Lection 4 Phase diagrams of unary and binary

Lection 4 Phase diagrams of unary and binary systems v Phase diagrams for unary systems, relations with the Gibbs energy, critical point; v Binary systems, relations with the Gibbs energy of phases, lever rule, types of phase diagrams: eutectic, peritectic, eutectoid, congruent transformation, monotectic etc. , relations with microstructure development Phase diagram is a chart showing conditions (pressure, temperature, composition , volume etc. ) at which thermodynamically distinct phases occur and coexist in equilibrium. Phase diagram consists of lines of equilibrium or phase boundaries where change of phase assemblage occurs (one phase disappears or appears).
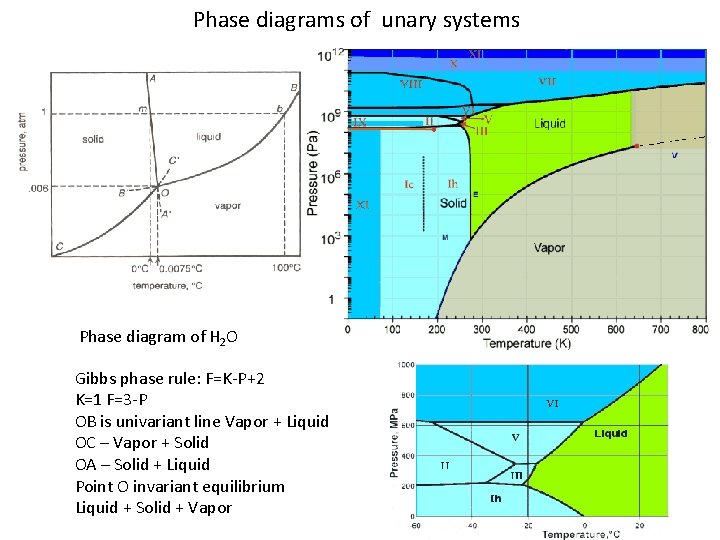
Phase diagrams of unary systems Phase diagram of H 2 O Gibbs phase rule: F=K-P+2 K=1 F=3 -P OB is univariant line Vapor + Liquid OC – Vapor + Solid OA – Solid + Liquid Point O invariant equilibrium Liquid + Solid + Vapor
