Lect 2 Biomolecules Other organic compounds Other organic

Lect 2: Biomolecules & Other organic compounds

Other organic compounds

n Take a cheeseburger. . hamburger, covered with American (yellow) cheese on a hamburger bun. . . yummy!

n Now, if you made this cheeseburger with Swiss cheese and put it on slices of rye bread, n (or used Buffalo beef and no bun)

nyou’d end up with a “cheeseburger” but one that tasted totally different. . . nyou would notice that the substitutions affected the taste. . .

n Chemists make similar changes to organic compounds. . . n these changes produce compounds called “ substituted hydrocarbons” n A substituted hydrocarbon has had one or more of its hydrogen atoms or groups of atoms replaced by atoms or groups of atoms of other elements.

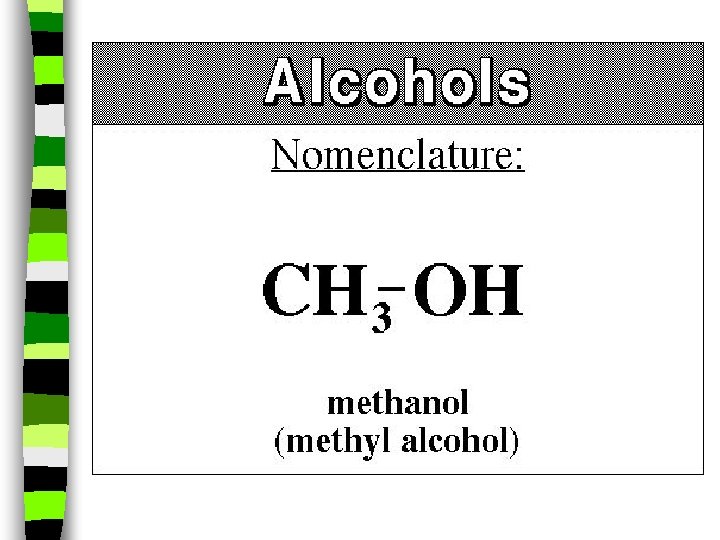
Alcohol: -OH n n n alcohol is the name of a family of compounds formed when a hydroxyl (-OH) group replaces one or more hydrogen atoms in a hydrocarbon. (ex: thanolis produced by sugar fermenting in corn, grains & fruits) Structure challenge: Isopropyl alcohol: The -OH is on the middle carbon of the 3 carbon chain Propyl alcohol: Has the -OH on the end C


