LEAD ACID BATTERY MODELING IEEE ESSB Summer 2016

LEAD ACID BATTERY MODELING IEEE ESSB Summer 2016 Meeting Frank X. Garcia 12 June 2016 1


Presentation Objectives • Explain lead acid cell operation at the atomic level • Present a Randles circuit model approximation of a lead acid cell • Examine computer simulations trending using Randles circuit model 3

Outline • Battery Overview – – Components Electrochemistry Double layer capacitance State of health parameters • Randles circuit model – Single cell approximation – Failure modes analysis – Computer Simulations 4
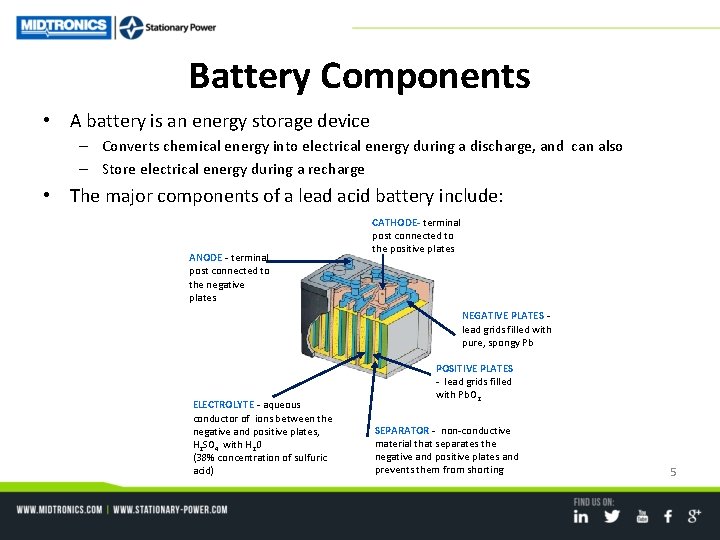
Battery Components • A battery is an energy storage device – Converts chemical energy into electrical energy during a discharge, and can also – Store electrical energy during a recharge • The major components of a lead acid battery include: ANODE - terminal post connected to the negative plates CATHODE- terminal post connected to the positive plates NEGATIVE PLATES - lead grids filled with pure, spongy Pb ELECTROLYTE - aqueous conductor of ions between the negative and positive plates, H 2 SO 4 with H 20 (38% concentration of sulfuric acid) POSITIVE PLATES - lead grids filled with Pb. O 2 SEPARATOR - non-conductive material that separates the negative and positive plates and prevents them from shorting 5
