Lec 6 4 th stage Organic Pharmaceutical Chemistry


- Slides: 16

Lec 6 4 th stage Organic Pharmaceutical Chemistry III 2018 -2019 Assist prof. Dr. Rita Sabah Elias College of Pharmacy, university of Basrah Textbook of Organic medicinal and pharmaceutical chemistry Wilson and Gisvold’s

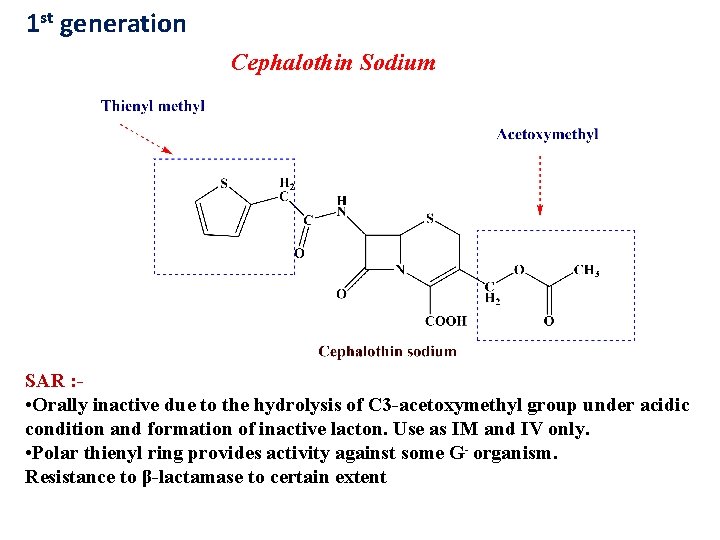
1 st generation Cephalothin Sodium SAR : • Orally inactive due to the hydrolysis of C 3 -acetoxymethyl group under acidic condition and formation of inactive lacton. Use as IM and IV only. • Polar thienyl ring provides activity against some G- organism. Resistance to β-lactamase to certain extent

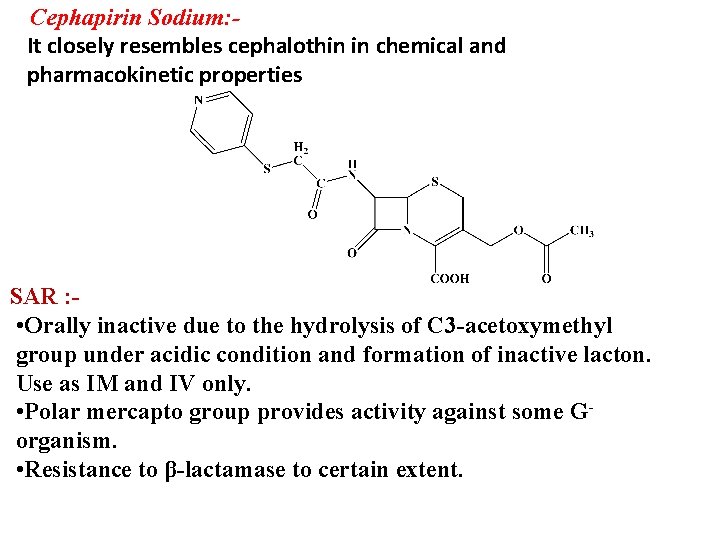
Cephapirin Sodium: It closely resembles cephalothin in chemical and pharmacokinetic properties SAR : • Orally inactive due to the hydrolysis of C 3 -acetoxymethyl group under acidic condition and formation of inactive lacton. Use as IM and IV only. • Polar mercapto group provides activity against some Gorganism. • Resistance to β-lactamase to certain extent.