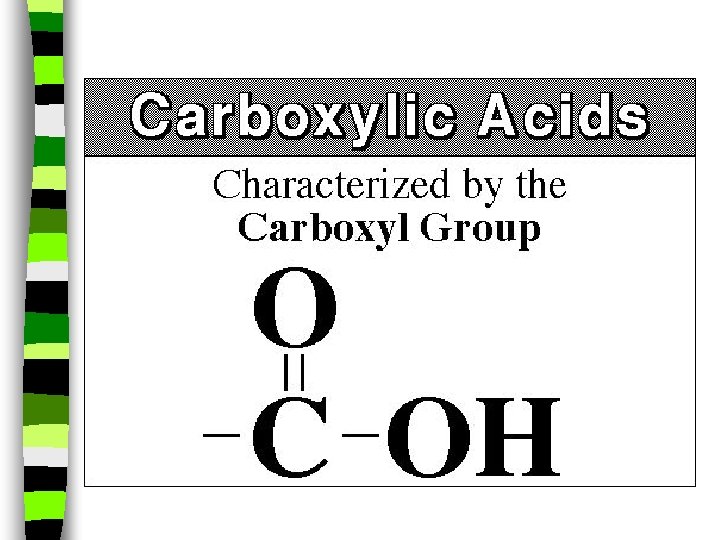
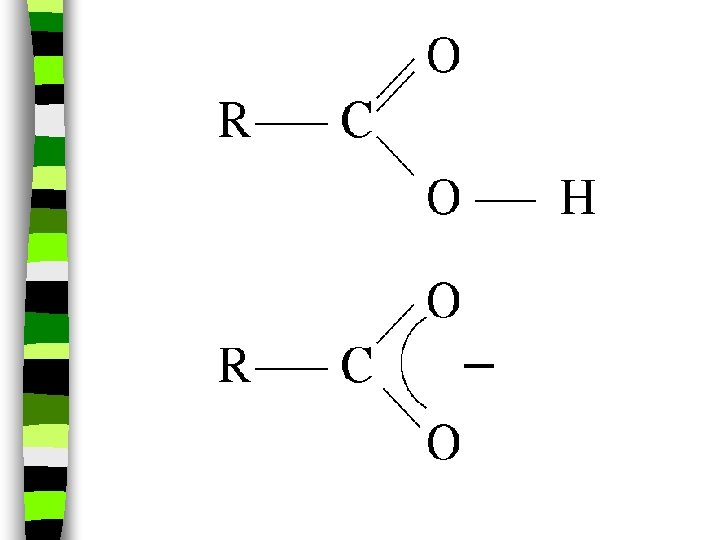
Carboxylic Acid: -COOH na carboxylic acid is formed when a -CH 3 group is replaced by a carboxyl (-COOH) group. n (The simplest carboxylic acid is methanic acid or formic acid which is made by ants and is injected into your skin when they bite you)
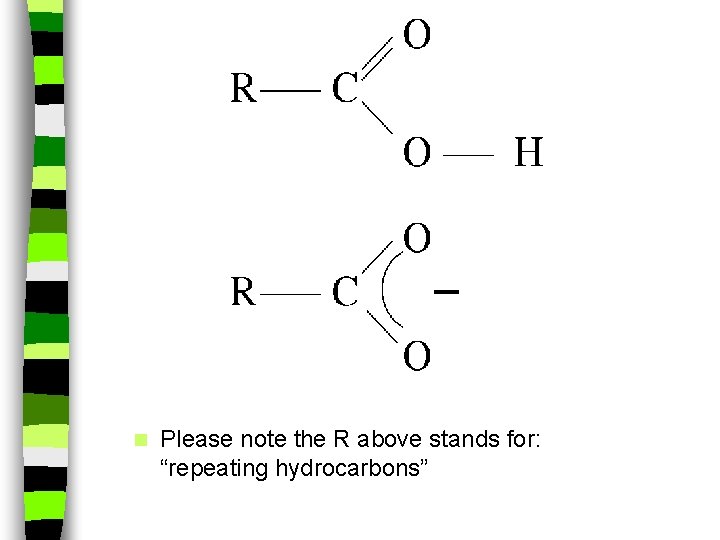
n Please note the R above stands for: “repeating hydrocarbons”

Amines: - NH 2 In this group, Nitrogen forms bonds with the carbon and hydrogen. n The amine group (-NH 2) replaces the hydrogen in the hydrocarbon. Mathylamine is the simplest amine. n (EX: novicane in the dentist’s office, caffeine in soft drinks. . . are all hydrocarbons substituted with nitrogen) n Example: Ethylmethylamine: CH 3 NHCH 2 CH 3 n