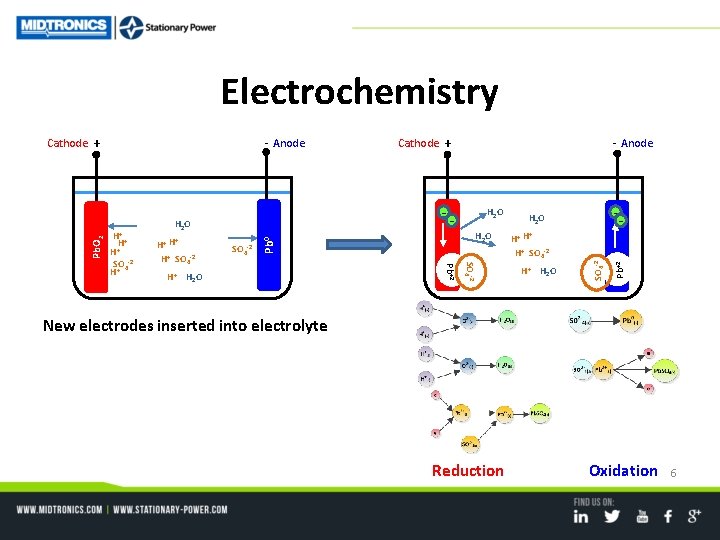
Electrochemistry H+ H 2 O -- H 2 O H+ H H+ SO 4 -2 + H+ H 2 O Pb+2 SO 4 -2 Pb 0 Pb. O 2 + H 2 O SO 4 -2 H+ H H+ SO 4 -2 Pb+2 H+ + H H+ SO 4 -2 H+ -- H 2 O - Anode Cathode + SO 4 -2 - Anode Cathode + New electrodes inserted into electrolyte Reduction Oxidation 6
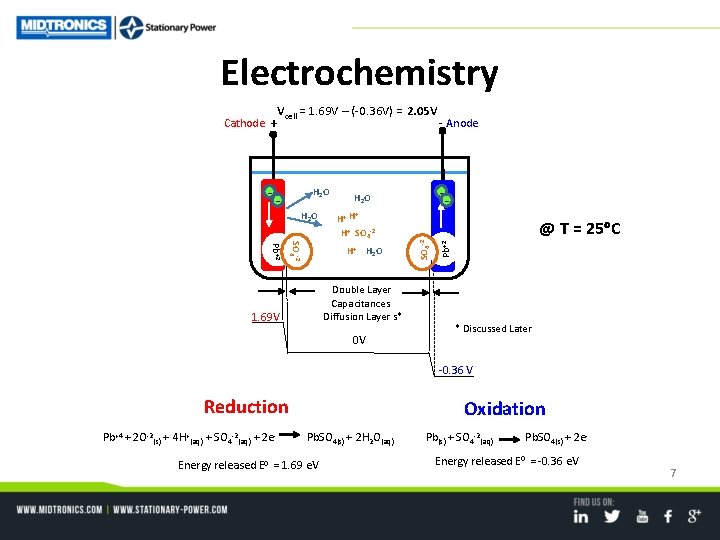
Electrochemistry Vcell = 1. 69 V – (-0. 36 V) = 2. 05 V H 2 O H+ H H+ SO 4 -2 Pb+2 Double Layer Capacitances Diffusion Layer s* 1. 69 V -- H 2 O H+ H 2 O 0 V - Anode @ T = 25 C Pb+2 -- SO 4 -2 Cathode + * Discussed Later -0. 36 V Reduction Pb+4 + 2 O-2(s) + 4 H+(aq) + SO 4 -2(aq) + 2 e- Oxidation Pb. SO 4(s) + 2 H 2 O(aq) Energy released E 0 = 1. 69 e. V Pb(s) + SO 4 -2(aq) Pb. SO 4(s) + 2 e- Energy released E 0 = -0. 36 e. V 7
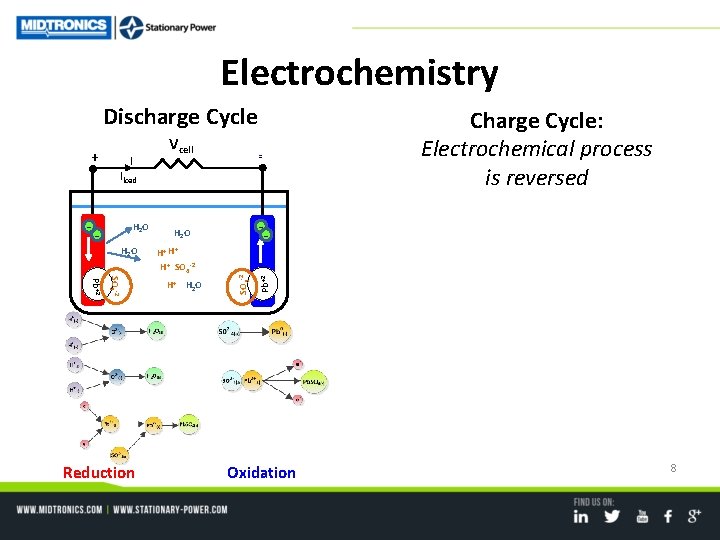
Electrochemistry Discharge Cycle + I Vcell - Iload H 2 O -- H 2 O H+ H H+ SO 4 -2 Pb+2 H+ H 2 O Pb+2 H 2 O SO 4 -2 -- Charge Cycle: Electrochemical process is reversed Reduction Oxidation 8