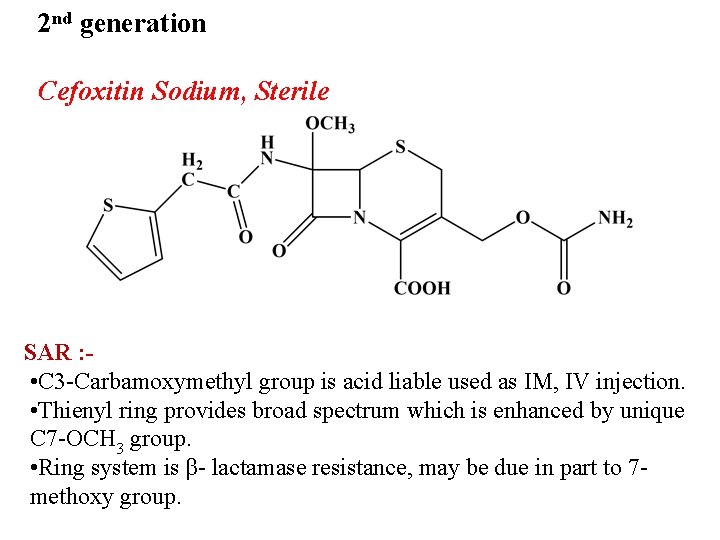
2 nd generation Cefoxitin Sodium, Sterile SAR : • C 3 -Carbamoxymethyl group is acid liable used as IM, IV injection. • Thienyl ring provides broad spectrum which is enhanced by unique C 7 -OCH 3 group. • Ring system is β- lactamase resistance, may be due in part to 7 methoxy group.

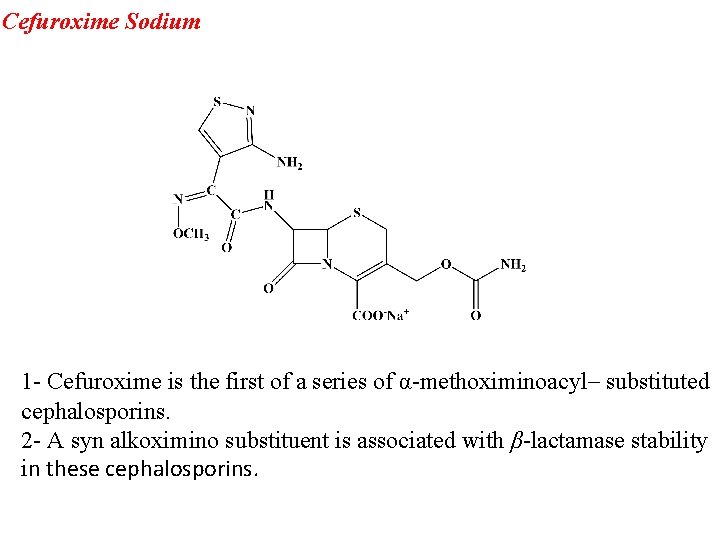
Cefuroxime Sodium 1 - Cefuroxime is the first of a series of α-methoximinoacyl– substituted cephalosporins. 2 - A syn alkoximino substituent is associated with β-lactamase stability in these cephalosporins.

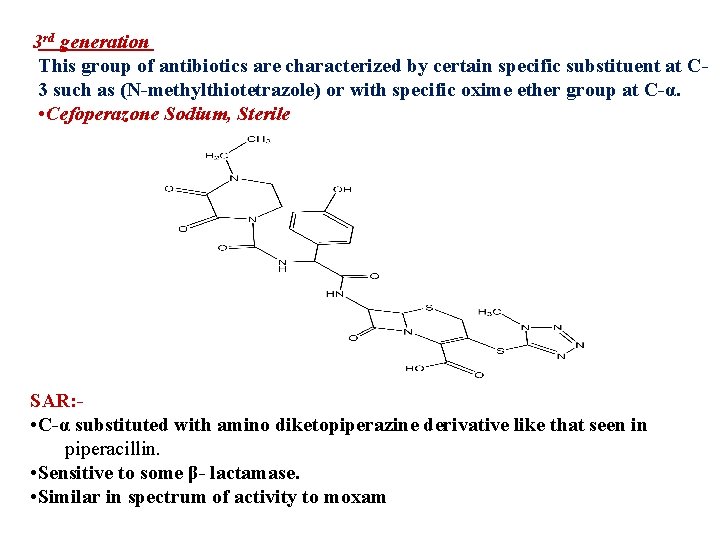
3 rd generation This group of antibiotics are characterized by certain specific substituent at C 3 such as (N-methylthiotetrazole) or with specific oxime ether group at C-α. • Cefoperazone Sodium, Sterile SAR: • C-α substituted with amino diketopiperazine derivative like that seen in piperacillin. • Sensitive to some β- lactamase. • Similar in spectrum of activity to moxam