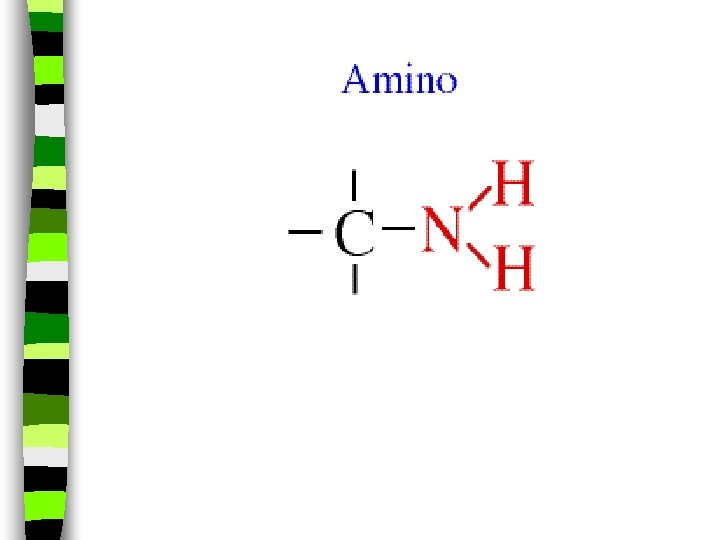

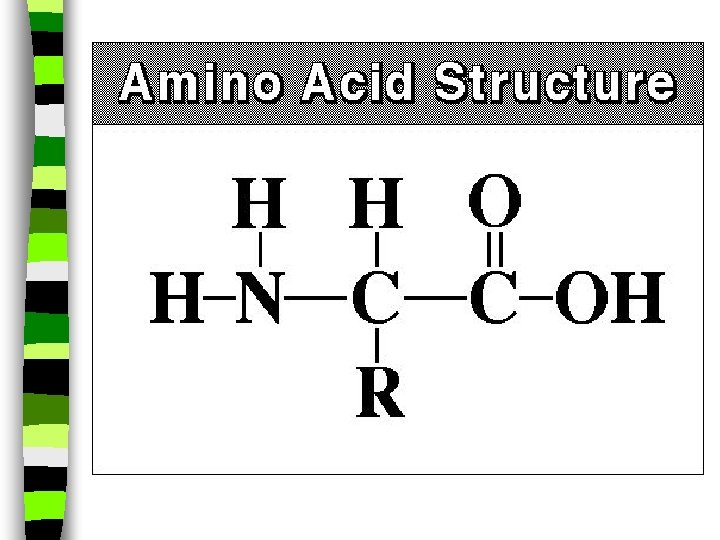
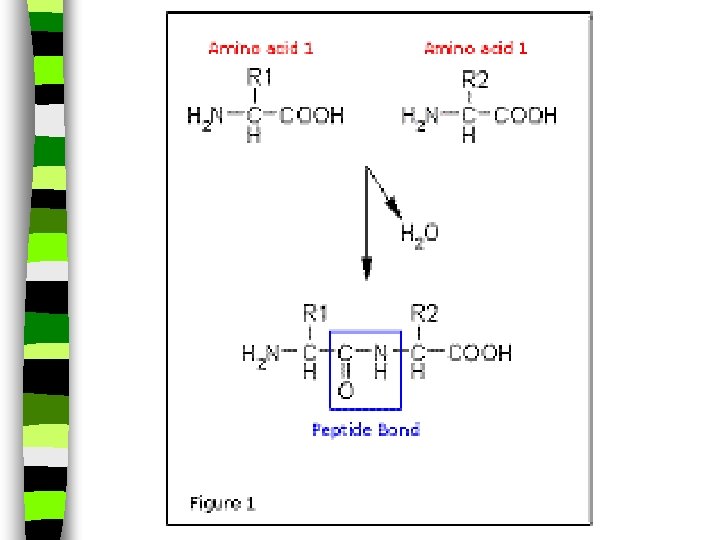
Amino Acids: n Amino acids have a -NH 2 group along as well as an acid group in it’s structure, and is a building block for proteins. n They also have both: -COOH and NH 2 groups ( a substituted hydrocarbon and more than one chemical group replacing its hydrogens at one time)

Polymers: n Milk, blood muscle, cassette tapes & athletic shoes are all made of organic compounds with very large molecules called Polymers. n Polymers are made up of smaller organic compounds that are linked together to form new bonds. n Polymers are also found in the biological compounds that make up living things.

polymers



What are Biomolecules? n n n Organic compounds made by living things Also called biochemicals Some are very large Biomolecules are based on the most important element to living organisms: Carbon There are thousands of different biomolecules but only 4 categories:

Click on a Category 1) Carbohydrates 2) Lipids 4) Nucleic Acids 3) Protein

1) Carbohydrate Facts n The simplest biomolecules n Carbohydrates are made of only 3 elements: – Carbon – Hydrogen – Oxygen n The word carbohydrate comes from the fact that these compounds have many carbon atoms bonded to hydroxide (OH) groups.

Carbohydrate Facts n Serve 2 main functions: 1. Source of chemical energy for cells in many living things. 2. Part of the structural material of plants n Come in all sizes, from small rings to long chains.

