Le Chatelier Equilibrium Review Objective Today I will

Le. Chatelier & Equilibrium Review

� Objective: �Today I will be able to: �Apply knowledge of Le Chatelier’s principle and Equilibrium Constants to practice � Evaluation/Assessment: �Informal assessment: monitoring student progress as they complete the practice �Formal assessment: analyzing student responses to the practice and exit ticket � Common Core Connection �Build Strong Content Knowledge �Make sense of problems and persevere in solving them �Reason abstactly and quantitatively

Lesson Sequence � Evaluate: Warm – Up � Evaluate: Le Chatelier/Equilibrium Practice � Evaluate: Exit Ticket

Warm-Up Complete the following question on a separate ½ sheet of paper: Given the following reaction: � Use Le Chatelier’s principle to determine what would happen if the following changes were made ◦ ◦ Increase the concentration of N 2 Increase the concentration of NH 3 Remove NH 3 from the reaction after it is made Increase volume

Objective � Today I will be able to: �Apply knowledge of Le Chatelier’s principle and Equilibrium Constants to practice

Homework � Study Quiz for a brief Equilibrium and Le. Chatelier

Agenda Warm – Up � Equilibrium/Le. Chatelier Condensed Notes � Equilibrium/Le Chatelier Review � Exit Ticket �

Quiz on Thursday � Quiz � You on Thursday is only 28 points will have 2 systems to label shifts � You will have 2 equilibrium calculation problems � Overall, the quiz may only take 15 -20 minutes!

Le Chatelier/Equilibrium Constant Condensed Notes

Equilibrium � Chemical reversible � What reactions have varying rates and are does equilibrium mean? ◦ Forward and reverse rates are equal NOT concentrations of products and reactants

Equilibrium � Dynamic Chemical Equilibrium: system is changing but there is not net reaction � How to: Equilibrium Expressions 1. Molecules written as concentrations � i. e. [A] 2. 3. 4. 5. Products go on top, reactants go on bottom Coefficients become exponents Solids and liquids are NOT included Gases and aqueous molecules are included

Le Chatelier Principle � Focus is on how equilibrium systems will respond (shift) during a stress � Stresses ◦ ◦ ◦ can come in many forms: Increase/decrease temperature Increase/decrease reactants Increase/decrease products Change volume Change pressure

A (g) + B (g) C (g) + X k. J � � Add a reactant – moves to product side to consume it Add a product – moves to reactant side to consume it Remove a reactant – moves to reactant side to produce more Remove a product – moves to product side to produce more
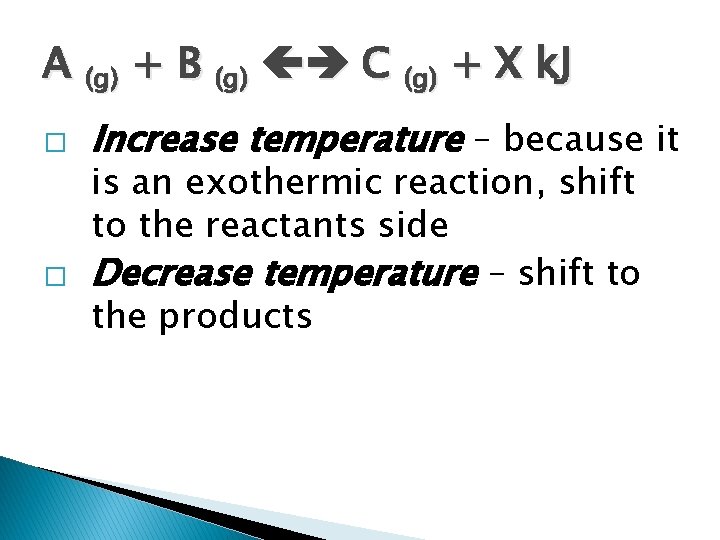
A (g) + B (g) C (g) + X k. J � � Increase temperature – because it is an exothermic reaction, shift to the reactants side Decrease temperature – shift to the products

