Lead Acid Battery Safety ChemistryCTE Lead Acid Battery

Lead Acid Battery Safety Chemistry/CTE

Lead Acid Battery Safety Topics: • • • Why is sulfuric acid hazardous? Personal Protective Equipment (PPE) Acid spills Overcharging MSDS Battery Disposal Concerns 2

What the big deal with sulfuric acid? ? ? Play 1 - Sulfuric acid experiment FYI: Battery Acid is 4 M!!! 3

Play 2 - Homer Simpson almost drinks sulfuric acid 4

Personal Protective Equipment (PPE) • • Goggles Apron Gloves Closed toe shoes 5

Play 3 - Draining a lead acid battery In the video: • What is this person doing right? • What is this person doing wrong? 6

What to do if there is an acid spill… 1. Notify your teacher and those around you (so no one accidentally comes into contact with the spill) 2. Put on PPE (if not already on) 3. Obtain an acid spill kit (we use solid sodium bicarbonate aka…baking soda) 4. Put a generous amount of sodium bicarbonate all around the spill. It will bubble up. Keep adding until it no longer bubbles. 5. At this point the acid is neutralized so you make finish cleaning it up using a paper towel and water if needed. 7

So what the chemical reaction for neutralization of the sulfuric acid? H 2 SO 4 (aq) + Na. HCO 3 (s) � Na 2 SO 4 (aq) + CO 2 (g) + H 2 O (l) 8

Hazard of Overcharging Play 4 - Hazard of overcharging • Overcharging comes from using a charging device that is faulty or using a voltage that is too high. • Overcharging produces hydrogen gas which is extremely flammable. 9

Teacher Balloon demo or video clip below Play 5 - Teach hydrogen balloon demo 10

Material Safety Data Sheets 11

1. 2. 3. 4. 5. 6. 7. 8. Chemical Product Information Composition/Ingredients Hazards Identification First Aid Measures Fire Fighting Measures Accidental Release Measures Handling and Storage Exposure Controls/Personal Protection 9. Physical and Chemical Properties 10. Stability and Reactivity 11. Toxicological Information 12. Ecological Information 13. Disposal Considerations 14. Transport Information 15. Regulatory Information 12 16. Additional Information

Lead Acid Battery Disposal Some facts… • More than 25 million motor vehicles are registered in California • Each vehicle uses a lead acid battery • The average battery contains 17. 5 pounds of lead and 1. 5 gallons of sulfuric acid 13

Why is lead bad? Low-level exposure: – Fatigue – Impaired central nervous symptoms – Impaired learning (which is why in 1977 lead paint was banned in residental homes) Severe lead poisoning: – – – Coma Convulsions Irreversible mental retardation Seizures Death Remember 17. 5 pounds per battery! 14

Disposal • If illegally dumped or placed in a landfill, lead and sulfuric acid can seep into the soil and contaminate ground water and/or run off into surface water and harm aquatic life as well. • Proper disposal: recycle! – You can exchange your battery when you buy a new one – Take to a household hazardous waste (HHW) collection facility • How do they do it? 15
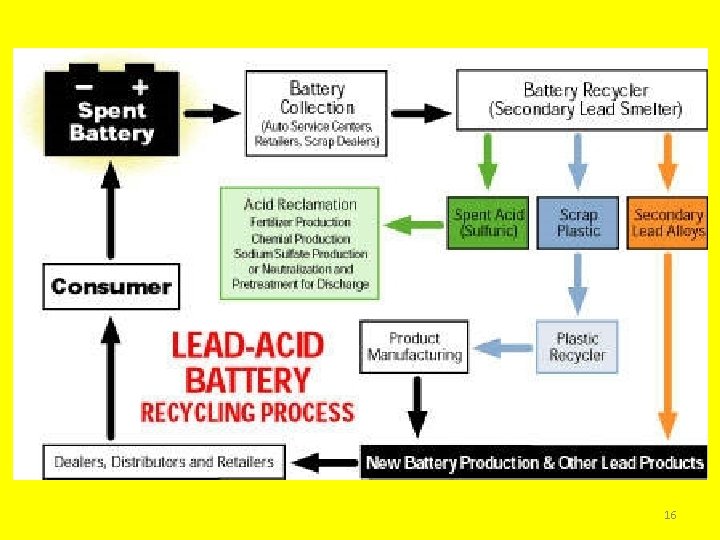
16

Assessment of Lead Acid Battery Safety 1. List the four items you should wear when working with lead acid batteries? 2. How do you clean up an acid spill? 3. What causes overcharging? 4. What flammable gas is created from overcharging? 1. Goggles, apron, gloves, closed toe shoes 2. Notify teacher, put on PPE, add Na. HCO 3 till no longer bubbles, clean up with paper towels and water 3. Faulty charging device or a too high voltage 4. Hydrogen, H 2 17

Assessment of Lead Acid Battery Safety 5. What does MSDS stand 5. Material Safety Data for? Sheets 6. List one piece of 6. Many possible answers information that can be Refer to Slide 12 found on a MSDS? 7. Sulfuric Acid, H 2 SO 4 and 7. What are the 2 chemicals Lead, Pb in a battery that cause it to H 2 SO 4: strong acid, be hazardous and need corrosive special disposal? For each Pb: mental impairment chemical, list at least one to death (see slide 14 characteristic that makes it for specifics) 18 hazardous.

Assessment of Lead Acid Battery Safety 8. Why do you think gasoline 8. Because the lead in sold now is “unleaded”? gasoline was an environmental hazard 9. How should you dispose of a lead acid battery? 9. Take to a battery recycling/HHW location 10. In your CTE class, we use sealed lead acid batteries 10. Since sealed, no acid (SLAs) instead of the type spills that is in your car. What is the major safety benefit of doing this? 19
- Slides: 19