Two Types of Carbohydrates Simple Carbohydrates Complex Carbohydrates STARCH CELLULOSE SUGAR

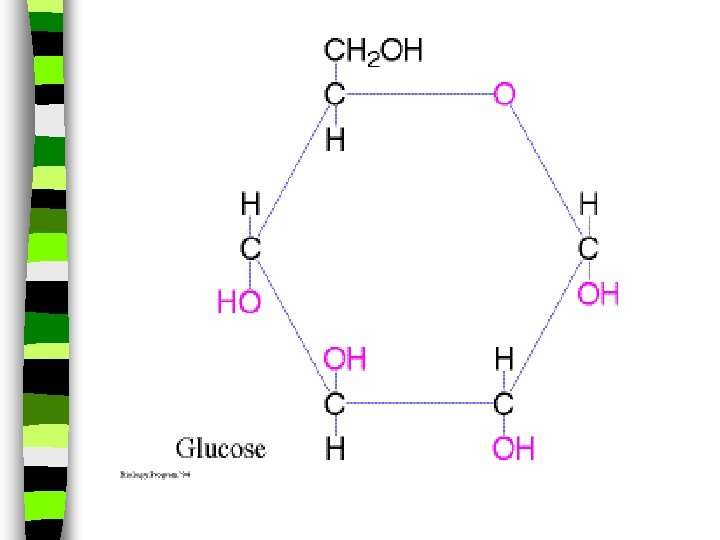
Sugar Break down quickly in the body n Provide a quick burst of energy or a “sugar rush” n Glucose is the most important & simplest sugar on Earth. n – Used in cells & created by photosynthesis – It comes in many forms

Examples of Simple Sugars

Starch Long chains of simple sugars joined together n These big molecules are called macromolecules, polysaccharides or polymers n Slower to break down in the body & provide energy for a longer period of time than regular sugars. n

Did you know that? Marathon runners, tri-athletes, cyclists like Lance Armstrong, and other endurance runners eat carbohydrates for weeks leading up to a big event. They call it “carbo-loading”. Even high school athletes occasionally have “pasta feeds” the night before a big game. What’s the point? As the athletes consume massive amounts of starch and pasta, the energy begins to store up in their body, saving itself for use during the event.

Starch Examples

Cellulose Unlike animal cells, plant cells have a cell wall. This cell wall is a touch protective layer made out of cellulose, a macromolecule (or BIG molecule). n The cell wall is a large part of vegetables such as lettuce & celery. n It also is what gives stems & wood strength. n

Cellulose Like starch, cellulose is made from chains of thousands of glucose molecules, but the difference is in how they’re linked. n Because of this small difference, your body can’t digest cellulose the same way it can starches & sugars. n

Carbohydrates: Carbohydrates are organic compounds in which there are twice as many hydrogen atoms as oxygen atoms. n Like fats, they contain carbon, hydrogen and oxygen. n However, in carbohydrates, the hydrogen and oxygen are present in a ratio of 2 hydrogen atoms to one oxygen atom. n This ratio is the same as water. n


Carbohydrates Summary: n The sugar in blood is called glucose. and has the formula C 6 H 12 O 6. n Sucrose C 12 H 22 O 11 is also a common sugars. n Glucose is found in honey and grapes. n Starches are larger molecules that occur naturally in wheat, rice and corn (a natural starch food is pasta and bread!!)


Brainpop: Carbohydrate Click to watch then answer the questions. Login as: mms 308, marshall

Quiz Yourself What is the difference between sugar, starch, and cellulose? Not Sure? Maybe you need to read the previous slides.
2) Lipids FACTS SATURATED & UNSATURATE D STRUCTU RE CHOLESTERO L

Lipid Facts Lipids include: Fats & Oils n Like carbohydrates most lipids are made of just carbon, hydrogen, and oxygen. n But unlike carbohydrates, fats are more complex & take much longer to break down. n So, fats are high-energy molecules that plants and animals use to store energy in reserves for longer periods. n

Lipid Facts Plants store energy in oil, like olive oil, corn oil, or peanut oil. n An oil is a fat that is liquid at room temperature. n
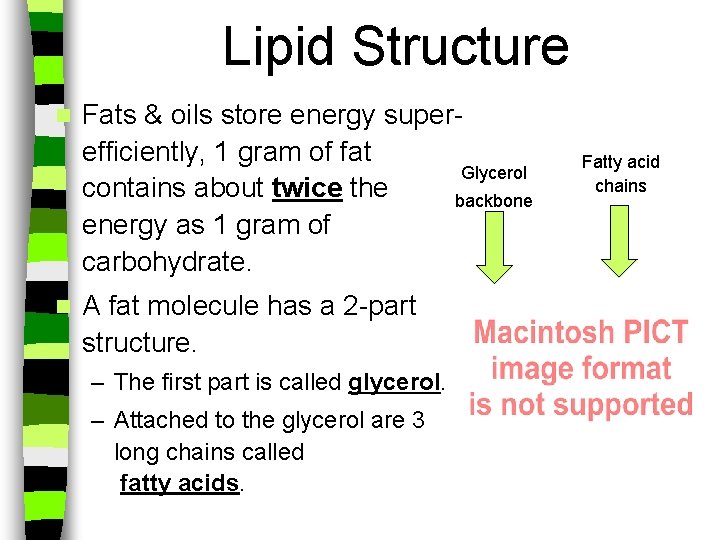
Lipid Structure n n Fats & oils store energy superefficiently, 1 gram of fat Glycerol contains about twice the backbone energy as 1 gram of carbohydrate. A fat molecule has a 2 -part structure. – The first part is called glycerol. – Attached to the glycerol are 3 long chains called fatty acids. Fatty acid chains