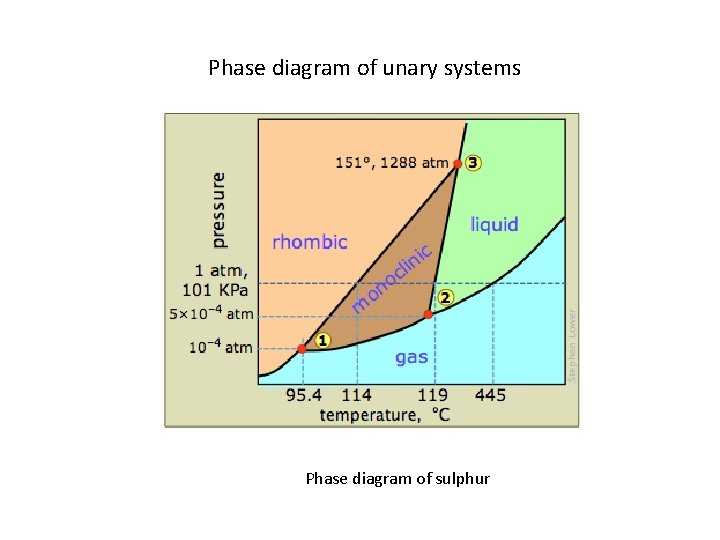
Phase diagram of unary systems Phase diagram of sulphur
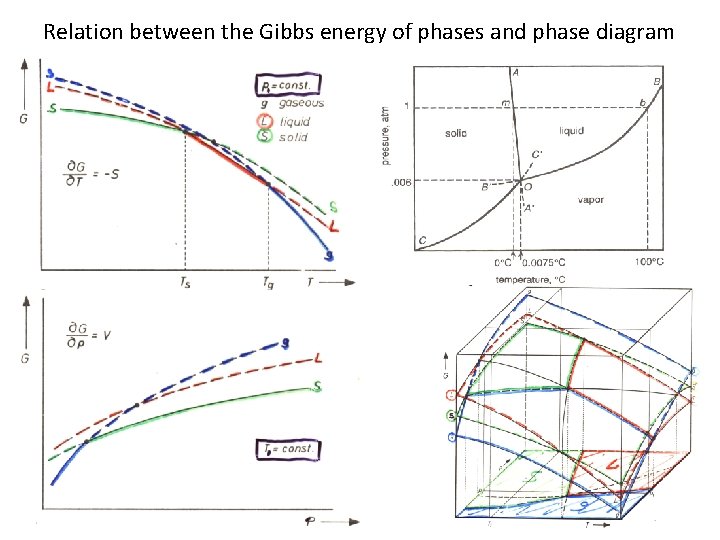
Relation between the Gibbs energy of phases and phase diagram
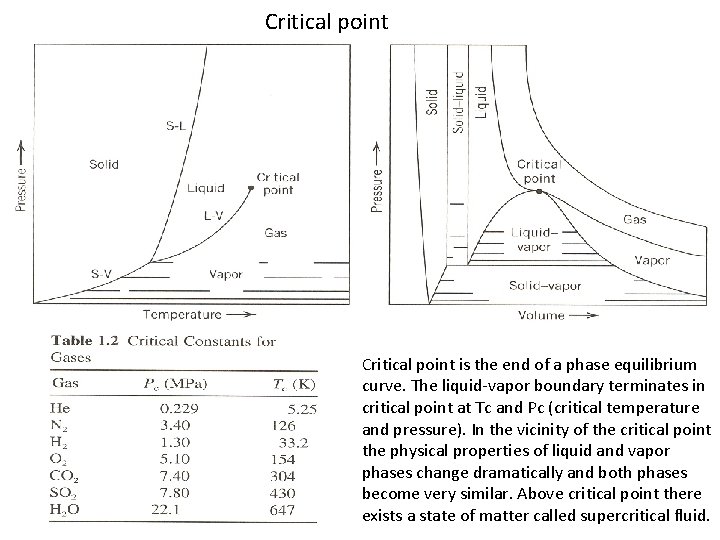
Critical point is the end of a phase equilibrium curve. The liquid-vapor boundary terminates in critical point at Tc and Pc (critical temperature and pressure). In the vicinity of the critical point the physical properties of liquid and vapor phases change dramatically and both phases become very similar. Above critical point there exists a state of matter called supercritical fluid.
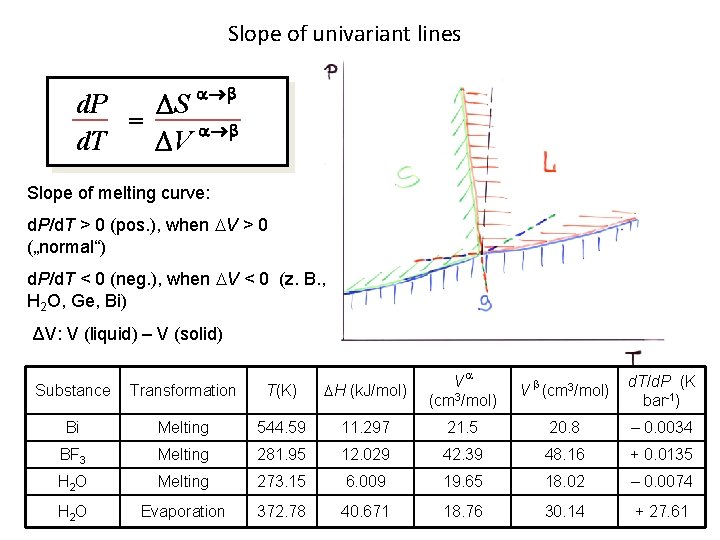
Slope of univariant lines d. P S = d. T V Slope of melting curve: d. P/d. T > 0 (pos. ), when V > 0 („normal“) d. P/d. T < 0 (neg. ), when V < 0 (z. B. , H 2 O, Ge, Bi) ΔV: V (liquid) – V (solid) Substance Transformation T(K) H (k. J/mol) V (cm 3/mol) Bi Melting 544. 