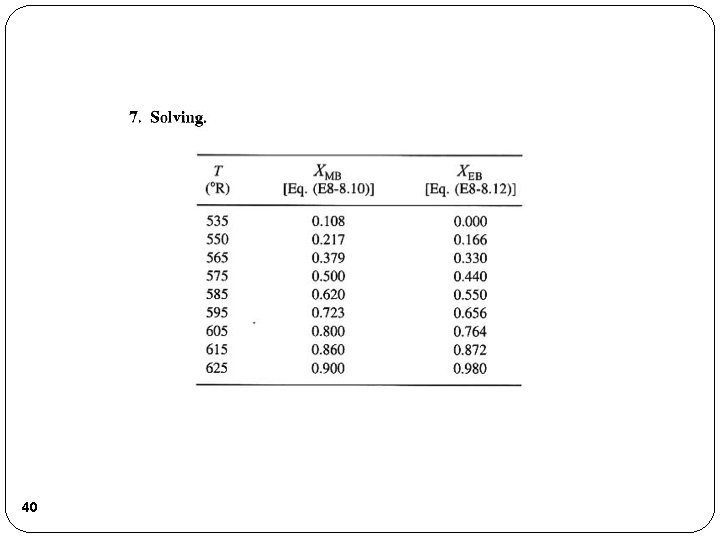
40

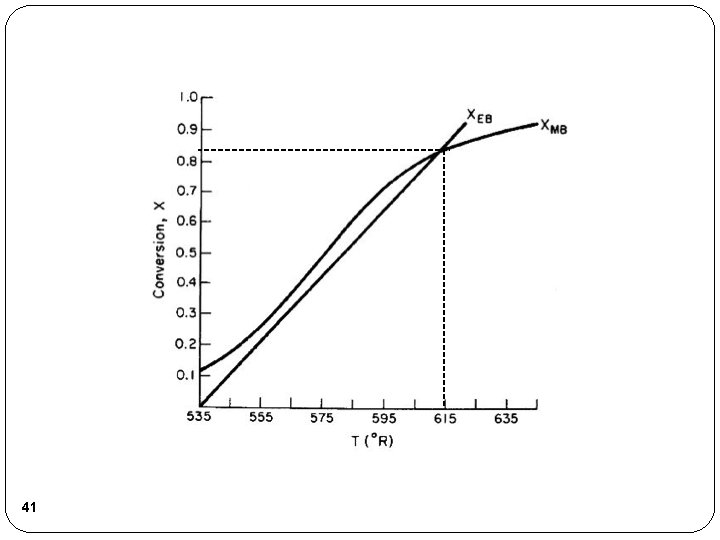
41

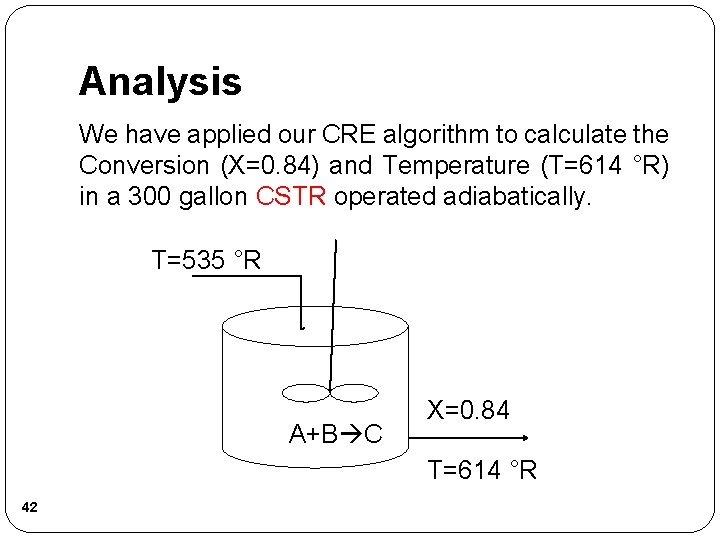
Analysis We have applied our CRE algorithm to calculate the Conversion (X=0. 84) and Temperature (T=614 °R) in a 300 gallon CSTR operated adiabatically. T=535 °R A+B C X=0. 84 T=614 °R 42