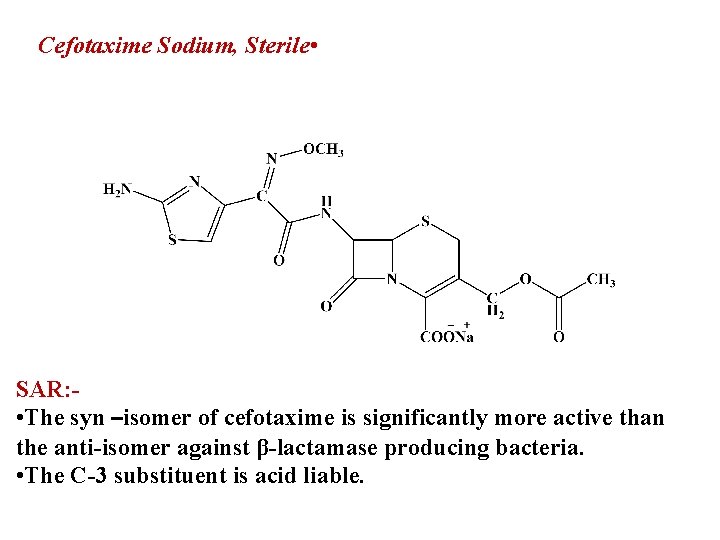
Cefotaxime Sodium, Sterile • SAR: • The syn –isomer of cefotaxime is significantly more active than the anti-isomer against β-lactamase producing bacteria. • The C-3 substituent is acid liable.

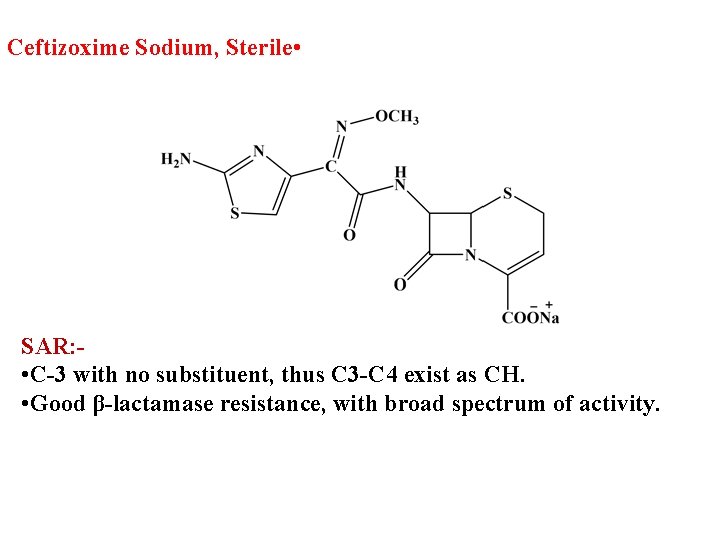
Ceftizoxime Sodium, Sterile • SAR: • C-3 with no substituent, thus C 3 -C 4 exist as CH. • Good β-lactamase resistance, with broad spectrum of activity.