59 11. 297 21. 5 20. 8 – 0. 0034 BF 3 Melting 281. 95 12. 029 42. 39 48. 16 + 0. 0135 H 2 O Melting 273. 15 6. 009 19. 65 18. 02 – 0. 0074 H 2 O Evaporation 372. 78 40. 671 18. 76 30. 14 + 27. 61 V (cm 3/mol) d. T/d. P (K bar-1)
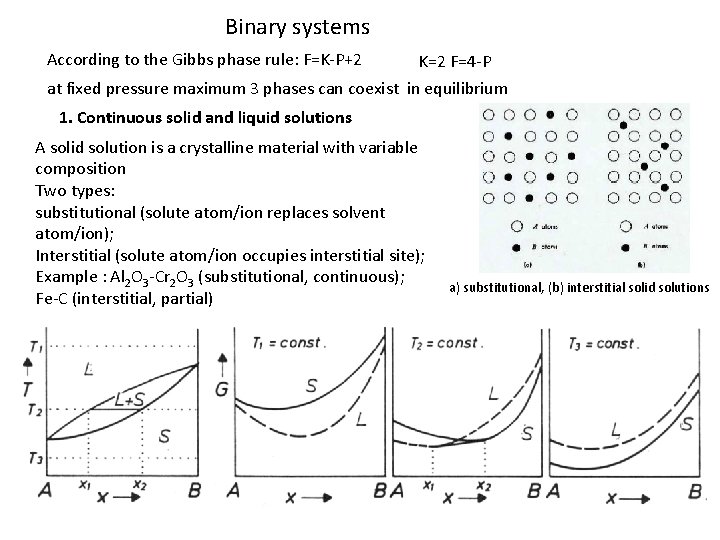
Binary systems According to the Gibbs phase rule: F=K-P+2 K=2 F=4 -P at fixed pressure maximum 3 phases can coexist in equilibrium 1. Continuous solid and liquid solutions A solid solution is a crystalline material with variable composition Two types: substitutional (solute atom/ion replaces solvent atom/ion); Interstitial (solute atom/ion occupies interstitial site); Example : Al 2 O 3 -Cr 2 O 3 (substitutional, continuous); Fe-C (interstitial, partial) a) substitutional, (b) interstitial solid solutions
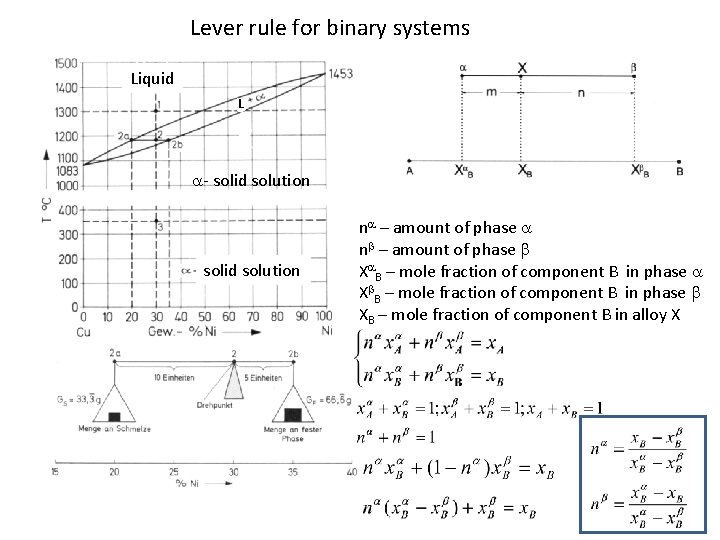
