Lec 10 Liquids Part 1 Buoyancy Archimedes Force

Lec 10: Liquids Part 1 • Buoyancy: Archimedes Force • Surface Tension and Capillaries • Fluids in Motion
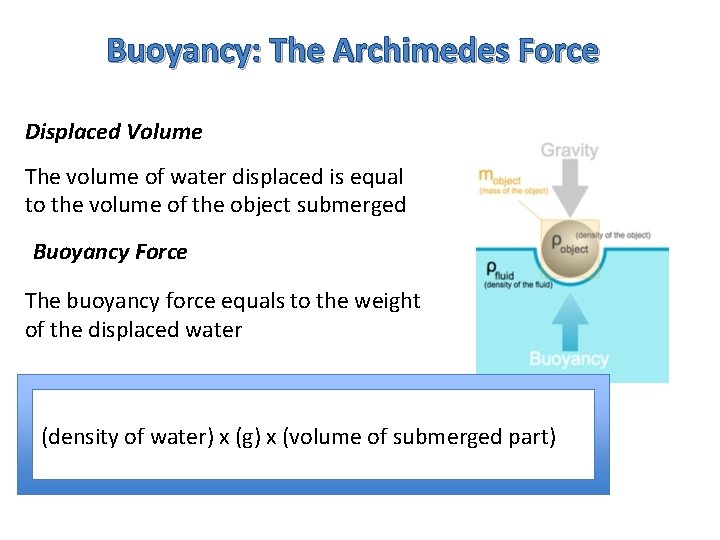
Buoyancy: The Archimedes Force Displaced Volume The volume of water displaced is equal to the volume of the object submerged Buoyancy Force The buoyancy force equals to the weight of the displaced water (density of water) x (g) x (volume of submerged part)
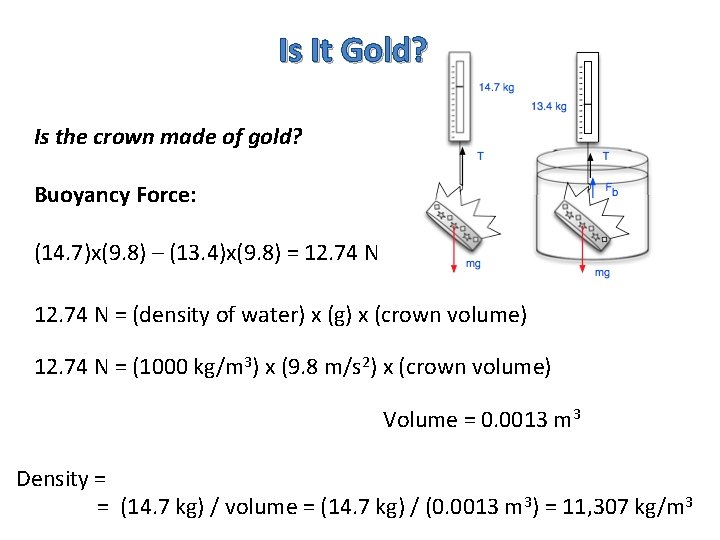
Is It Gold? Is the crown made of gold? Buoyancy Force: (14. 7)x(9. 8) – (13. 4)x(9. 8) = 12. 74 N = (density of water) x (g) x (crown volume) 12. 74 N = (1000 kg/m 3) x (9. 8 m/s 2) x (crown volume) Volume = 0. 0013 m 3 Density = = (14. 7 kg) / volume = (14. 7 kg) / (0. 0013 m 3) = 11, 307 kg/m 3

Surface Tension What keeps the water spiders from sinking into the water Water molecules interact with each other and normally each molecule is surrounded with other such molecules. Except for the surface, where molecules will have water molecules on one side and air (or other material) on the other. Depending on which forces water-water or water- material are stronger, the liquid will be ”pulled” and ”curved” in the corresponding direction.

Surface Tension What is the shape of a drop of water in outer space? http: //www. cnn. com/videos/world/2015/10/15/water-bubble-international-spacestation-nasa-orig. cnn/video/playlists/digital-studios-topicals/ Surface tension will cause liquids to ”try” to minimize their surface. A spherical shape gives the smallest surface-to-volume ratio.

Surface Tension And the Capillary Effect When the adhesive force (water sticking to the walls) exceeds the cohesive force (water-water attraction), water molecules at the water-wall surface will be pulled upward. We talk about capillary action. Question: Does soapy water have more or less surface tension? (a) Soap decreases surface tension (b) Soap increases surface tension (c) Soap does not affect surface tension

The Capillary Effect The surface tension depends on the curvature of the water surface and in tiny tubes (capillaries) it can exceed the force of gravity exerted on the fluid. The fluid will be ”pulled up” along that tube. • The larger the tension the higher the fluid • The smaller the radius the higher the fluid • The smaller the density the higher the fluid

Puzzles P 1. An object weighs 10 N in air and 8 N in water. How much is the buoyancy force on it in water? Will it sink or float P 2. A 500 -g object displaces 185 m. L of water. How much is its density? P 3. Water forms and stays in the shape of spherical droplets. Why not cubes? Or cylinders? Or other shape? P 4. Will water rise higher in a 1 -mm or 1 -cm radius tube? P 5. Many sports clothing materials are praised highly for their ”wicking” properties? What does it mean to ”wick” the sweat? How is water really ”wicked away”? And how do common paper towels absorb liquid?

Fluids in Motion Reynolds Number • Less than 2000 (Laminar) • More than 4000 (Turbulent)
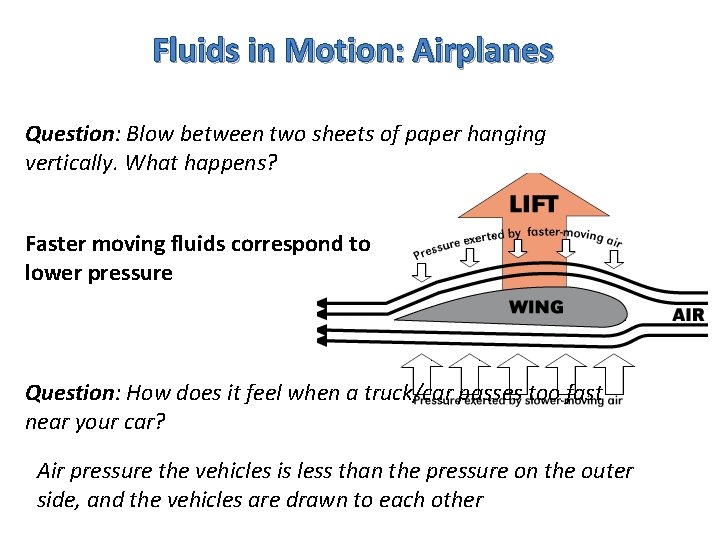
Fluids in Motion: Airplanes Question: Blow between two sheets of paper hanging vertically. What happens? Faster moving fluids correspond to lower pressure Question: How does it feel when a truck/car passes too fast near your car? Air pressure the vehicles is less than the pressure on the outer side, and the vehicles are drawn to each other

Viscous Fluids in Motion: Blood Question: What makes honey flow differently from water? Viscosity: • The more viscous the fluid, the more difficult to flow • The longer the hoe, the more opportunity the viscous forces have to slow down the fluid, the less amount of fluid flows • Pressure determines how hard the fluid is being pushed through the hose. The larger the pressure difference, the larger amount • The amount of fluid flowing is proportional to the 4 th power of the hose diameter

Poiseuille’s Equation Question: How do we want blood to flow in the blood vessels? (a) In laminar flow (b) In a turbulent flow (c) We don’t care
- Slides: 12