Lipids: n What do butter, margarine and oil in salad dressing have in common? ? n They are all lipids. n Lipids are organic compounds that feel greasy and will not dissolve in water (they are insoluable) n Fats, oils. waxes, etc. , make up this group.

Lipids contain the same elements: Carbon, H, O that carbohydrates do, but they are in different proportions. n Lipids are a more concentrated source of energy than carbohydrates. n They provide twice as much energy per gram as carbohydrates. n

Saturated vs Unsaturated Fats: Fats and oils are classified as saturated and unsaturated according to the types of bonds in their carbon chains. n Saturated fats such as cheese, whipped cream, ice cream only contain single bonds between carbons. n Unsaturated fats: contain one or more double bonds between atoms n

Saturated and Unsaturated n Saturated fats – only single bonds in the carbon chain – Most animal fats – “Bad” fats – Diets high in saturated fat are linked to heart disease n Unsaturated fats – one or more double bonds in the carbon chain – Most oils from plants – “Good” fats

Quiz Yourself List 2 examples of saturated fats and 2 examples unsaturated fats.

Fat Examples Saturated Fats Unsaturated Fats

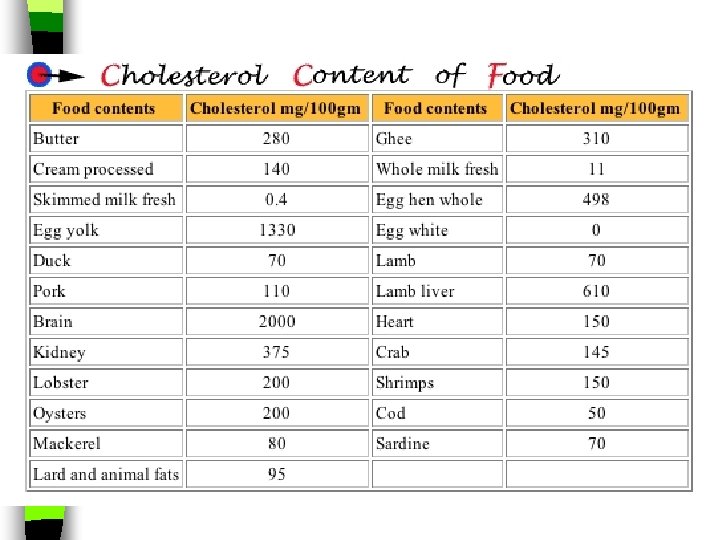
Cholesterol n Cholesterol is another lipid in cell membranes n It is also needed to make hormones like adrenaline n Your body makes the cholesterol that it needs, but it is also found in many foods that come from animals, like meat and eggs. n Although you need cholesterol, eating too much of it can block arteries and lead to heart disease.

Brainpop: Fats Click to watch then answer the questions. Login as: mms 308, marshall

Quiz Yourself How are lipids different from carbohydrates? Not Sure? Maybe you need to read the previous slide.

Video 6: Fueling the Body: Carbohydrates & Fats Click to watch then answer the questions.

3) Proteins FACTS AMINO ACIDS ENZYMES PROTEINS IN THE DIET VITAMINS

Protein Facts n n n Proteins are big molecules called macromolecules and are made of smaller molecules called amino acids Made of carbon, hydrogen, oxygen, nitrogen, sulfur, & some other elements There at least 100, 000 proteins in your body Each has a different structure that gives it a specific job. There are 4 types of structure, including coils & curls.

