Learning Objectives l To recognize the components of

Learning Objectives l To recognize the components of chemical equations l To recognize three types of chemical equations l To understand the law of conservation of mass

Chemicals and Their Reactions Chapter 6

Where do we find Chemical Reactions? l Everywhere!

What is a chemical reaction? l a reaction between 2 or more elements or compounds to form new substances, with new properties.

Describing Chemical Reactions l Equations (either word or chemical) are used for reactions l Equations are balanced
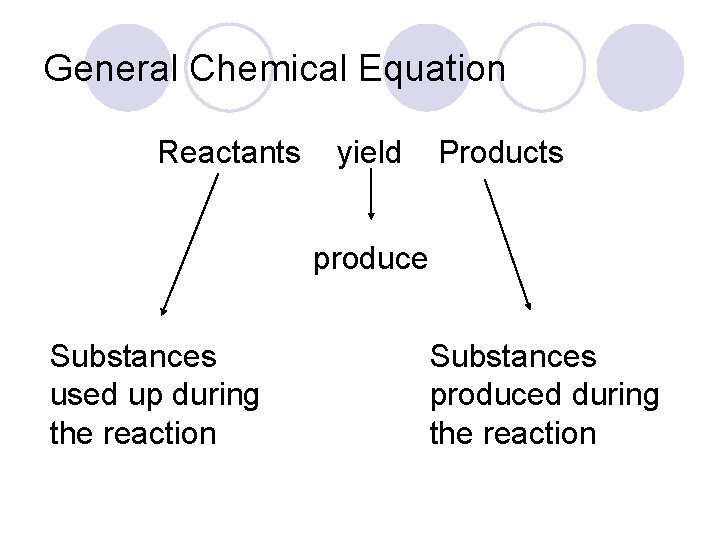
General Chemical Equation Reactants yield Products produce Substances used up during the reaction Substances produced during the reaction

Word Equations Iron + sulfur iron (II) sulfide + energy l The arrow indicates the direction of the reaction l The ‘+’ sign on reactant side means the substances must be in contact l The ‘+’ sign on product side means more than one product
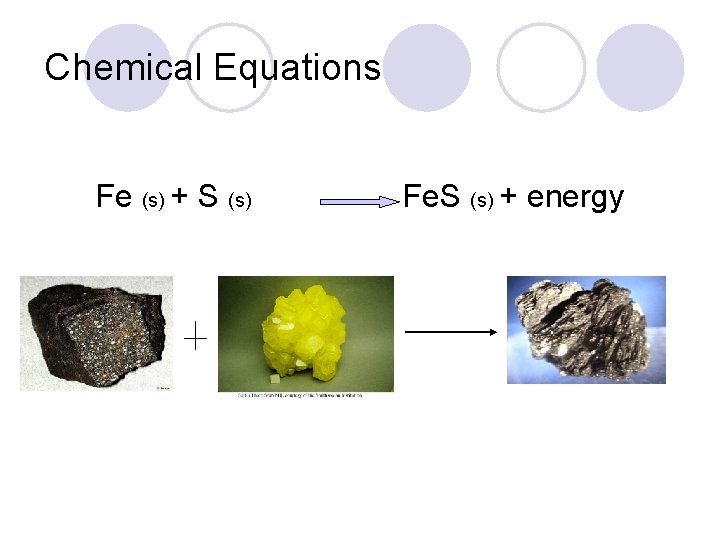
Chemical Equations Fe (s) + S (s) Fe. S (s) + energy

Word vs Chemical Equations? l chemical equations provide more detail such as: l. Chemical formulas of substances involved l. The ratio of substances involved l. State of substances involved

State Symbols for Equations Symbol Meaning (s) Solid (l) Liquid (g) Gas (aq) Aqueous (dissolved in water)
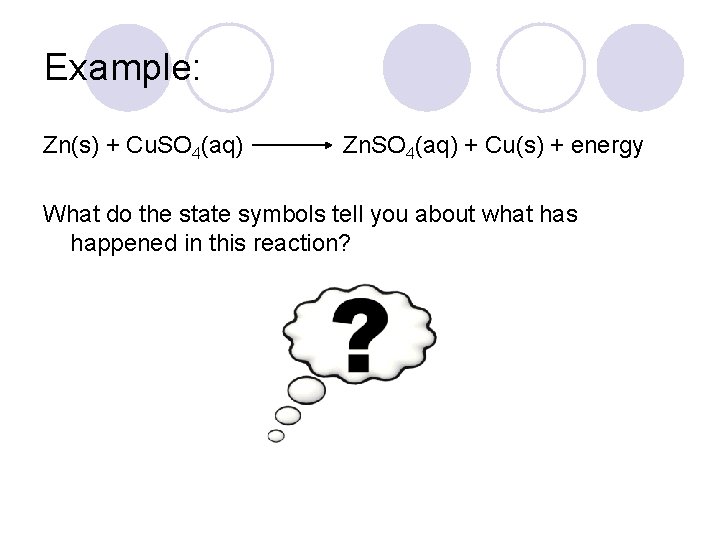
Example: Zn(s) + Cu. SO 4(aq) Zn. SO 4(aq) + Cu(s) + energy What do the state symbols tell you about what has happened in this reaction?