Lever rule for binary systems Liquid d L - solid solution n – amount of phase n X B – mole fraction of component B in phase XB – mole fraction of component B in alloy X
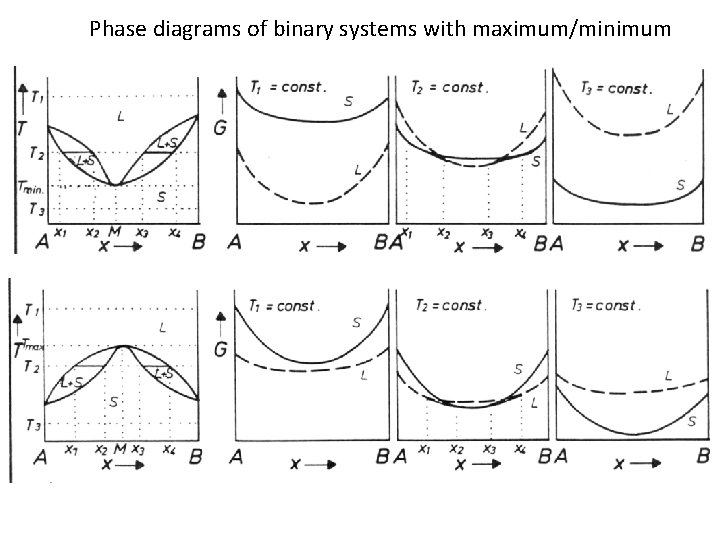
Phase diagrams of binary systems with maximum/minimum
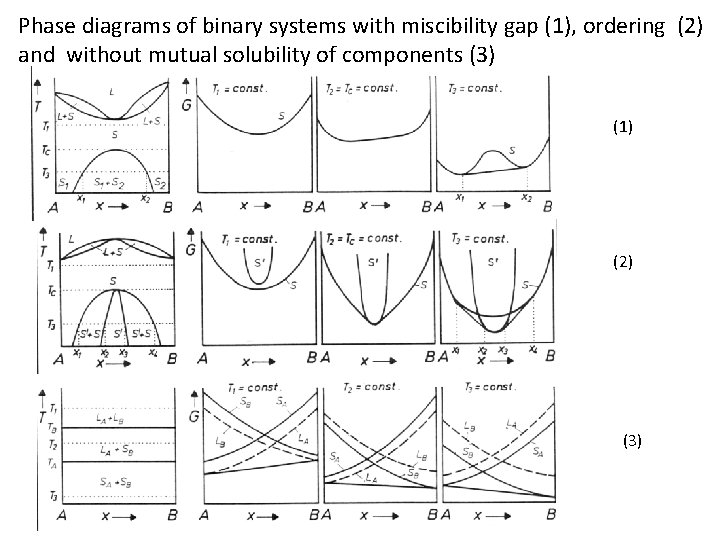
Phase diagrams of binary systems with miscibility gap (1), ordering (2) and without mutual solubility of components (3) (1) (2) (3)
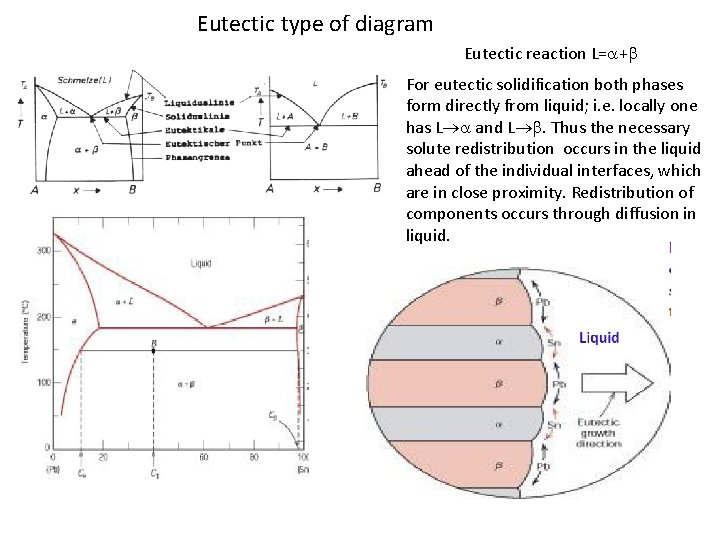
Eutectic type of diagram Eutectic reaction L= + For eutectic solidification both phases form directly from liquid; i. e. locally one has L and L . Thus the necessary solute redistribution occurs in the liquid ahead of the individual interfaces, which are in close proximity. Redistribution of components occurs through diffusion in liquid.

Different types of eutectic microstructures More complex arrangements of the two phases occurs if interface attachment kinetics are sluggish (usually encountered for crystals that