Lecture 02 Fundamental Properties of Solids 14 Bravais

Lecture 02 Fundamental Properties of Solids
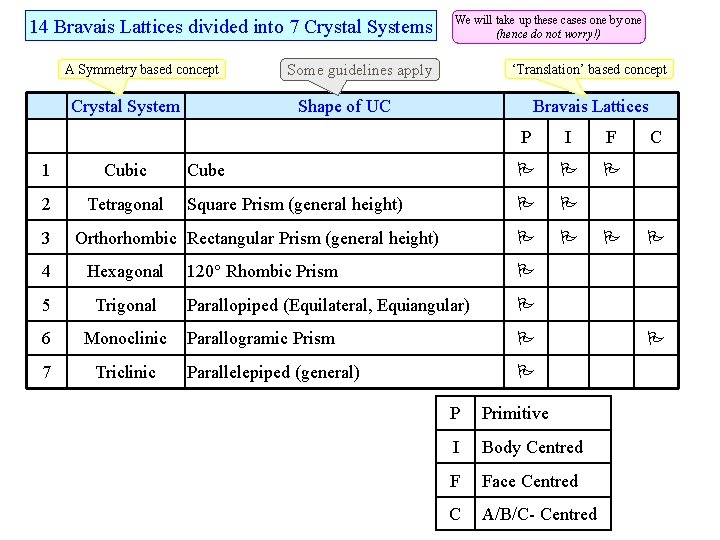
14 Bravais Lattices divided into 7 Crystal Systems A Symmetry based concept Crystal System 1 Cubic 2 Tetragonal 3 We will take up these cases one by one (hence do not worry!) ‘Translation’ based concept Some guidelines apply Shape of UC Bravais Lattices P I F Cube Square Prism (general height) Orthorhombic Rectangular Prism (general height) 4 Hexagonal 5 Trigonal 6 Monoclinic 7 Triclinic 120 Rhombic Prism Parallopiped (Equilateral, Equiangular) Parallogramic Prism Parallelepiped (general) P Primitive I Body Centred F Face Centred C A/B/C- Centred C
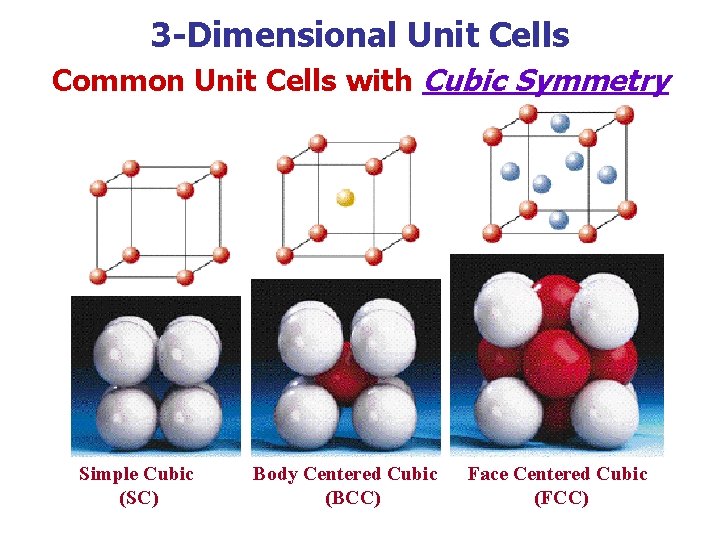