Double Layer Capacitance • • Ions adsorbed to the surface of the electrode held by electrostatic force Positive ions too large to penetrate electrode metal surface Only electrons can travel through the metal conduction bands Solvated ions encapsulated by water molecules can migrate through diffusion layer A second layer of solvated ions create the outer layer forming capacitive double layer Double layer has an ionic density gradient which allow Ionic migration in the diffusion layer Bulk region maintains an equal concentration of electrolyte 9

State of Health Parameters • Float voltage – Set to recommended range for optimal battery life – Life expectancy decreases as float voltage increases • Float current – A high float current indicates aging battery • Negative post temperature – Indefinite 10 o C temperature rise decrease battery life by 50% • Electrolyte Specific Gravity – Sulfuric acid (H 2 SO 4) concentration can indicate state of charge – Fully charged: 1. 26 to 1. 3 specific gravity • Admittance/Impedance/Resistance Trending – Admittance will decrease as the battery ages – Impedance and Resistance will increase as battery ages 10

Complete Randles Circuit Model Rm+ Rm- Lumped, metallic resistance of the posts, bus bars, grids, plates, and metallic joints at the cathode, Rm+, and the anode, Rm- Cdl+ Cdl- Cbulk Double layer capacitance at the interface between the positive plate surface and the electrolyte, Cdl+, and the negative plate surface and the electrolyte interface, Cdl-. Cdl depends on the effective surface area of each plate, and the battery state of charge, and it is a combination of the series connection of a double layer capacitor and a diffusion layer capacitor. Equivalent charge transfer resistance at the interface between the positive plate and the electrolyte due to the ionization losses of Pb, Rct+, and the interface between the negative plate and the electrolyte, Rct-. These are resistive paths that are tunneled through Cdl+ and Cdl-, respectively. Diffusion layer impedance in the diffusion layer near the positive plate, Zdiff+, and the negative plate, Zdiff-. These elements are modeled using the Warburg Diffusion Element, which has a constant phase at all frequencies and a magnitude inversely proportional to the frequency. Refer to Appendix A for a review of the Warburg Diffusion Element. Equivalent bulk capacitance of the electrolyte material. Note: Cbulk >> Cdl+, Cdl-. Rbulk Equivalent bulk resistance of the electrolyte material 1. 69 V Voltage potential at the cathode relative to the bulk electrolyte, stemming from the Reduction release of energy -0. 36 V Voltage potential at the negative plate relative to the bulk electrolyte, stemming from the Oxidation release of energy Rct+ Rct. Zdiff+ Zdiff- 11
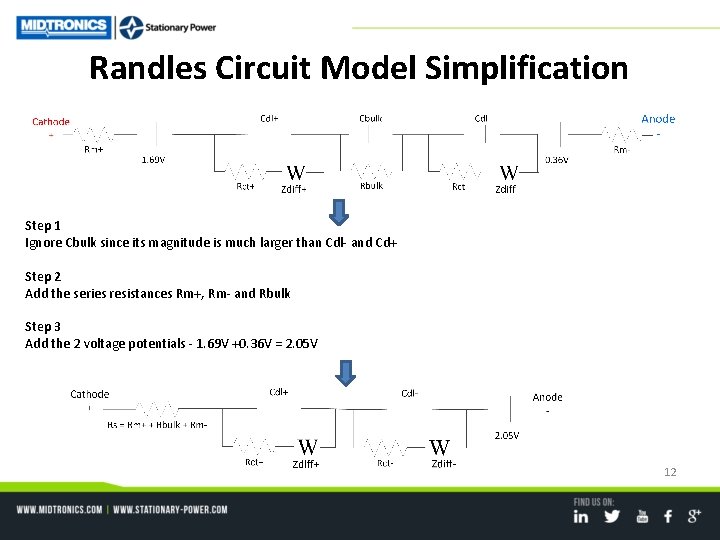
Randles Circuit Model Simplification Step 1 Ignore Cbulk since its magnitude is much larger than Cdl- and Cd+ Step 2 Add the series resistances Rm+, Rm- and Rbulk Step 3 Add the 2 voltage potentials - 1. 69 V +0. 36 V = 2. 05 V 12
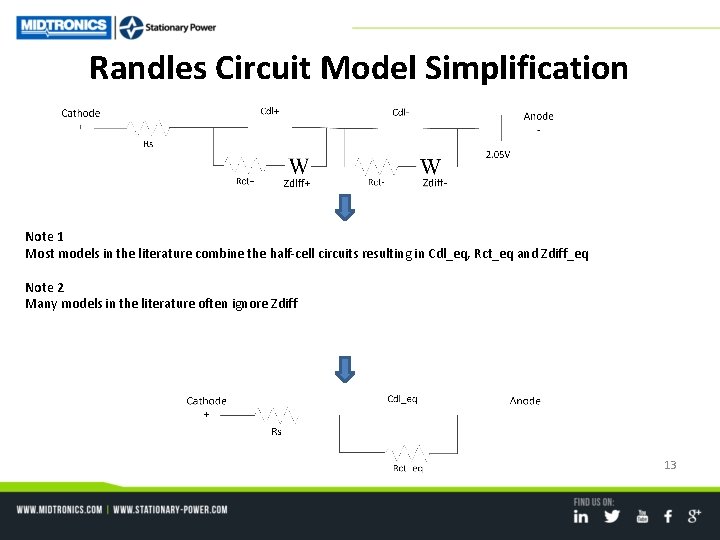
Randles Circuit Model Simplification Note 1 Most models in the literature combine the half-cell circuits resulting in Cdl_eq, Rct_eq and Zdiff_eq Note 2 Many models in the literature often ignore Zdiff 13