grow from liquid with crystallographic facets). Then two solid phases grow independently from the melt with very little communication of the solute fields in the liquid. This leads to much coarser mixture of the two solid phases (divorced eutectic). a - globular eutectic b – acicular (needle-like) eutectic c - lamellar eutectic d – Chinese script
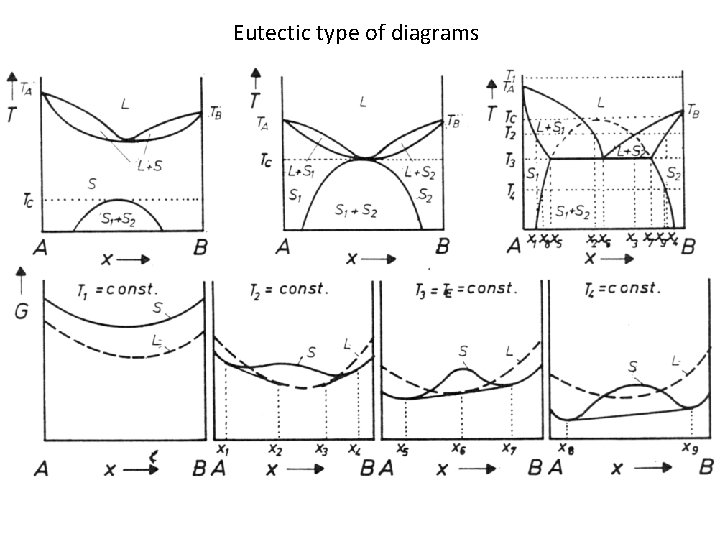
Eutectic type of diagrams
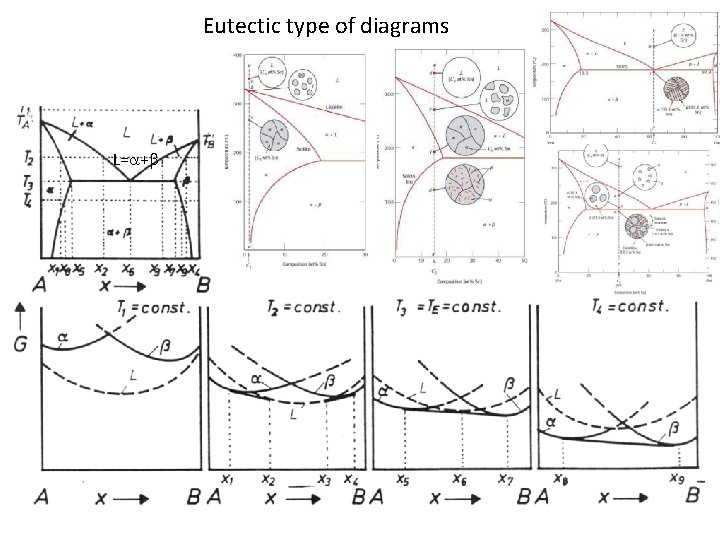
Eutectic type of diagrams L= +
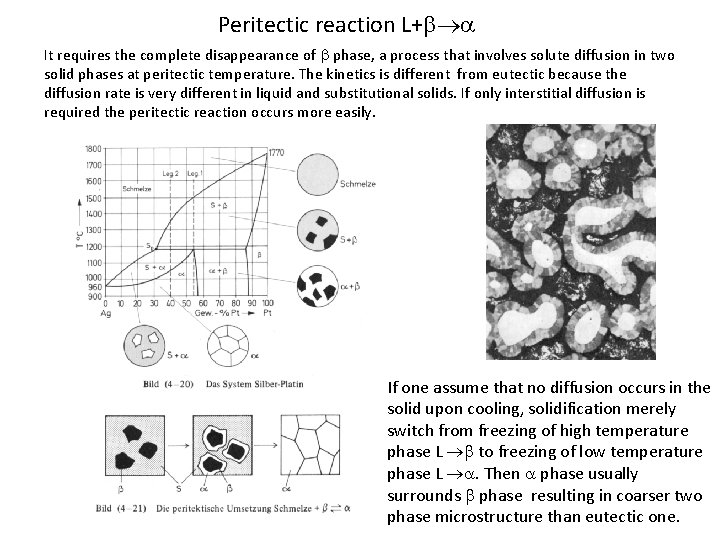
Peritectic reaction L+ It requires the complete disappearance of phase, a process that involves solute diffusion in two solid phases at peritectic temperature. The kinetics is different from eutectic because the diffusion rate is very different in liquid and substitutional solids. If only interstitial diffusion is required the peritectic reaction occurs more easily. If one assume that no diffusion