3 -Dimensional Unit Cells Common Unit Cells with Cubic Symmetry Simple Cubic (SC) Body Centered Cubic (BCC) Face Centered Cubic (FCC)
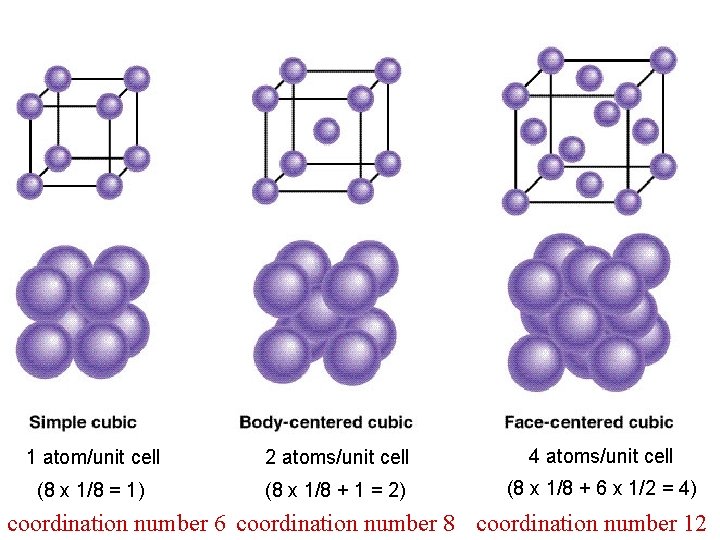
1 atom/unit cell 2 atoms/unit cell 4 atoms/unit cell (8 x 1/8 = 1) (8 x 1/8 + 1 = 2) (8 x 1/8 + 6 x 1/2 = 4) coordination number 6 coordination number 8 coordination number 12
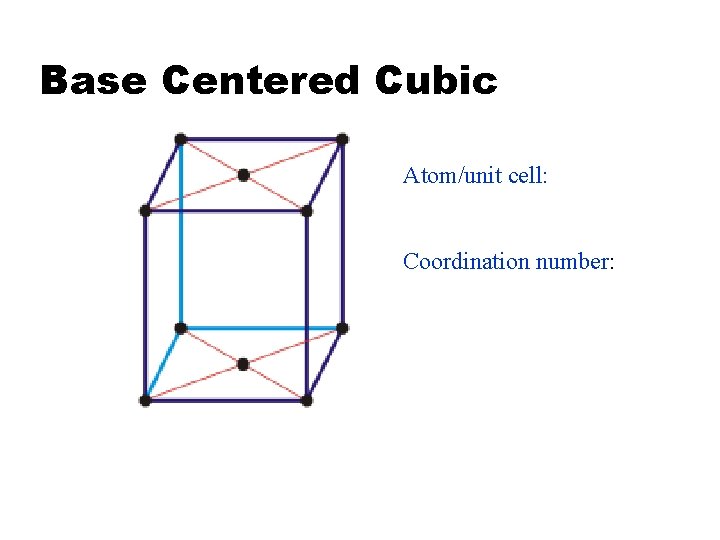
Base Centered Cubic Atom/unit cell: Coordination number:

Primitive & Conventional Unit Cells Unıt Cell Types Primitive Conventional (Non-primitive) A single lattice point per cell More than one lattice point per cell Volume (area) = integer multiple of The smallest area in 2 dimensions, or that for primitive cell The smallest volume in 3 dimensions Simple Cubic (sc) Body Centered Cubic (bcc) Conventional Cell = Primitive cell Conventional Cell ≠ Primitive cell