Energy and Reactions l Exothermic reactions release energy l Energy will be on the product side of the equation l Exothermic = exit l Examples:

Energy and Reactions l Endothermic reactions require energy in order to occur (absorb/consume energy) l Energy will be on the reactant side of the equation l Examples:

Energy and Reactions l A reaction is exothermic if more energy is produced than was put into the reaction l A reaction is endothermic if more energy is required to run the reaction than is produced
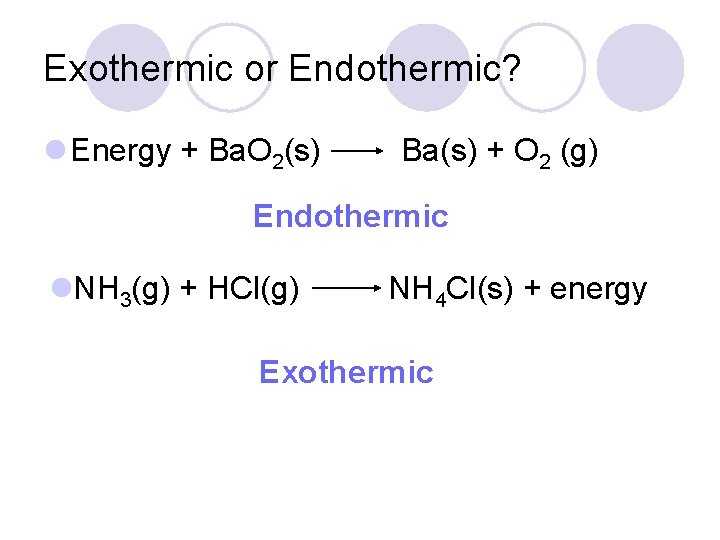
Exothermic or Endothermic? l Energy + Ba. O 2(s) Ba(s) + O 2 (g) Endothermic l. NH 3(g) + HCl(g) NH 4 Cl(s) + energy Exothermic

Exothermic or Endothermic? C 6 H 12 O 6(s) + O 2 (g) CO 2 (g) + H 2 O(l) + energy Exothermic (This is cellular respiration) CO 2 (g) + H 2 O(l) + energy C 6 H 12 O 6(s) + O 2 (g) Endothermic (This is photosynthesis)