Keeping Up 43

Separations Filtration Distillation Adsorption These topics do not build upon one another. 44

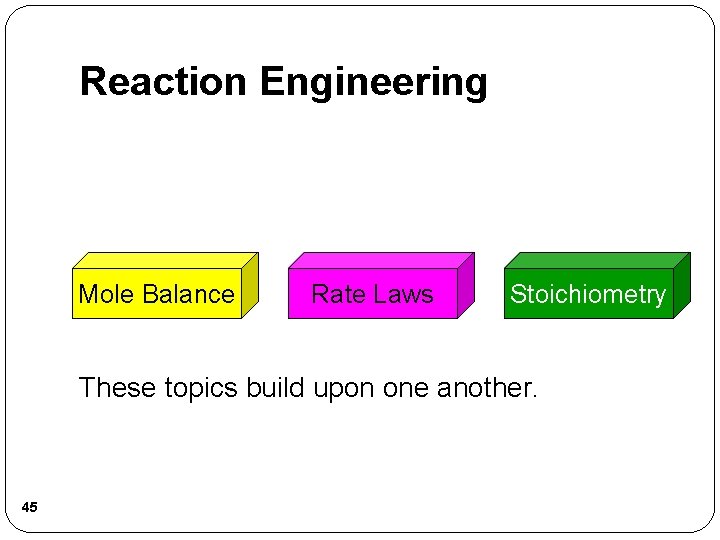
Reaction Engineering Mole Balance Rate Laws Stoichiometry These topics build upon one another. 45

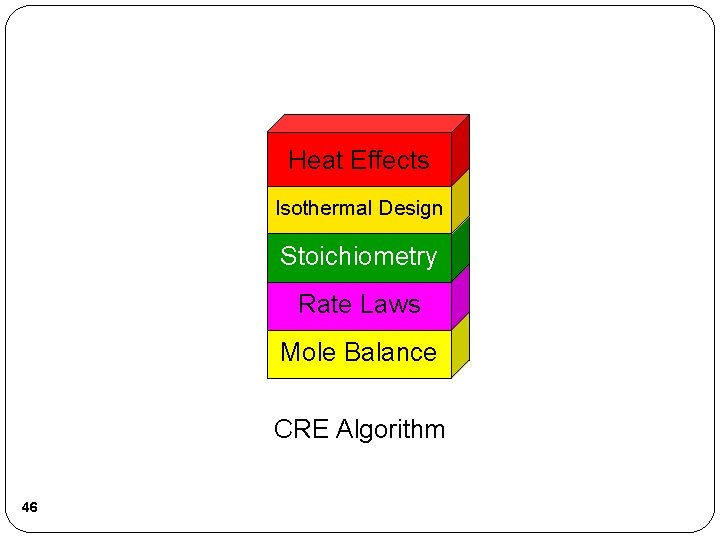
Heat Effects Isothermal Design Stoichiometry Rate Laws Mole Balance CRE Algorithm 46

Mole Balance Rate Laws Be careful not to cut corners on any of the CRE building blocks while learning this material! 47

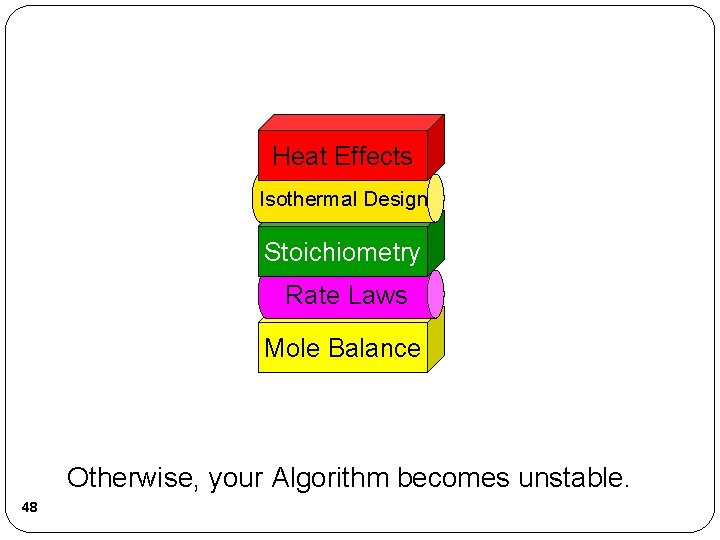
Heat Effects Isothermal Design Stoichiometry Rate Laws Mole Balance Otherwise, your Algorithm becomes unstable. 48