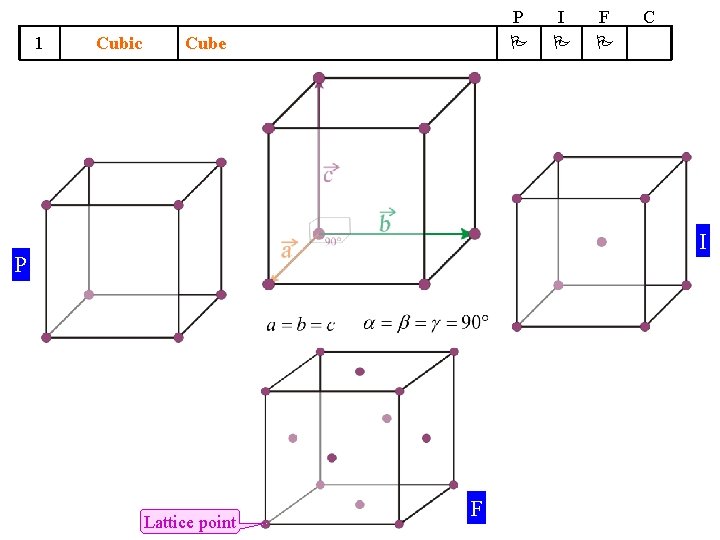
1 Cubic P Cube I F C I P Lattice point F
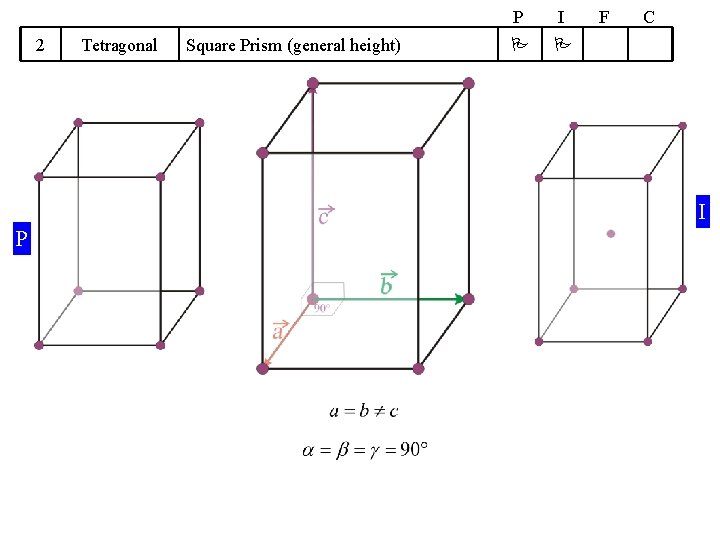
2 Tetragonal Square Prism (general height) P I F C I P
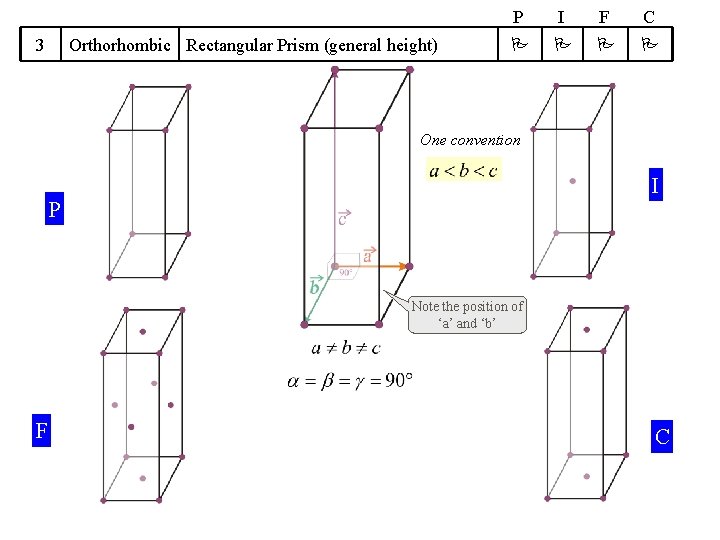
3 Orthorhombic Rectangular Prism (general height) P I F C One convention I P Note the position of ‘a’ and ‘b’ F C
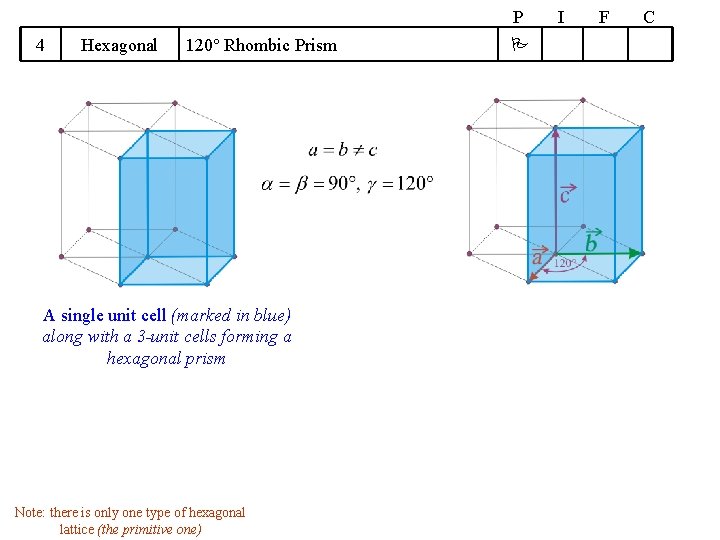
P 4 Hexagonal 120 Rhombic Prism A single unit cell (marked in blue) along with a 3 -unit cells forming a hexagonal prism Note: there is only one type of hexagonal lattice (the primitive one) I F C
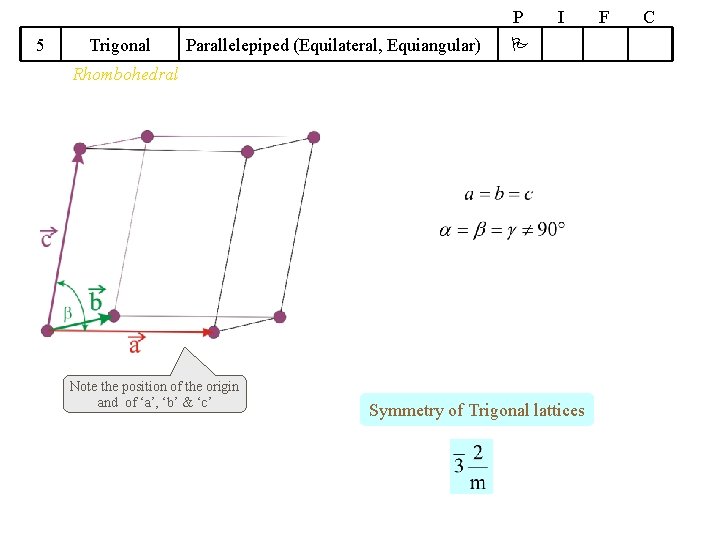
P 5 Trigonal Parallelepiped (Equilateral, Equiangular) I Rhombohedral Note the position of the origin and of ‘a’, ‘b’ & ‘c’ Symmetry of Trigonal lattices F C