Proteins: n Milk and fish contain protein, a particular kind of hydrocarbon that is necessary for all living cells. n Proteins are polymers formed by linking together monomers called amino acids. n Think of proteins as being like a word. Amino acids are the letters in that word. n Rearranging the letters makes words with different meanings.

Amino Acids n Proteins are in your muscles, hair, every living cell in your body. n 8 of the __20__ amino acids used by our bodies are absolutely essential for us to function properly, and our bodies DON'T make them. _12__ amino acids are made in our bodies, but the other _8_ must be obtained by eating _foods__that contain them.
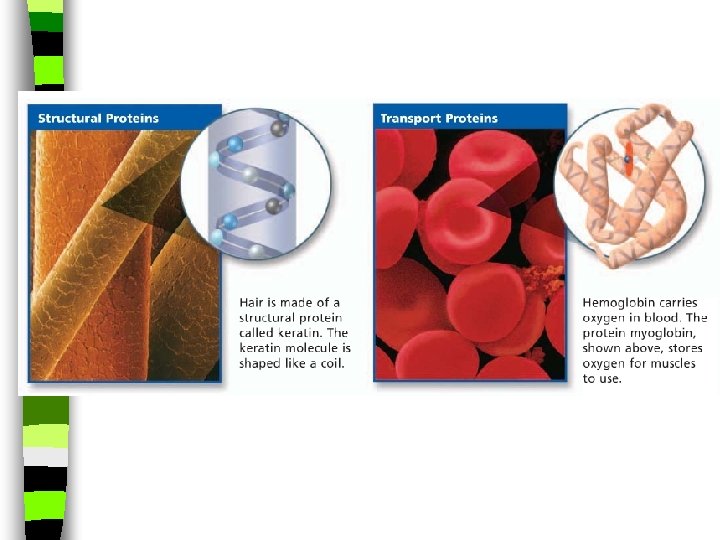

Proteins in the Diet It’s important to have lots of protein in your diet! n Proteins in foods such as meats, soybeans, & nuts are broken down into amino acids. n Without protein, your body can’t function perfectly. . n This is why it’s important for vegetarians to find protein from non-animal sources. n

Enzymes n n n Some proteins curl up into a shape like a ball of enzymes. An enzyme is a special protein & a catalyst for a chemical reaction in living things. Catalysts speed up the rate of a reaction. Enzymes are needed for many chemical reactions in your body. Without them, these reactions would occur too slowly to keep you alive.

Nucleic Acids Largest & most complex bio-molecule n Includes: – DNA : deoxyribonucleic acid – RNA : ribonucleic acid Huge, complex carbon-based molecules. . n Their Job: Contain information that cells use to make proteins n Made of: – Carbon Hydrogen Oxygen Nitrogen Phosphorous n

Protein Synthesis n Every cell in your body has a complete set of nucleic acids. n The process of making proteins from amino acids is called protein synthesis. n How does protein synthesis work?

DNA Facts Deoxyribonucleic acid n One of the largest molecules n A single DNA molecule has more than 1 million atoms. n

DNA Structure A DNA molecule is a twisted ladder or double helix n The sides of the ladder are made of: n – 5 carbon sugar molecules called deoxyribose and – phosphate groups n The “rungs” of the ladder are made of: – Nitrogen bases
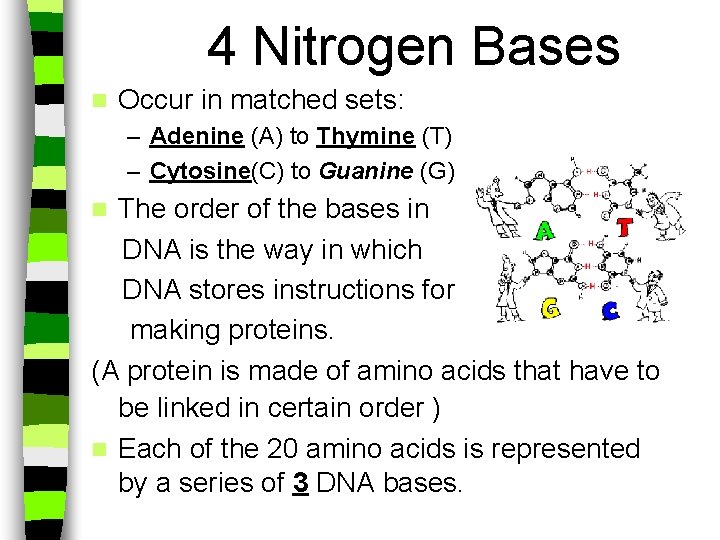
4 Nitrogen Bases n Occur in matched sets: – Adenine (A) to Thymine (T) – Cytosine(C) to Guanine (G) The order of the bases in DNA is the way in which DNA stores instructions for making proteins. (A protein is made of amino acids that have to be linked in certain order ) n Each of the 20 amino acids is represented by a series of 3 DNA bases. n