occurs in the solid upon cooling, solidification merely switch from freezing of high temperature phase L to freezing of low temperature phase L . Then phase usually surrounds phase resulting in coarser two phase microstructure than eutectic one.
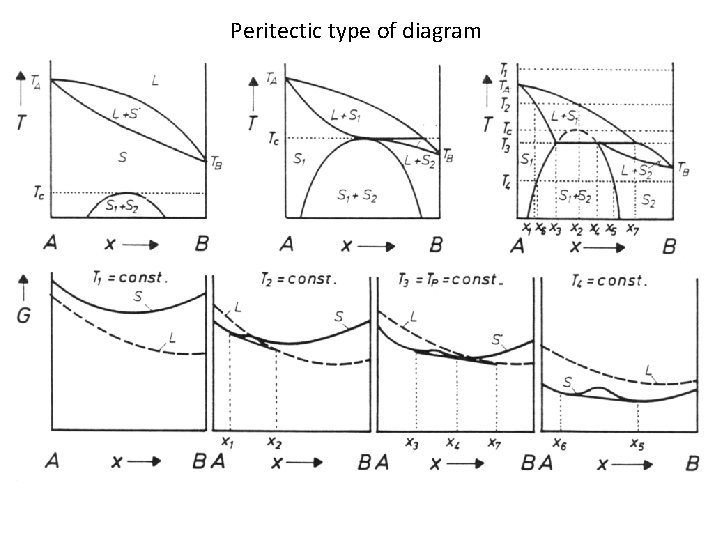
Peritectic type of diagram
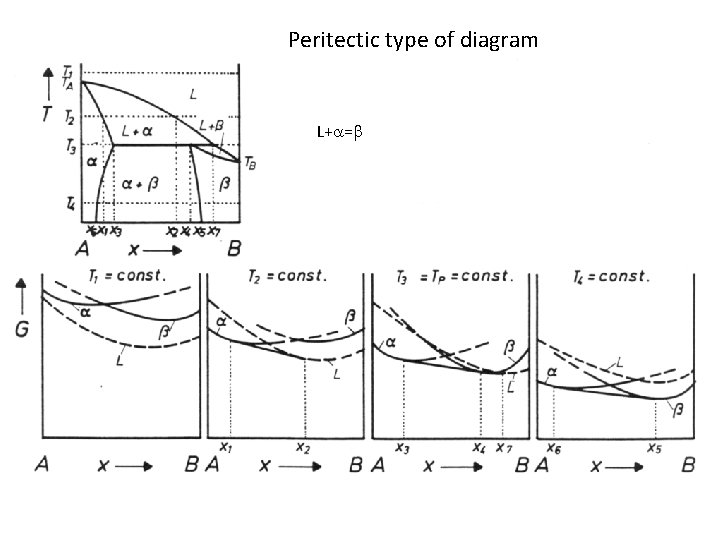
Peritectic type of diagram L+ =
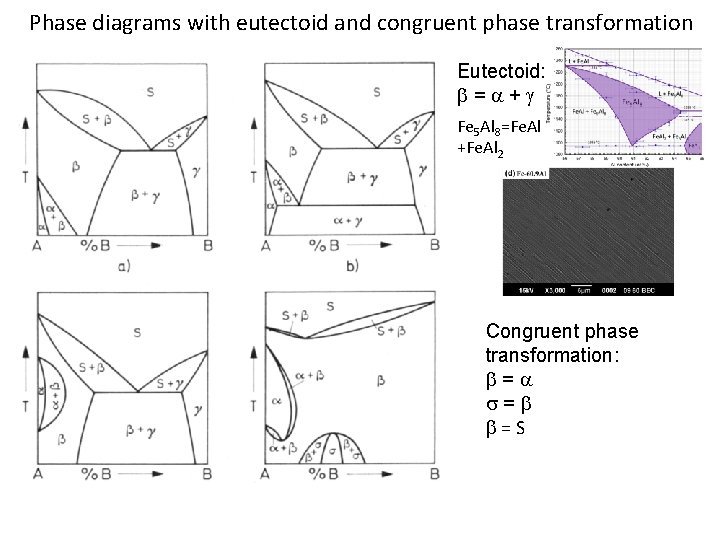
Phase diagrams with eutectoid and congruent phase transformation Eutectoid: = + Fe 5 Al 8=Fe. Al +Fe. Al 2 Congruent phase transformation: = s= =S
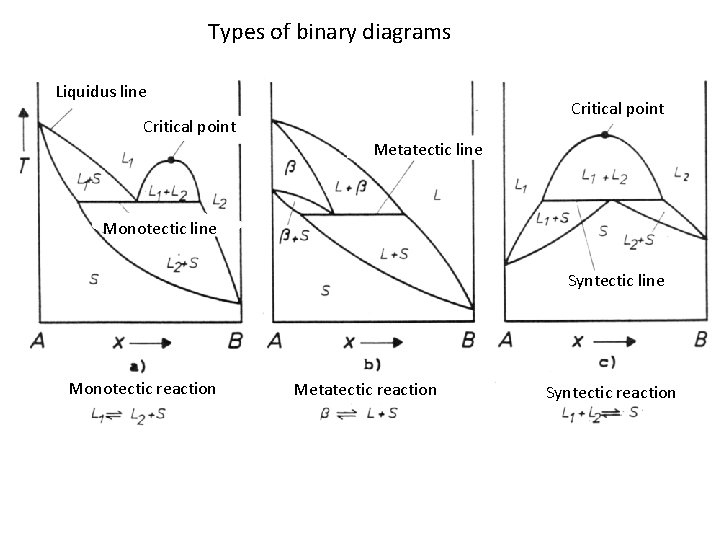
Types of binary diagrams Liquidus line Critical point Metatectic line Monotectic line Syntectic line Monotectic reaction Metatectic reaction Syntectic reaction
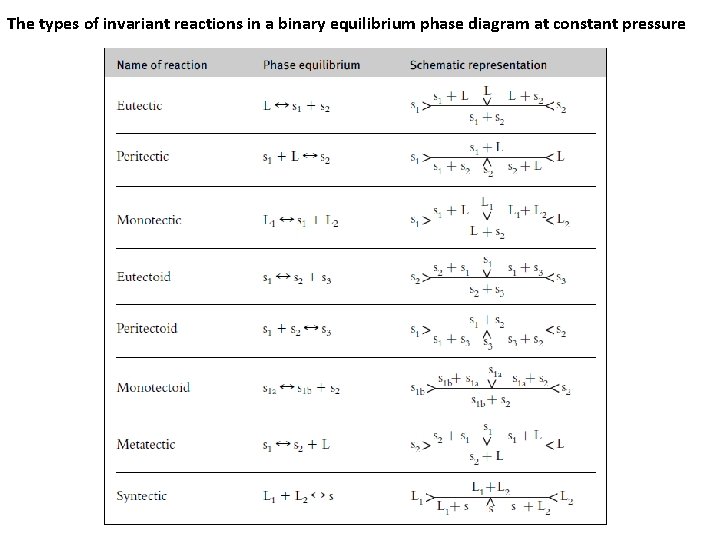
The types of invariant reactions in a binary equilibrium phase diagram at constant pressure
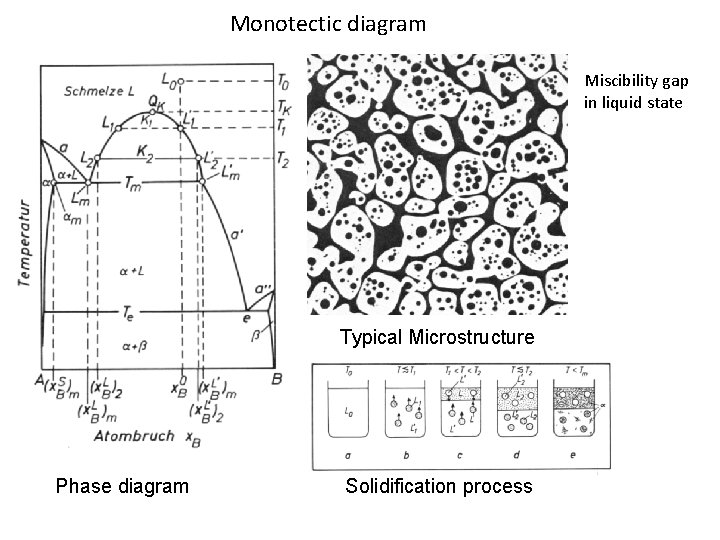