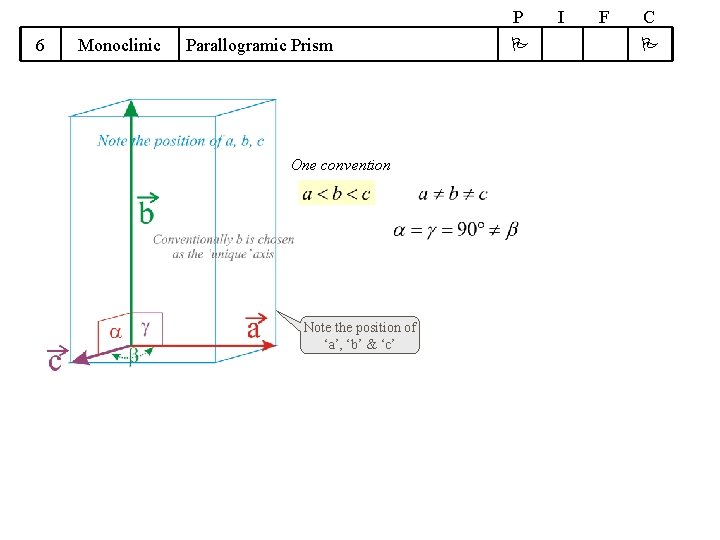
P 6 Monoclinic Parallogramic Prism One convention Note the position of ‘a’, ‘b’ & ‘c’ I F C
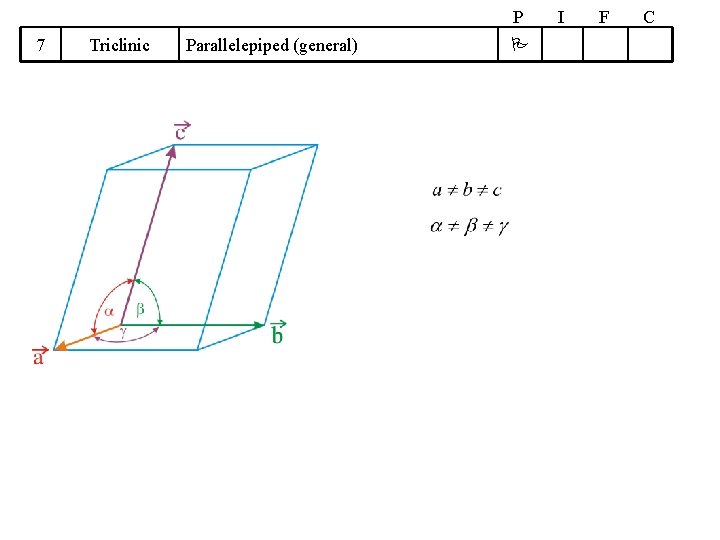
P 7 Triclinic Parallelepiped (general) I F C

Closed-packed structures z There an infinite number of ways to organize spheres to maximize the packing fraction. The centres of spheres at A, B, and C positions (from Kittel) There are different ways you can pack spheres together. This shows two ways, one by putting the spheres in an ABAB… arrangement, the other with ACAC…. (or any combination of the two works)

Hexagonal Close Packed z. Cell of an HCP lattice is visualized as a top and bottom plane of 7 atoms, forming a regular hexagon around a central