Vitamins n n n Most of the chemical needed for life can by made by your own body, like proteins. However, there are certain chemicals that your body does not automatically make. We call these vitamins & minerals. Important daily vitamins & minerals include calcium, Vitamin A, Vitamin C, Vitamin D, and a whole bunch of B vitamins. The only place to get these: food!!


Vitamin C Ascorbic acid, or vitamin C, is needed for several important processes in your brain & nervous system. n Scurvy results from a lack of vitamin C in your diet. n It causes of spotting on the skin, spongy gums, and bleeding membranes, and can eventually lead to death. n The British Royal Navy were among the first to discover this vitamin deficiency, when they noticed their sailors would get sick without fresh fruits & vegetables. n

Brainpop: DNA Click to watch then answer the questions. Login as: mms 308, marshall

Brainpop: Body Chemistry Click to watch then answer the questions. Login as: mms 308, marshall
QUIZ TIME! n Answer the quiz questions on your lecture notes first and then check your answers!

Review: n 1. Carbon’s unique ability to form four covalent bonds with other atoms enables it to make a huge number of compounds.

n The structure of its compound determines its properties in the following ways: n shorter hydrocarbons are lighter molecules. n In general, they have low boiling points and the evaporate and burn easier. n Longer hydrocarbons are heavy molecules and exist as solids or liquids at room temp

n 2. Hydrocarbons can be composed of hydrogen and carbon alone, or other chemical groups may be substituted fro hydrogen on the molecule to form new compounds, as with vitamin C. Three additional types of substituted hydrocarbons include:

alcohol: hydroxyl group (-OH) n replaces 1 or more hydrogen atoms, produced naturally by sugar fermentation in fruit and grain. n
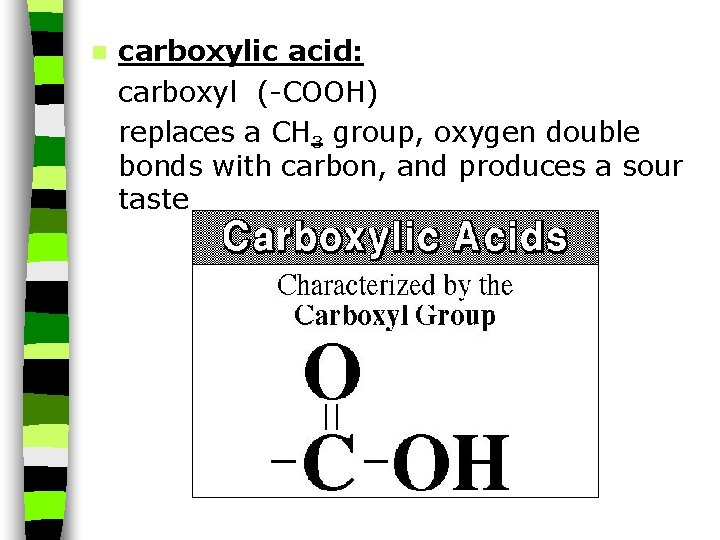
n carboxylic acid: carboxyl (-COOH) replaces a CH 3 group, oxygen double bonds with carbon, and produces a sour taste

n amine: amine group (-NH 2) replaces a hydrogen, found in caffeine, novocaine, and in some vitamins.

n 3. food providing protein: milk, fish, meat, poultry n food providing carbohydrates: pasta, bread, vegetables, sugar n food providing lipids: butter, margarine, oils, solid shortening

n 4. Some examples of biological compounds at work: n proteins: make up muscles n Glucose : found in grapes and honey n Vegetables contain carbohydrates in the form of starch

n 5. What do they do for our bodies: n proteins: provide growth & renewal n Carbohydrates and lipids: provide energy
- Slides: 85