Monotectic diagram Miscibility gap in liquid state Typical Microstructure Phase diagram Solidification process
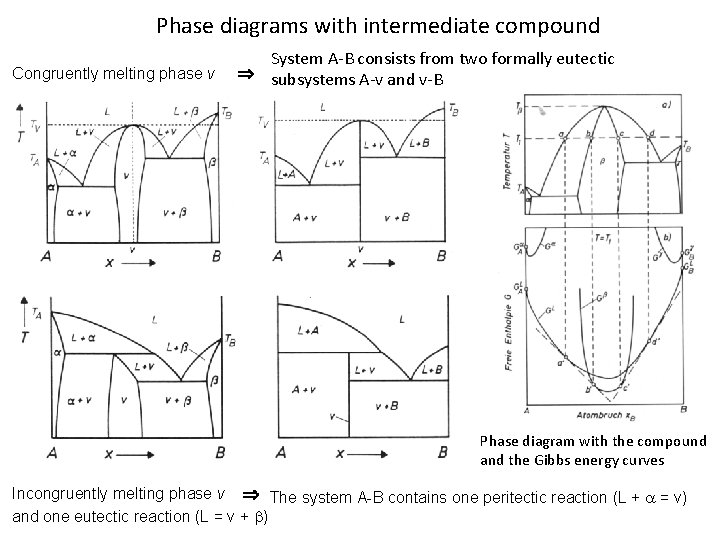
Phase diagrams with intermediate compound Congruently melting phase v System A-B consists from two formally eutectic subsystems A-v and v-B Phase diagram with the compound and the Gibbs energy curves Incongruently melting phase v The system A-B contains one peritectic reaction (L + = v) and one eutectic reaction (L = v + )
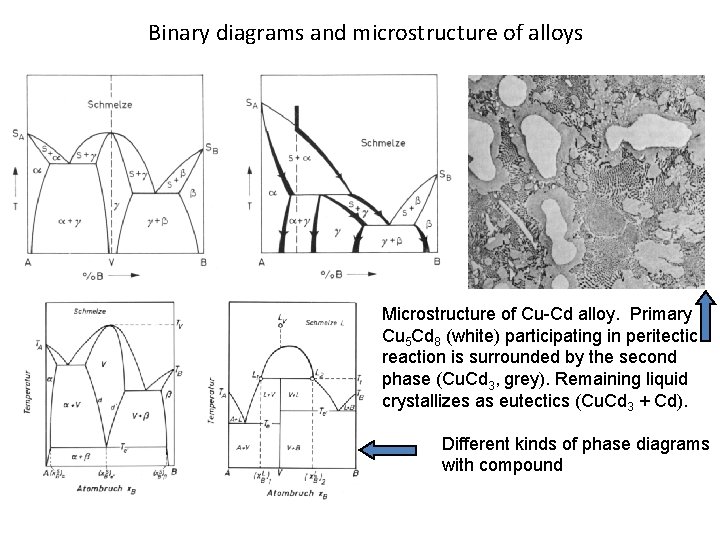
Binary diagrams and microstructure of alloys Microstructure of Cu-Cd alloy. Primary Cu 5 Cd 8 (white) participating in peritectic reaction is surrounded by the second phase (Cu. Cd 3, grey). Remaining liquid crystallizes as eutectics (Cu. Cd 3 + Cd). Different kinds of phase diagrams with compound
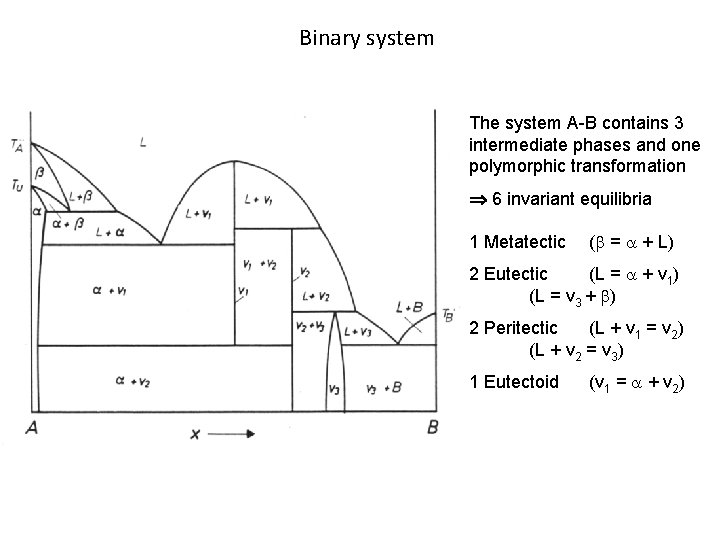