atom. In between these planes is a half-hexagon of 3 atoms. Be, Sc, Te, Co, Zn, Y, Zr, Tc, Ru, Gd, Tb, Py, Ho, Er, Tm, Lu, Hf, Re, Os, Tl

Hexagonal Close Packed z. There are two lattice parameters in HCP, a and c, representing the basal and height parameters respectively. In the ideal case, the c/a ratio is 1. 633, however, deviations do occur. z. Coordination number for HCP are exactly the same as those for FCC: 12 z. This is because they are both considered close packed structures.
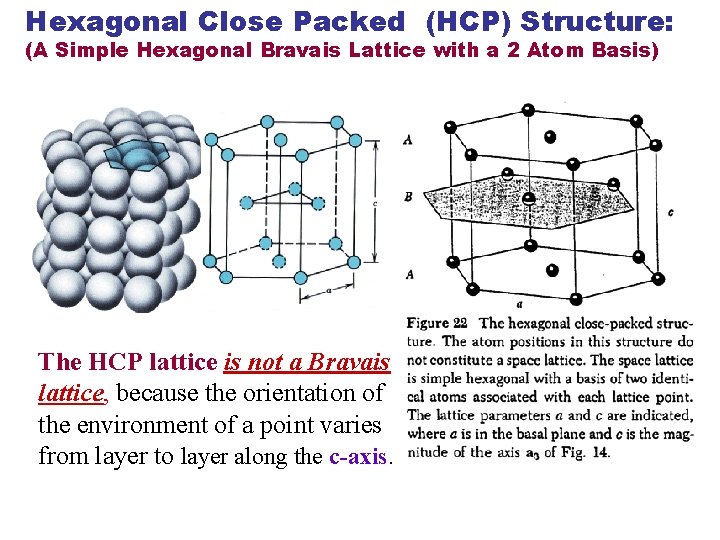
Hexagonal Close Packed (HCP) Structure: (A Simple Hexagonal Bravais Lattice with a 2 Atom Basis) The HCP lattice is not a Bravais lattice, because the orientation of the environment of a point varies from layer to layer along the c-axis.