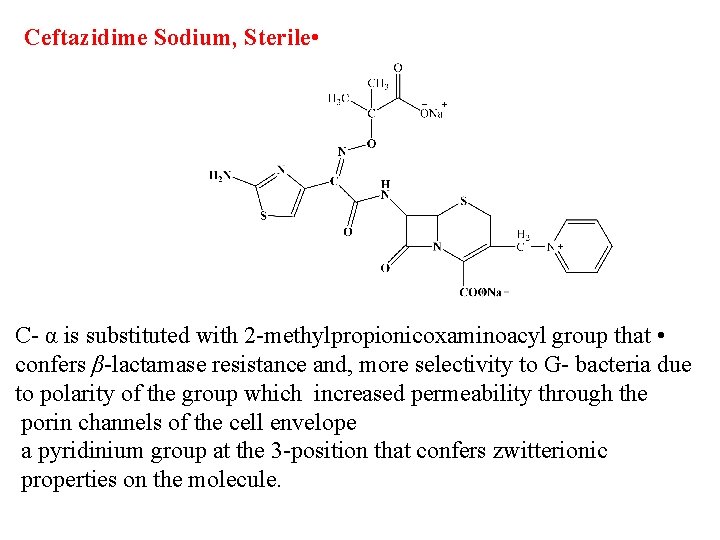
Ceftazidime Sodium, Sterile • C- α is substituted with 2 -methylpropionicoxaminoacyl group that • confers β-lactamase resistance and, more selectivity to G- bacteria due to polarity of the group which increased permeability through the porin channels of the cell envelope a pyridinium group at the 3 -position that confers zwitterionic properties on the molecule.

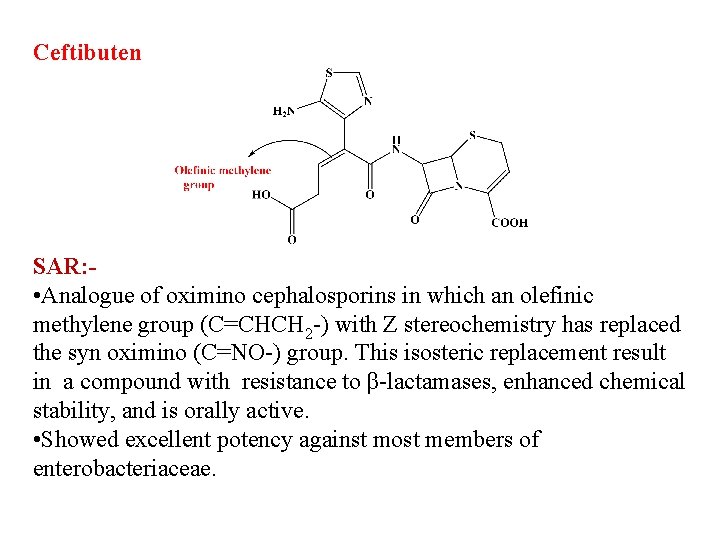
Ceftibuten SAR: • Analogue of oximino cephalosporins in which an olefinic methylene group (C=CHCH 2 -) with Z stereochemistry has replaced the syn oximino (C=NO-) group. This isosteric replacement result in a compound with resistance to β-lactamases, enhanced chemical stability, and is orally active. • Showed excellent potency against most members of enterobacteriaceae.

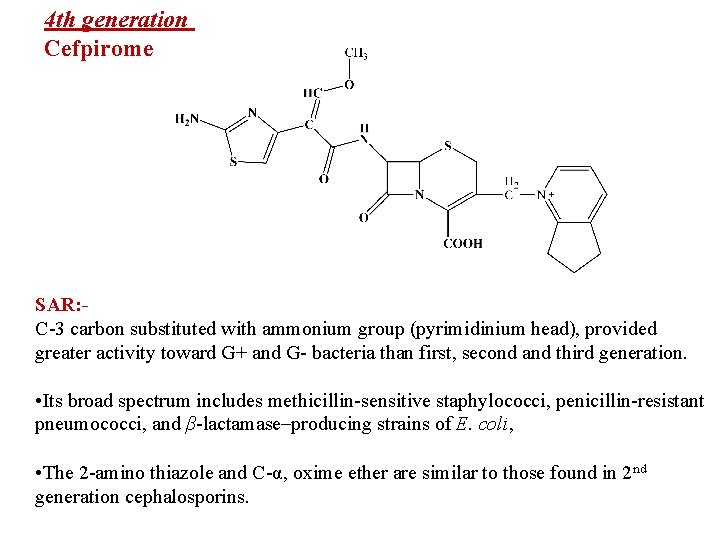
4 th generation Cefpirome SAR: C-3 carbon substituted with ammonium group (pyrimidinium head), provided greater activity toward G+ and G- bacteria than first, second and third generation. • Its broad spectrum includes methicillin-sensitive staphylococci, penicillin-resistant pneumococci, and β-lactamase–producing strains of E. coli, • The 2 -amino thiazole and C-α, oxime ether are similar to those found in 2 nd generation cephalosporins.

