LEC DEVICE Group Members Kevin Mihelc Amine Hallab

LEC DEVICE Group Members: Kevin Mihelc Amine Hallab Jen Bacior Hiroki Meguro Advisors: Kelly Dympna, MD John Patzer, Ph. D 1 February 2005

Project Overview • Intended Use: Open Liver Biopsy • Combines 3 Functions 1. Removal of Tissue 2. Cauterization 3. Biopsy Tissue Protection
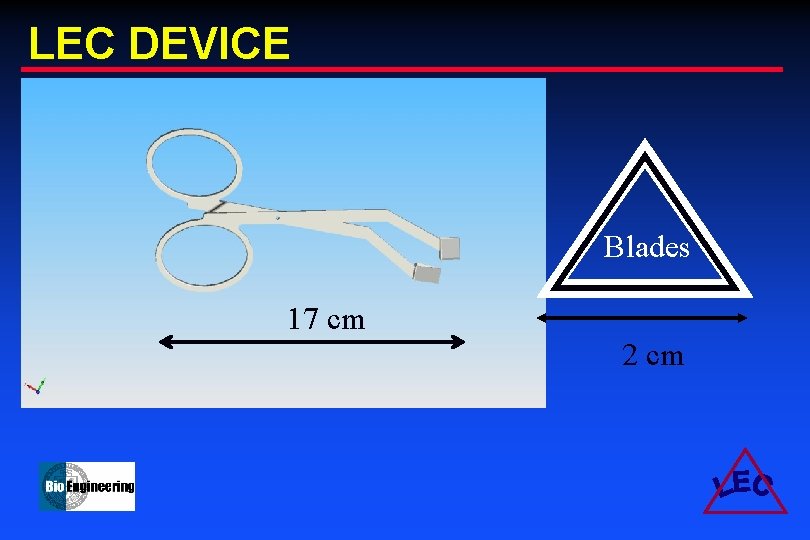
LEC DEVICE Blades 17 cm 2 cm

Individual Responsibilities • Blade Redesign • Testing Protocol • Design History File Update

Secondary Blade Design • Remove Closed Faces 1. Facilitate Easy Removal of Tissue Sample 2. Simplify Fabrication Process • Round Triangular Edges 1. Wedge Point is difficult to cauterize 2. Simplify Fabrication by removing joints

Testing Protocol • Preliminary Testing Using Cosmos. Works - Examine stresses subjected on device • Discarded Livers from Animal Research Study 1. 2. 3. 4. Cutting Ability Cauterization Effectiveness Cauterization Time Heat and Current Protection

Testing Protocol (Cont. ) • Sterilization Test -Multiple Autoclave Cleansings • User Survey 1. Ease of Operation 2. Comfortableness 3. Comparison to Previous Biopsy Technique

Design History File - Revise and Update DHF to ensure proper representation of the LEC tool

Upcoming Milestones • Functional Computer Prototype – 2 weeks • Submit for Fabrication – 2 weeks • Full Testing Completed – 4/7/05 • Revised Design History File – 4/7/05

Acknowledgements • Mark Gartner • Department of Bio. Engineering • LEC Group Members • John Patzer, Ph. D • Kelly Dympna, MD • Stephan Safta and Richard Miller
- Slides: 10