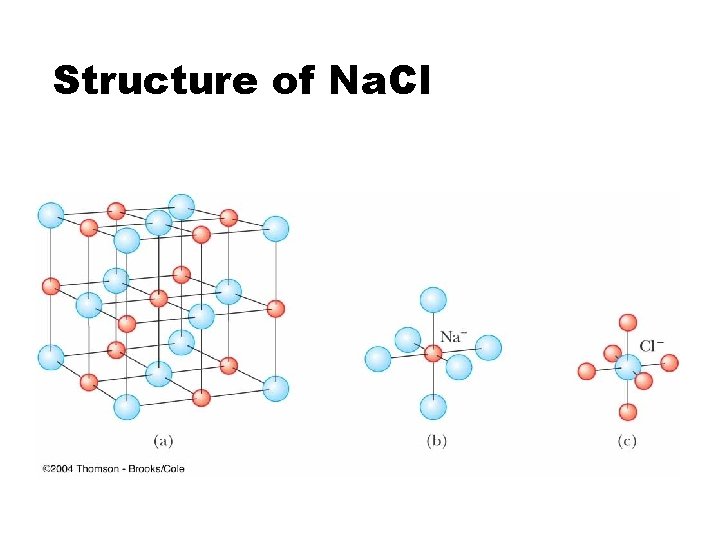
Structure of Na. Cl
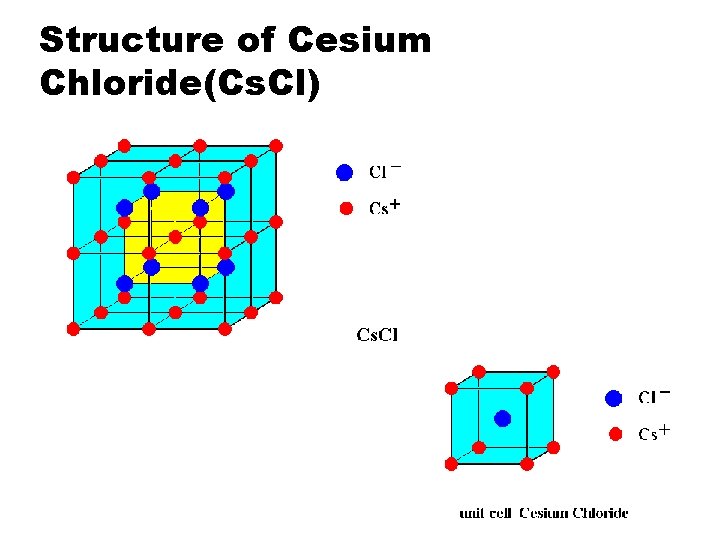
Structure of Cesium Chloride(Cs. Cl)
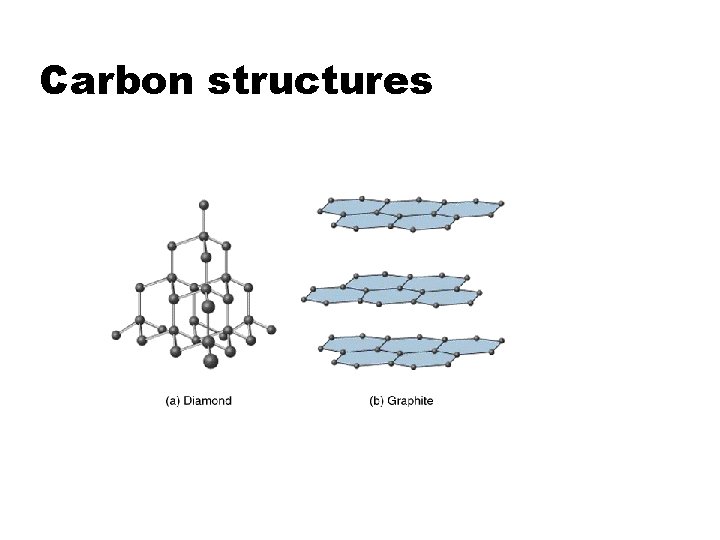
Carbon structures
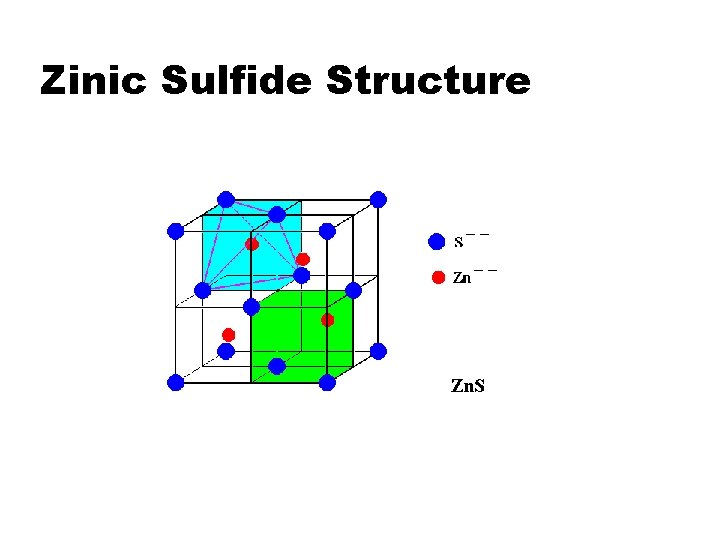
Zinic Sulfide Structure

Why are planes in a lattice important? (A) Determining crystal structure Diffraction methods directly measure the distance between parallel planes of lattice points. This information is used to determine the lattice parameters in a crystal and measure the angles between lattice planes. (B) Plastic deformation Plastic (permanent) deformation in metals occurs by the slip of atoms past each other in the crystal. This slip tends to occur preferentially along specific lattice planes in the crystal. Which planes slip depends on the crystal structure of the material.