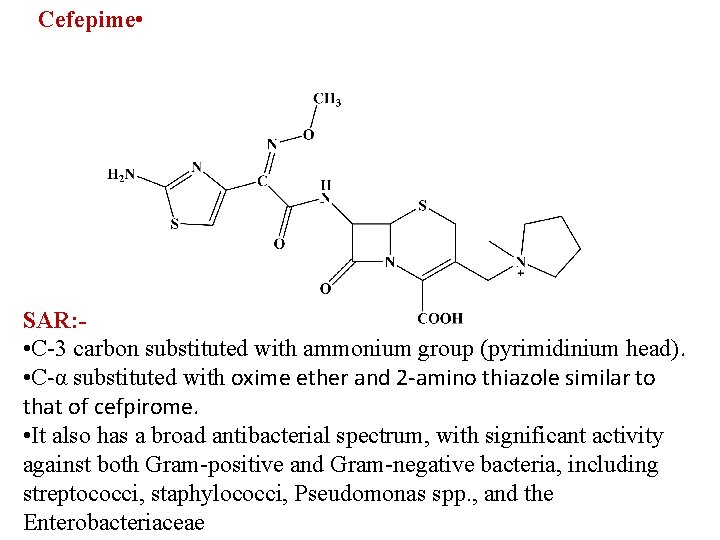
Cefepime • SAR: • C-3 carbon substituted with ammonium group (pyrimidinium head). • C-α substituted with oxime ether and 2 -amino thiazole similar to that of cefpirome. • It also has a broad antibacterial spectrum, with significant activity against both Gram-positive and Gram-negative bacteria, including streptococci, staphylococci, Pseudomonas spp. , and the Enterobacteriaceae

General SAR for Cephalosporins • C-3, acetoxymethyl liable to acidic conditions C-3 carbamate, relatively stable to acidic condition. C-3 = H, CH 3, Cl, C=CH 2 -CH=CHCH 2, -CH 2 -O-CH 3→ provide oral activity, • or acid stability. • C-7= α-OCH 3, provide greater resistance to β-lactamase. • C-α= oxime ether broaden spectrum of activity and β-lactamase resistance. • C-α= oxime ether with acidic founctional group ( carboxyl) increase spectrum of activity in a particular towards G- microorganism. • C-3 thiomethyl heterocyclic group usually are of second generation cephalosporine. • C-3 methylene immonium ion provide greater stability to β-lactamase and broader spectrum of activity and most of these antibiotics are either third or fourth generation cephalosporins. • C-α aryl or aryl heterocyclic contribute to the spectrum of activity, in general aryl heterocyclic classified within 2 nd, 3 rd, and recently 4 th generation cephalosporins.

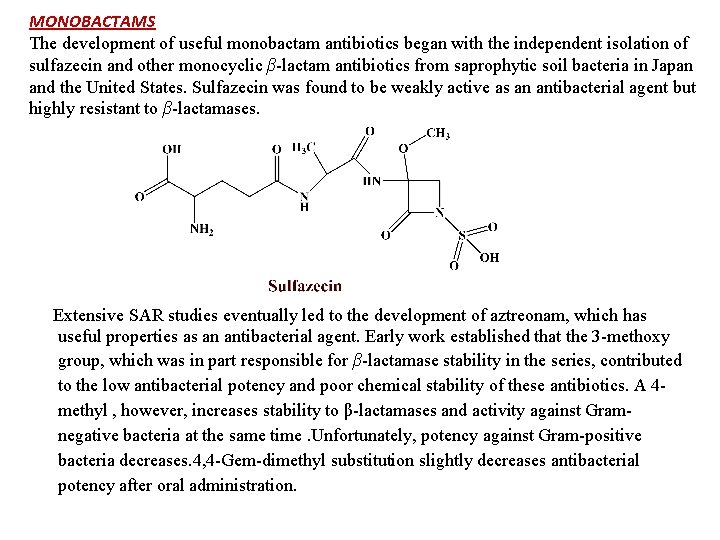
MONOBACTAMS The development of useful monobactam antibiotics began with the independent isolation of sulfazecin and other monocyclic β-lactam antibiotics from saprophytic soil bacteria in Japan and the United States. Sulfazecin was found to be weakly active as an antibacterial agent but highly resistant to β-lactamases. Extensive SAR studies eventually led to the development of aztreonam, which has useful properties as an antibacterial agent. Early work established that the 3 -methoxy group, which was in part responsible for β-lactamase stability in the series, contributed to the low antibacterial potency and poor chemical stability of these antibiotics. A 4 methyl , however, increases stability to β-lactamases and activity against Gramnegative bacteria at the same time. Unfortunately, potency against Gram-positive bacteria decreases. 4, 4 -Gem-dimethyl substitution slightly decreases antibacterial potency after oral administration.

