Learning Objectives What is halflife How does halflife

Learning Objectives: • What is half-life • How does half-life allow scientists to tell how old a fossil is • BE able to solve basic half-life problems

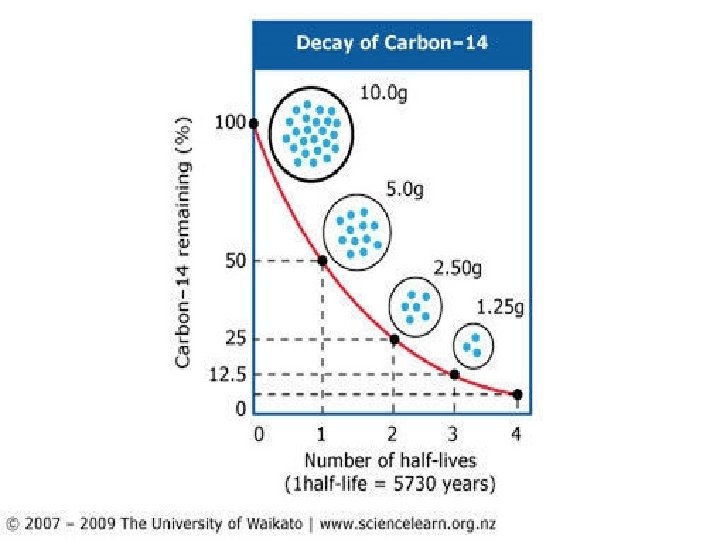
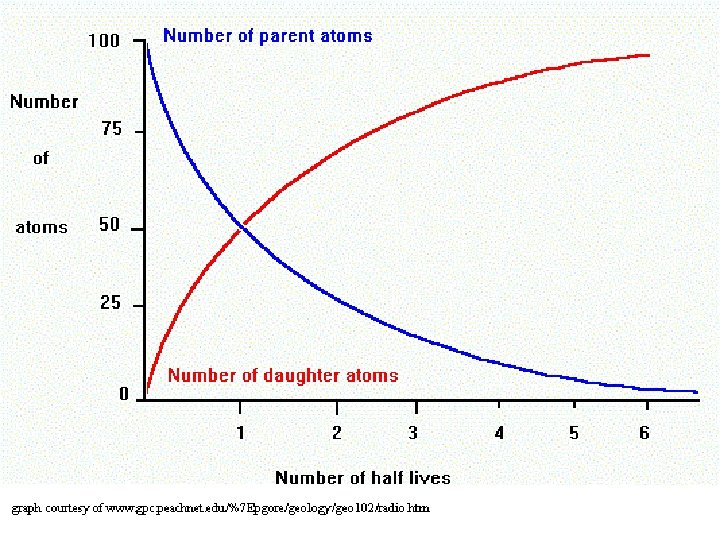
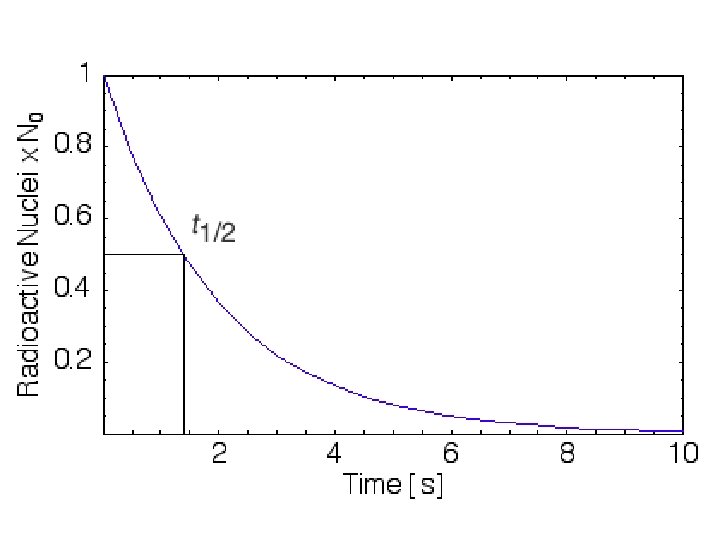
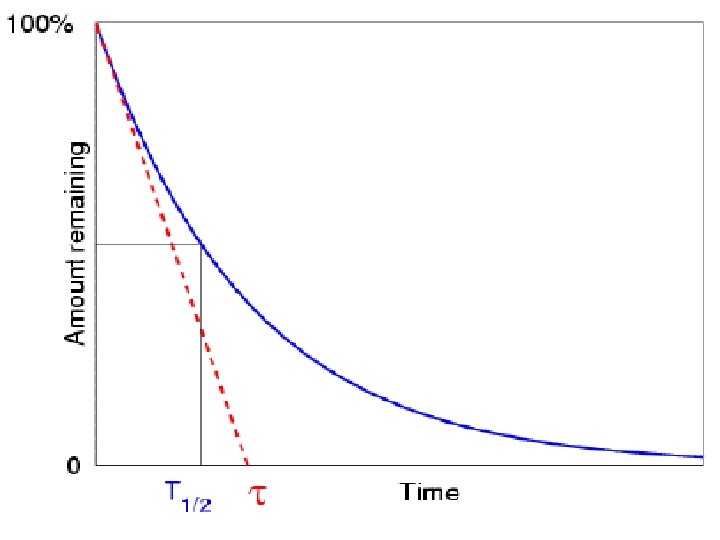
Radiometric Dating: Absolute Time Half-Life (λ) : The time it takes radioactive elements to to partially (50%) decay to a more stable form Ex: Carbon-14 (C 14) decays to Nitrogen-14 and has λ = 5, 730 years Potassium-40 (K 40) decays to Argon -40 λ = 1. 3 Billon years (1, 300, 000 years) Uranium 238 (U 238) decays to Lead-206 λ = 4. 4 Billon years (4, 400, 000 years)
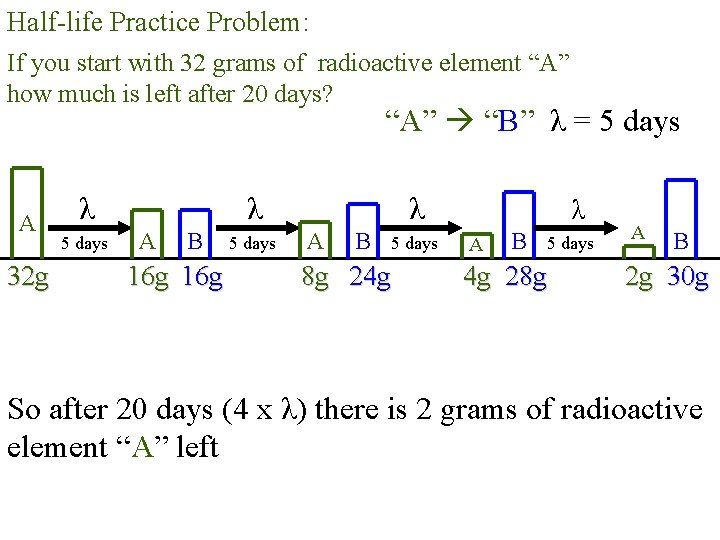
Half-life Practice Problem: If you start with 32 grams of radioactive element “A” how much is left after 20 days? “A” “B” λ = 5 days A 32 g λ 5 days λ A B 16 g 5 days λ A B 5 days 8 g 24 g λ A B 5 days 4 g 28 g A B 2 g 30 g So after 20 days (4 x λ) there is 2 grams of radioactive element “A” left

Half-life Practice Problem: “C” “D” λ = 7 days If you start with 80 grams of “C” how much is left after 35 days = 5 half-lives (λ =7 days) 7 days C = 80 g D = 0 g λ C = 40 g λ D = 40 g C=5 g D = 75 g λ C = 2. 5 g D = 77. 5 g C = 20 g D = 60 g λ C = 10 g D = 70 g λ

Half-life Practice Problem: “X” “Y” λ = 4 years If you start with 60 grams of “X” How much “Y” is created after 12 years? 12 years = 3 half-lives (λ =4 years) X = 60 g λ Y = 0 g X = 30 g Y = 30 g λ X = 15 g λ Y = 45 g X = 7. 5 g Y = 52. 5 g

A Real Half-life Problem: C 14 N 14 λ = 5, 730 years A bone fossil of animal has 75 g of N 14 and 25 g of C 14. Assuming there was 0 g of N 14 when the animal died. Estimate the age of the fossil C 14 = 100 g N 14 = 0 g Animal Died λ 5, 730 C 14 = 50 g N 14 = 50 g λ 5, 730 2 x λ (5, 730 years) = 11, 460 years old C 14 = 25 g N 14 = 75 g Fossil Found
- Slides: 10