Conserving Mass in Reactions Law of Conservation of Mass l The total mass of the reactants equals the total mass of the products
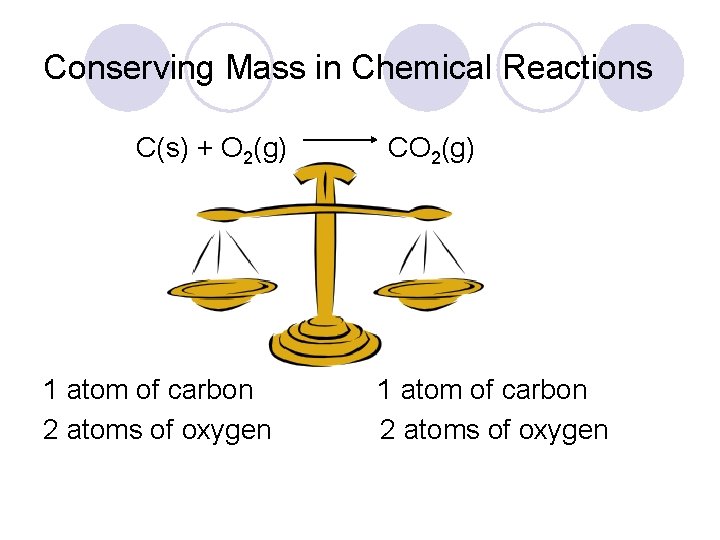
Conserving Mass in Chemical Reactions C(s) + O 2(g) 1 atom of carbon 2 atoms of oxygen CO 2(g) 1 atom of carbon 2 atoms of oxygen
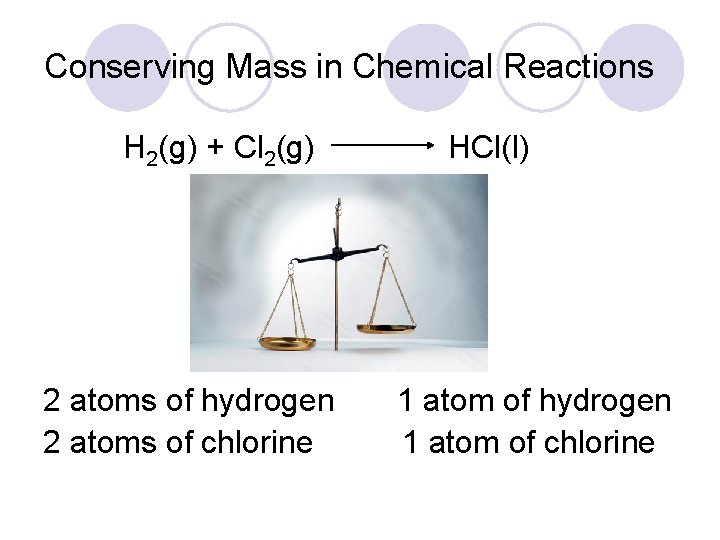
Conserving Mass in Chemical Reactions H 2(g) + Cl 2(g) 2 atoms of hydrogen 2 atoms of chlorine HCl(l) 1 atom of hydrogen 1 atom of chlorine
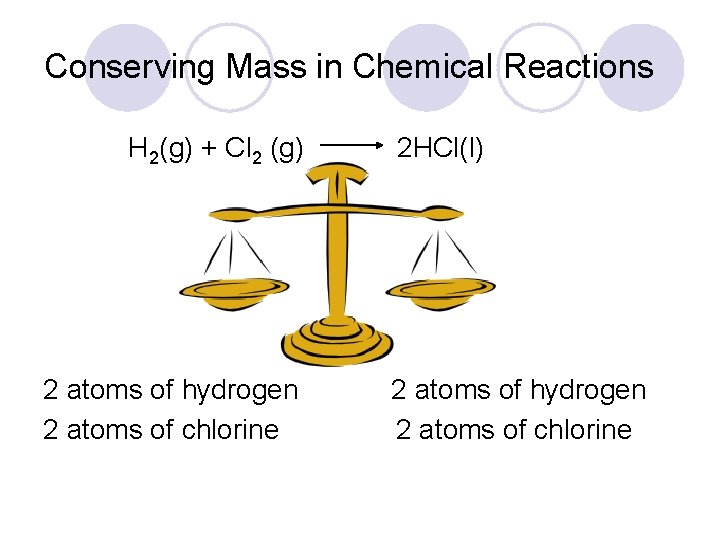
Conserving Mass in Chemical Reactions H 2(g) + Cl 2 (g) 2 atoms of hydrogen 2 atoms of chlorine 2 HCl(l) 2 atoms of hydrogen 2 atoms of chlorine
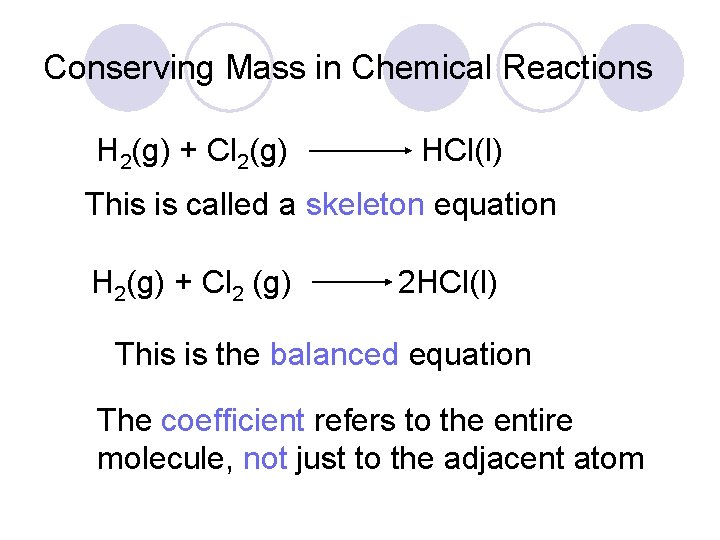
Conserving Mass in Chemical Reactions H 2(g) + Cl 2(g) HCl(l) This is called a skeleton equation H 2(g) + Cl 2 (g) 2 HCl(l) This is the balanced equation The coefficient refers to the entire molecule, not just to the adjacent atom

To balance things out…. l If you did your homework: l Page 227 # 6, 8, 9 l Page 232 # 2 – 4, 6, 7 l If you DID NOT do your homework l Page 227 # 1 – 9 l Page 232 # 2 – 4, 6, 7
- Slides: 22