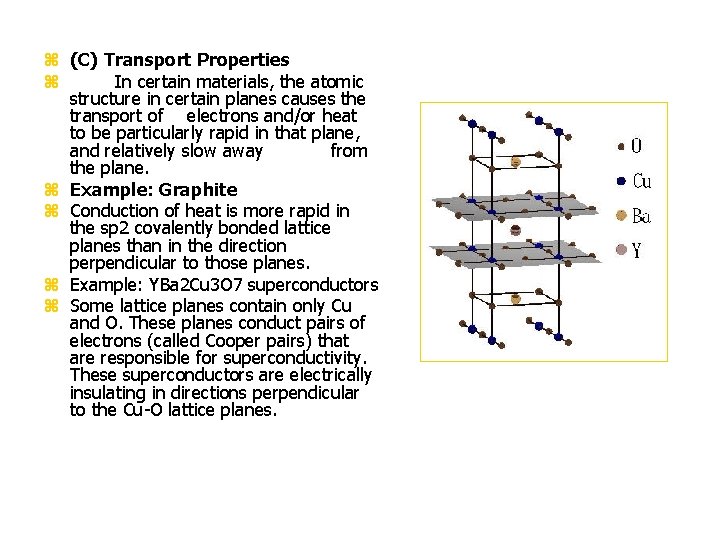
z (C) Transport Properties z In certain materials, the atomic structure in certain planes causes the transport of electrons and/or heat to be particularly rapid in that plane, and relatively slow away from the plane. z Example: Graphite z Conduction of heat is more rapid in the sp 2 covalently bonded lattice planes than in the direction perpendicular to those planes. z Example: YBa 2 Cu 3 O 7 superconductors z Some lattice planes contain only Cu and O. These planes conduct pairs of electrons (called Cooper pairs) that are responsible for superconductivity. These superconductors are electrically insulating in directions perpendicular to the Cu-O lattice planes.

(GPa)
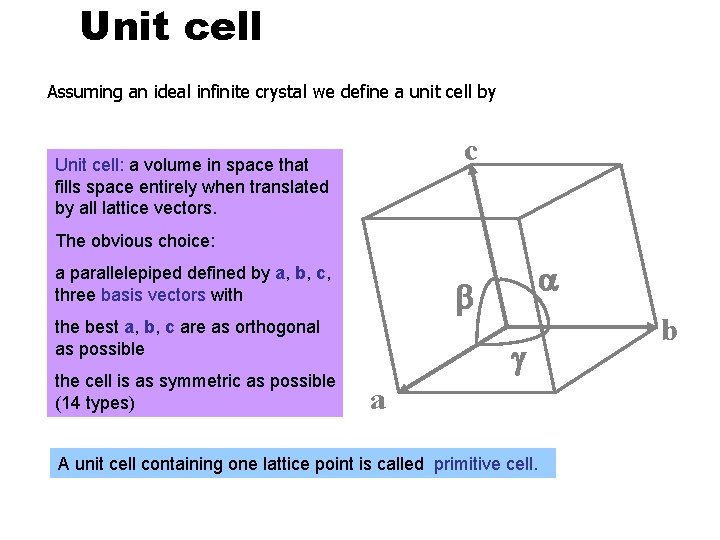
Unit cell Assuming an ideal infinite crystal we define a unit cell by c Unit cell: a volume in space that fills space entirely when translated by all lattice vectors. The obvious choice: a parallelepiped defined by a, b, c, three basis vectors with b the best a, b, c are as orthogonal as possible the cell is as symmetric as possible (14 types) a g a A unit cell containing one lattice point is called primitive cell. b
- Slides: 25