End of Lecture 1 49

Supplemental Slides Additional Applications of CRE 50

Supplemental Slides Additional Applications of CRE 51

Supplemental Slides Additional Applications of CRE 52

Supplemental Slides Additional Applications of CRE Hippo Digestion (Ch. 2) 53

Supplemental Slides Additional Applications of CRE 54

Supplemental Slides Additional Applications of CRE 55

Supplemental Slides Additional Applications of CRE Smog (Ch. 1) 56

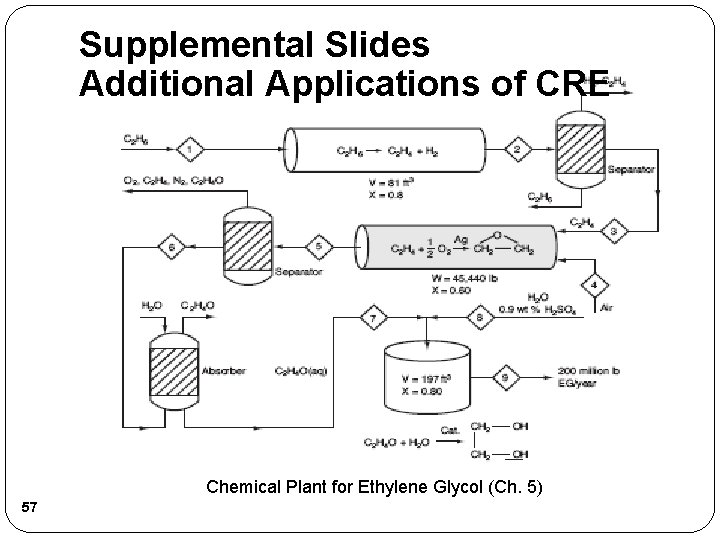
Supplemental Slides Additional Applications of CRE Chemical Plant for Ethylene Glycol (Ch. 5) 57

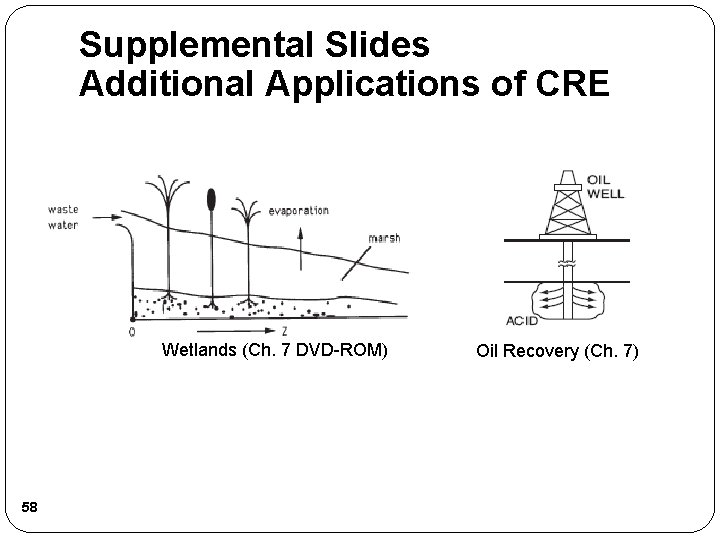
Supplemental Slides Additional Applications of CRE Wetlands (Ch. 7 DVD-ROM) 58 Oil Recovery (Ch. 7)

Supplemental Slides Additional Applications of CRE Cobra Bites (Ch. 8 DVD-ROM) 59

Supplemental Slides Additional Applications of CRE Lubricant Design (Ch. 9) 60

Supplemental Slides Additional Applications of CRE Plant Safety (Ch. 11, 12, 13) 61