A (g) + B (g) + X k. J C (g) � � Increase temperature – because it is an endothermic reaction, shift to the products side Decrease temperature – shift to the reactants

A (g) + B (g) C (g) + X k. J � � Increase pressure – move to side with less moles of gas (PIL) Decrease pressure – move to side with more moles of gas (PDL) Increase volume – move to side with more moles of gas (VIM) Decrease volume – move to side with less moles of gas (VDL)

Le Chatelier/Equilibrium Constant Review Lets review our homework about these two topics!
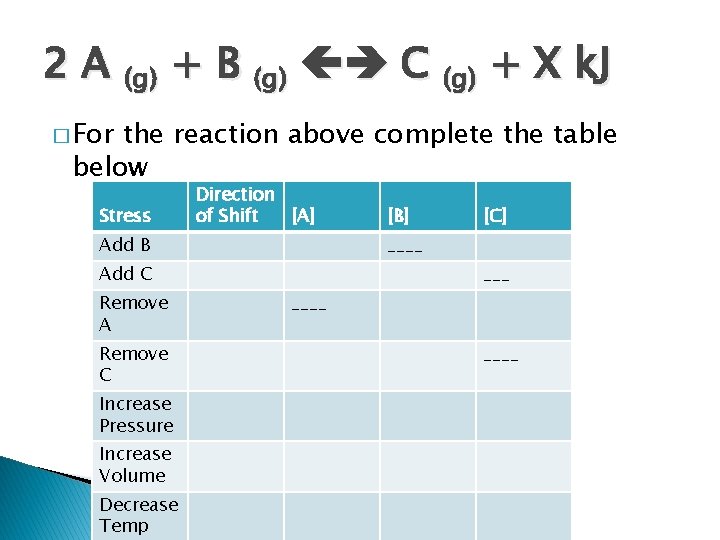
2 A (g) + B (g) C (g) + X k. J � For the reaction above complete the table below Stress Direction of Shift [A] Add B Remove C Increase Pressure Increase Volume Decrease Temp [C] ____ Add C Remove A [B] ____
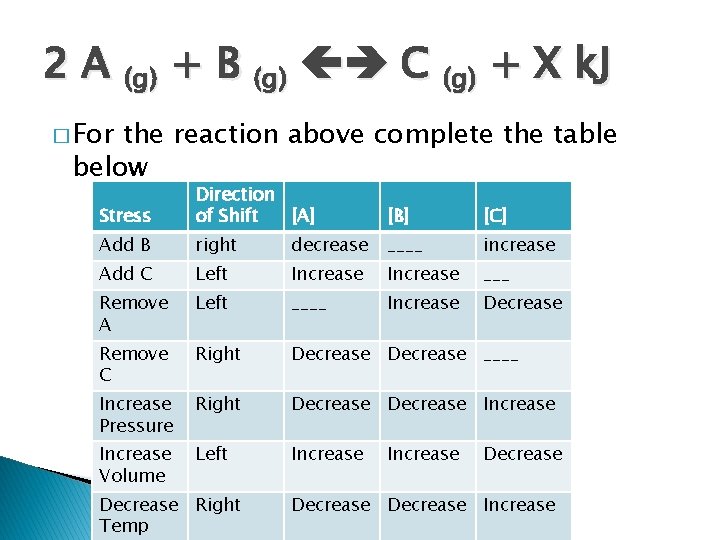
2 A (g) + B (g) C (g) + X k. J � For the reaction above complete the table below Stress Direction of Shift [A] [B] [C] Add B right decrease ____ increase Add C Left Increase ___ Remove A Left ____ Increase Decrease Remove C Right Decrease ____ Increase Pressure Right Decrease Increase Volume Left Increase Decrease Right Temp Increase Decrease Increase

Exit Discussion � Any questions regarding equilibrium and Le. Chatelier?
- Slides: 20