Binary system The system A-B contains 3 intermediate phases and one polymorphic transformation 6 invariant equilibria 1 Metatectic ( = + L) 2 Eutectic (L = + v 1) (L = v 3 + ) 2 Peritectic (L + v 1 = v 2) (L + v 2 = v 3) 1 Eutectoid (v 1 = + v 2)
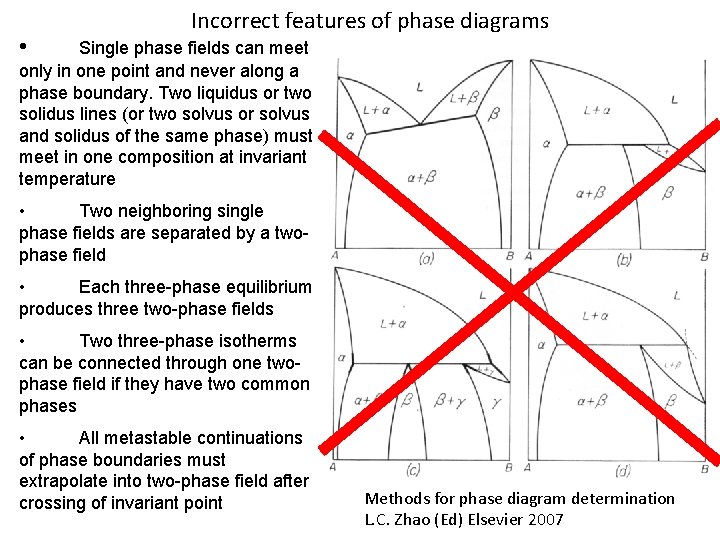
• Incorrect features of phase diagrams Single phase fields can meet only in one point and never along a phase boundary. Two liquidus or two solidus lines (or two solvus or solvus and solidus of the same phase) must meet in one composition at invariant temperature • Two neighboring single phase fields are separated by a twophase field • Each three-phase equilibrium produces three two-phase fields • Two three-phase isotherms can be connected through one twophase field if they have two common phases • All metastable continuations of phase boundaries must extrapolate into two-phase field after crossing of invariant point Methods for phase diagram determination L. C. Zhao (Ed) Elsevier 2007
- Slides: 25