Le Chateliers Principle Review Objective Today I will

Le Chatelier’s Principle Review

Objective: Today I will be able to: Apply Le Chatelier’s Principle to explaining how equilibrium systems adjust to stresses of concentration, temperature, pressure and volume Evaluation/Assessment: Informal assessment: Monitoring student interactions and questions as they complete the Le Chatelier’s Principle practice. Formal assessment: Analyzing student responses to practice and the exit ticket Common Core Connection Build Strong Content Knowledge Reason abstractly and quantitatively Make sense of problems and persevere in solving them

Lesson Sequence Evaluate: Warm – Up Explain: Le Chatelier’s Principle Notes Elaborate: Le Chatelier’s Principle Practice Evaluate: Exit Ticket

Warm - Up Write the equilibrium constant expression for the following reactions a. O 3 (g) + NO(g) → O 2(g) + NO 2(g) b. 2 CO(g) + O 2(g) → 2 CO 2(g) c. NH 4 NO 3(s) → N 2 O(g) + 2 H 2 O(l) d. 2 H 2 O(g) → 2 H 2(g) + O 2(g)

Objective Today I will be able to: Apply Le Chatelier’s Principle to explaining how equilibrium systems adjust to stresses of concentration, temperature, pressure and volume

Homework Finish Le Chatelier’s Principle Practice Wear closed toe shoes for lab Wednesday and Thursday Study for Equilibrium Constant/ Le Chatelier’s Quiz Friday

Agenda Warm – Up Le Chatelier’s Principle Notes Le Chatelier’s Principle Practice Exit Ticket

Le Chatelier’s Principle Review Answer the following questions with a partner

Determine whether the following statements are true or false At equilibrium: 1. The rate of the forward reaction equals the rate of the reverse reaction 2. The concentration of reactants and products is always equal 3. The concentrations of reactants and products remain constant

Answers 1. True 2. False 3. True

True or False Le Chatelier’s principle describes how a system at equilibrium restores equilibrium after it has been “stressed”

