Learning Objective To be able to Describe how

Learning Objective To be able to: Describe how some metals are extracted using carbon Key Words: carbon, element, reactivity

09 September 2020 Extraction of metals using carbon Date and title in books. You need a pen, pencil & ruler. Starter Activity – 5 minutes 1. Name two reactive metals 2. Name two unreactive metals 3. Give two reasons why copper is used in cooking pans 4. Give two reasons why gold is used for jewellery.
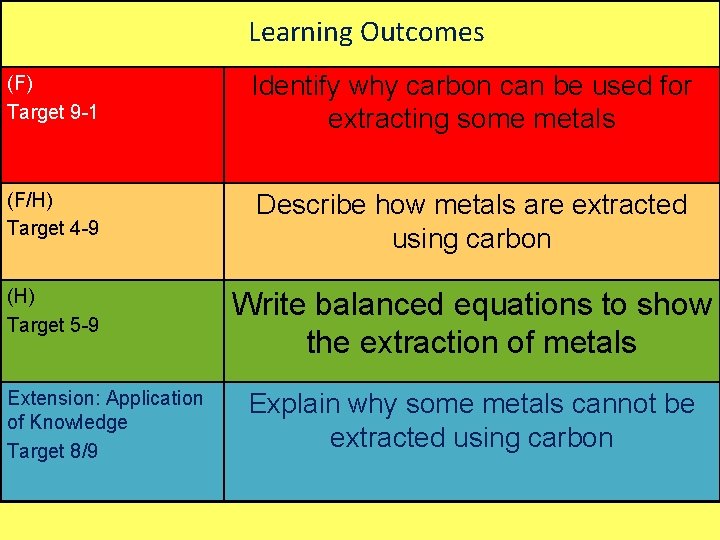
Learning Outcomes (F) Target 9 -1 Identify why carbon can be used for extracting some metals (F/H) Target 4 -9 Describe how metals are extracted using carbon (H) Target 5 -9 Write balanced equations to show the extraction of metals Extension: Application of Knowledge Target 8/9 Explain why some metals cannot be extracted using carbon

Extracting Metals • Many metals need to be extracted from its ore. • An ore is a rock which contains a mineral or metal. • Some examples include: • Iron Ore – to get Iron • Aluminium Ore or Bauxite – to get aluminium

How is carbon used to extract metals? Metals that are less reactive than carbon can be extracted from their ores by burning with carbon. raw materials Iron is extracted by this method in a blast furnace. The iron ore is heated with carbon -rich coke at very high temperatures. hot air The iron collected from a blast furnace is only 96% pure. Usually, this product will be treated further because the impurities make iron brittle. molten iron molten slag

Blast furnace in a modern steel works
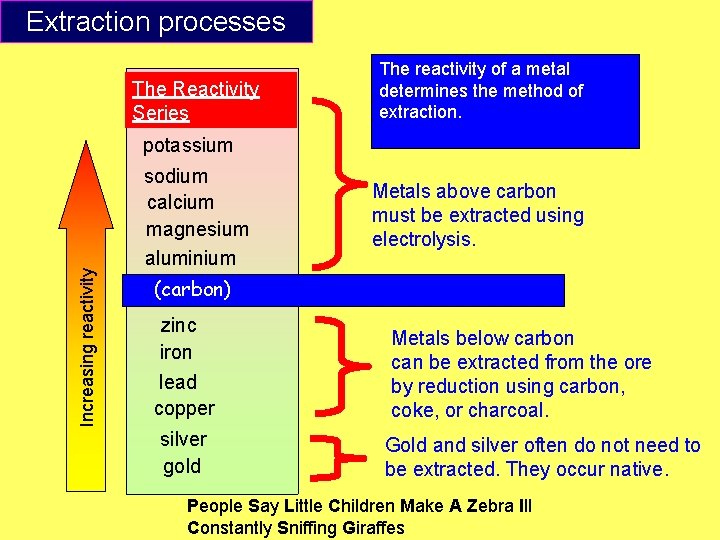
Extraction processes The Reactivity Series The reactivity of a metal determines the method of extraction. Increasing reactivity potassium sodium calcium magnesium aluminium Metals above carbon must be extracted using electrolysis. (carbon) zinc iron lead copper silver gold Metals below carbon can be extracted from the ore by reduction using carbon, coke, or charcoal. Gold and silver often do not need to be extracted. They occur native. People Say Little Children Make A Zebra Ill Constantly Sniffing Giraffes

Extracting Copper and Aluminium Iron by reaction with carbon: iron oxide + carbon iron + carbon dioxide Can you write the balanced symbol equation?

Ores Activity Foundation Questions (1 mark each) 1. What is an ore? – Most metals must be extracted from rocks, called ores, in the Earth's crust. Ores contain the metal, or a compound of the metal, in a high enough concentration to make it worth extracting the metal. 2. Name two metals and their ores – Iron oxide and aluminium oxide

Ores Activity Standard Questions (3 marks each) 1. Describe how iron is extracted using carbon – Many ores contain metal oxides, therefore many metals can be extracted from their ores by reduction reactions. The method used to extract a given metal depends on how reactive it is. Iron may be extracted by reduction with carbon. In this reaction, the iron oxide is reduced to iron, and the carbon is oxidised to carbon dioxide. iron oxide + carbon → iron + carbon dioxide 2 Fe 2 O 3 + 3 C → 4 Fe + 3 CO 2
- Slides: 10