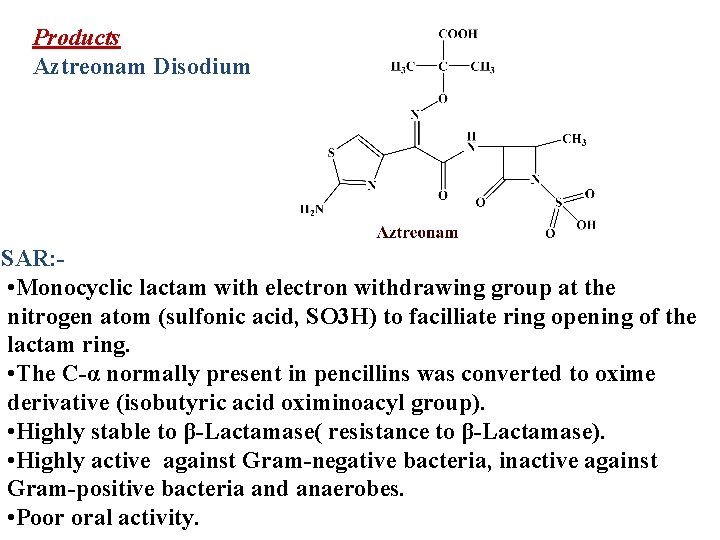
Products Aztreonam Disodium SAR: • Monocyclic lactam with electron withdrawing group at the nitrogen atom (sulfonic acid, SO 3 H) to facilliate ring opening of the lactam ring. • The C-α normally present in pencillins was converted to oxime derivative (isobutyric acid oximinoacyl group). • Highly stable to β-Lactamase( resistance to β-Lactamase). • Highly active against Gram-negative bacteria, inactive against Gram-positive bacteria and anaerobes. • Poor oral activity.

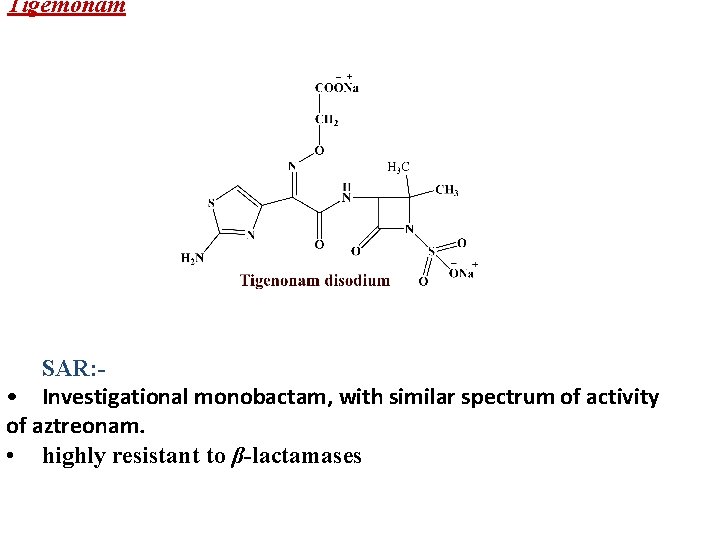
Tigemonam SAR: • Investigational monobactam, with similar spectrum of activity of aztreonam. • highly resistant to β-lactamases