Answer True

What different “stresses” can be applied to a system? Adding/ Removing Reactants Adding/ Removing Products Changing Pressure Changing Volume Changing Temperature
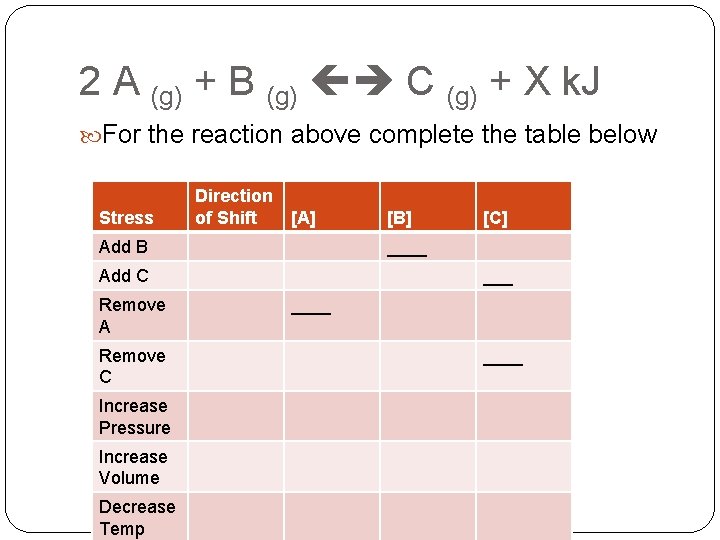
2 A (g) + B (g) C (g) + X k. J For the reaction above complete the table below Stress Direction of Shift [A] Add B Remove C Increase Pressure Increase Volume Decrease Temp [C] ____ Add C Remove A [B] ____
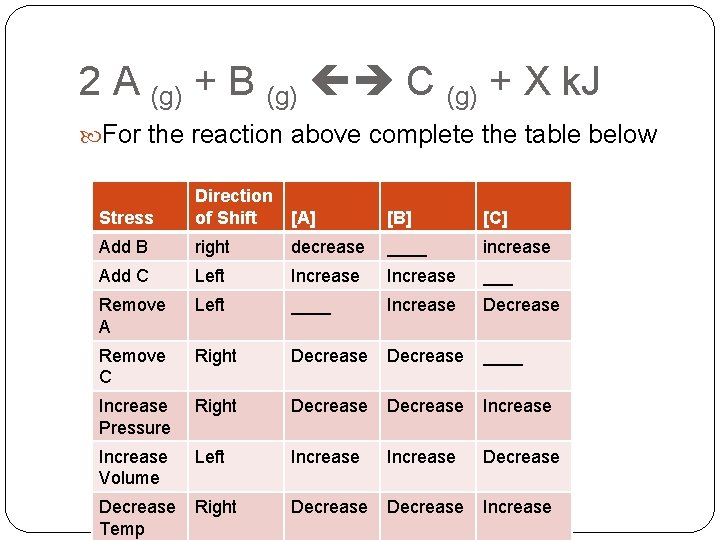
2 A (g) + B (g) C (g) + X k. J For the reaction above complete the table below Stress Direction of Shift [A] [B] [C] Add B right decrease ____ increase Add C Left Increase ___ Remove A Left ____ Increase Decrease Remove C Right Decrease ____ Increase Pressure Right Decrease Increase Volume Left Increase Decrease Temp Right Decrease Increase
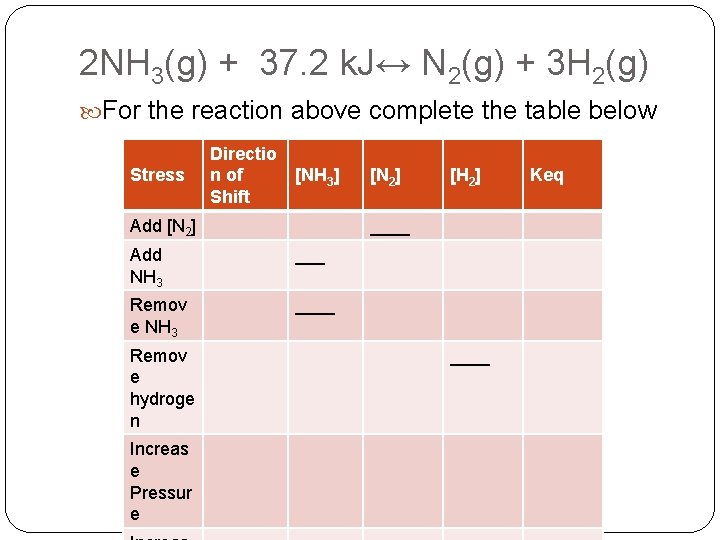
2 NH 3(g) + 37. 2 k. J↔ N 2(g) + 3 H 2(g) For the reaction above complete the table below Stress Directio n of Shift [NH 3] Add [N 2] ___ Remov e NH 3 ____ Increas e Pressur e [H 2] ____ Add NH 3 Remov e hydroge n [N 2] ____ Keq

Le Chatelier’s Principle Notes

Le Chatelier’s Principle When an equilibrium system is subjected to a change, the system will “shift” to counteract the change Example reaction A (g) + B (g) C (g) + X k. J

A (g) + B (g) C (g) + X k. J Add a reactant – moves to product side to consume it Add a product – moves to reactant side to consume it Remove a reactant – moves to reactant side to produce more Remove a product – moves to product side to produce more

A (g) + B (g) C (g) + X k. J Increase temperature – because it is an exothermic reaction, shift to the reactants side Decrease temperature – shift to the products The opposite would be true for an endothermic reaction

A (g) + B (g) C (g) + X k. J Increase pressure – move to side with less moles of gas Decrease pressure – move to side with more moles of gas Increase volume – move to side with more moles of gas Decrease volume – move to side with less moles of gas

Le Chatelier’s Principle Practice

Exit Ticket �Given the following reaction: Use Le Chatelier’s principle to determine what would happen if the following changes were made Increase the concentration of N 2 Increase the concentration of NH 3 Remove NH 3 from the reaction after it is made
- Slides: 23