Failure Modes Analysis Failure Mode Description Corrosion Negative strap corrosion Plate lugs, plate strap/lead corrosion Positive grid corrosion (grid expansion) Part of aging process Dry-out Electrolyte Reduction due to overcharging Premature capacity loss Shedding of positive active material due to heavy cycling Battery Admittance Decreases Sulphation Negative electrode irreversible sulphation (lead sulfate crystallizing on plate due to undercharging) Electrolyte Stratification Electrolyte settles to the bottom of the cell when not in use or not fully charged Loss of Compression VRLA glass mat 14
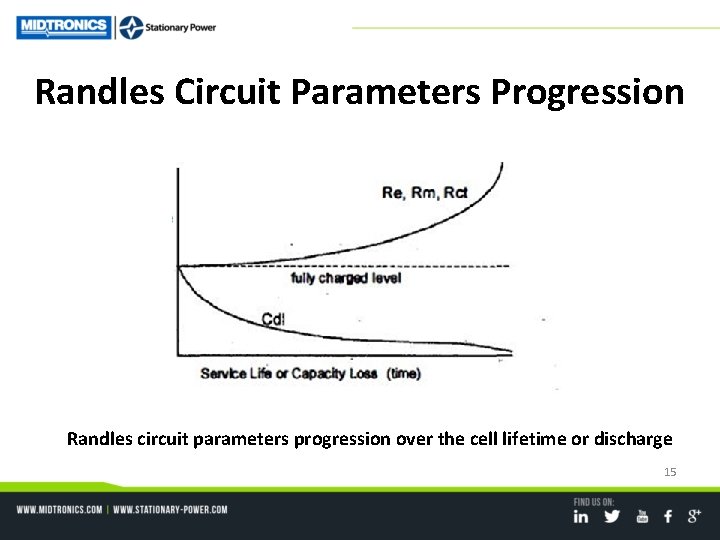
Randles Circuit Parameters Progression Randles circuit parameters progression over the cell lifetime or discharge 15

• Strategy Computer Simulations – Examine degradation of a cell using complete Randles Circuit Model – Simulate Nyquist plots of baseline, +20% and +40% impedance due to cell aging – Synthesize component values to determine parametric changes • Circuit Model (one cell) Schematic Designation Equivalent, Complete Randles Model R 3 Rm+ + Rm- R 1 Rct+ C 1 Cdl+ R 2 Rct- C 2 Cdl- X 1 Zdiff+ (Warburg Constant Phase Element, cpe) X 2 Zdiff- (Warburg Constant Phase Element, cpe) C 3 Cbulk R 4 Rbulk 16
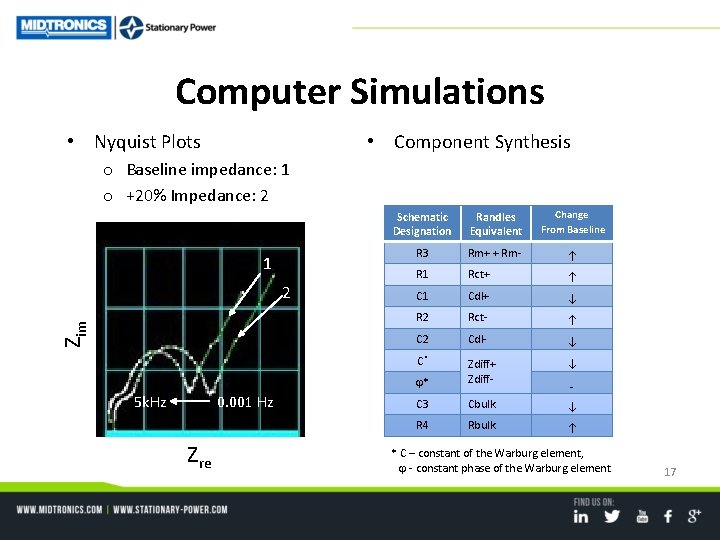
Computer Simulations • Nyquist Plots • Component Synthesis o Baseline impedance: 1 o +20% Impedance: 2 1 Zim 2 5 k. Hz 0. 001 Hz Zre Schematic Designation Randles Equivalent Change From Baseline R 3 Rm+ + Rm- R 1 Rct+ C 1 Cdl+ R 2 Rct- C 2 Cdl- C* * Zdiff+ Zdiff- C 3 Cbulk R 4 Rbulk - * C – constant of the Warburg element, - constant phase of the Warburg element 17

Computer Simulations • Nyquist Plots • Component Synthesis o Baseline impedance: 1 o +40% Impedance: 2 1 Zim 2 5 k. Hz 0. 001 Hz Zre Schematic Designation Randles Equivalent Change From Baseline R 3 Rm+ + Rm- + R 1 Rct+ + C 1 Cdl+ - R 2 Rct- + C 2 Cdl- - C* - * Zdiff+ Zdiff- C 3 Cbulk - R 4 Rbulk + - * C – constant of the Warburg element, - constant phase of the Warburg element 18

Summary • Randles circuit model approximates electrochemistry of a lead acid cell • Simplified Randles circuit model reduces analysis accuracy • Trending in Randles element values add visibility to battery state of health – Baseline immitance using Discrete Frequency Immitance Spectroscopy – Synthesize Randles circuit model battery elements – Analyze battery degradation by comparing element value changes from a known reference 19
- Slides: 19