Le 050310 IPSEN 1 Sophie Compagnon Melodie Debacker

Le 05/03/10 IPSEN 1 Sophie Compagnon, Melodie Debacker, Gaelle Datchoua, Catherine Lucas

INTRODUCTION 2

About IPSEN • Specialized world pharmaceutical group • More than 20 products marketed in the world • Originally, French independent company • Headed by Beaufour Family • Third independent pharmaceutical firm in France (sales) 3
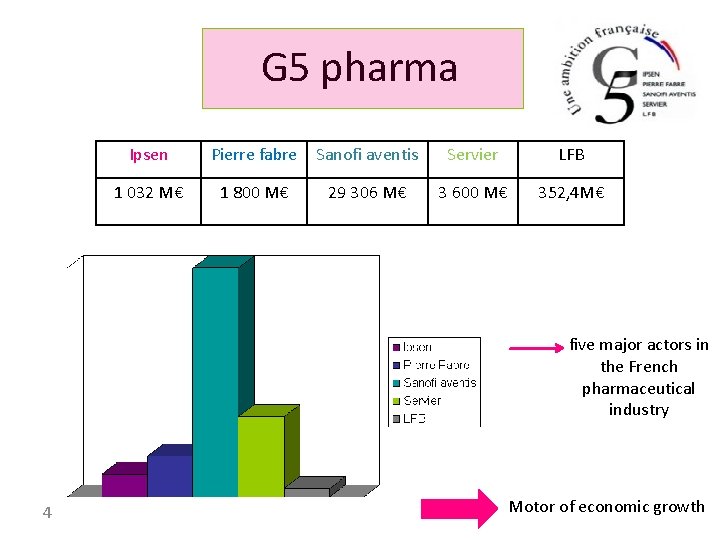
G 5 pharma Ipsen Pierre fabre Sanofi aventis Servier LFB 1 032 M€ 1 800 M€ 29 306 M€ 3 600 M€ 352, 4 M€ five major actors in the French pharmaceutical industry 4 Motor of economic growth
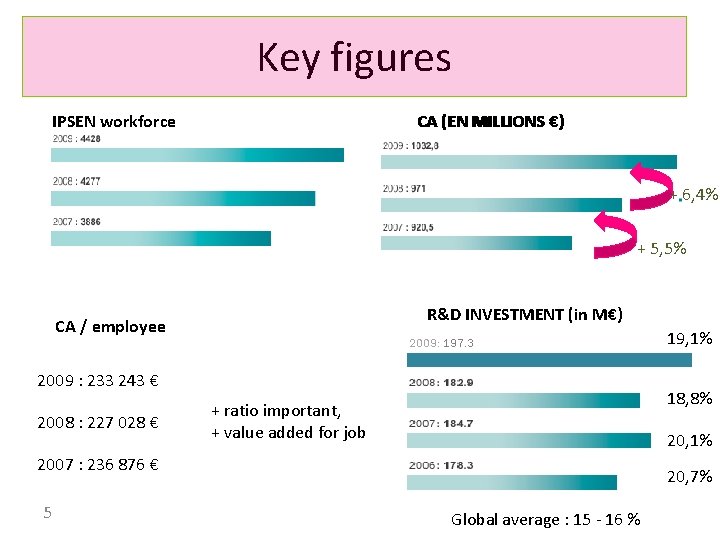
Key figures CA (EN MILLIONS €) IPSEN workforce + 6, 4% + 5, 5% R&D INVESTMENT (in M€) CA / employee 2009: 197. 3 2009 : 233 243 € 2008 : 227 028 € 18, 8% + ratio important, + value added for job 20, 1% 2007 : 236 876 € 5 19, 1% 20, 7% Global average : 15 - 16 %
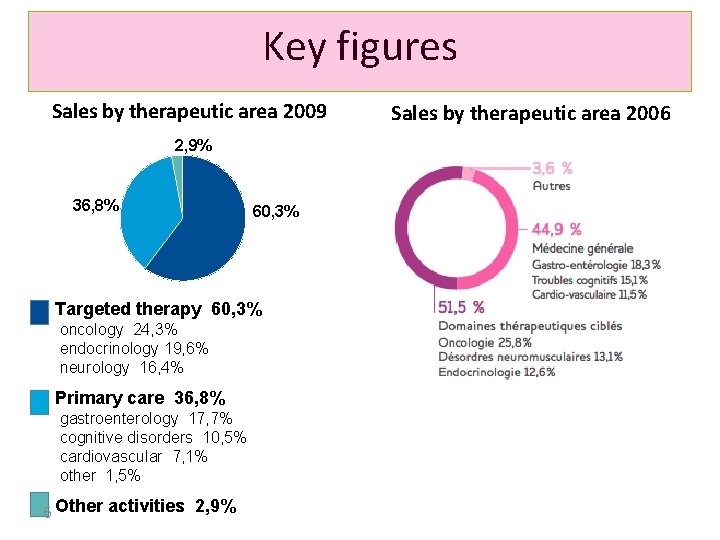
Key figures Sales by therapeutic area 2009 2, 9% 36, 8% 60, 3% Targeted therapy 60, 3% oncology 24, 3% endocrinology 19, 6% neurology 16, 4% Primary care 36, 8% gastroenterology 17, 7% cognitive disorders 10, 5% cardiovascular 7, 1% other 1, 5% 6 Other activities 2, 9% Sales by therapeutic area 2006
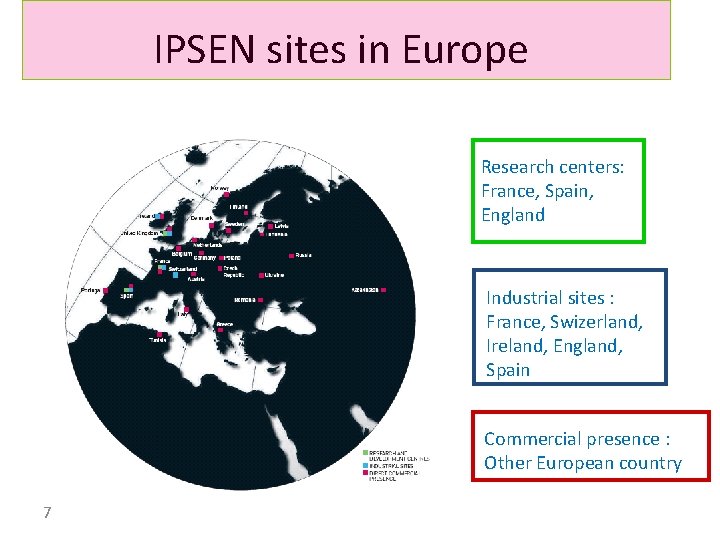
IPSEN sites in Europe Research centers: France, Spain, England Industrial sites : France, Swizerland, Ireland, England, Spain Commercial presence : Other European country 7
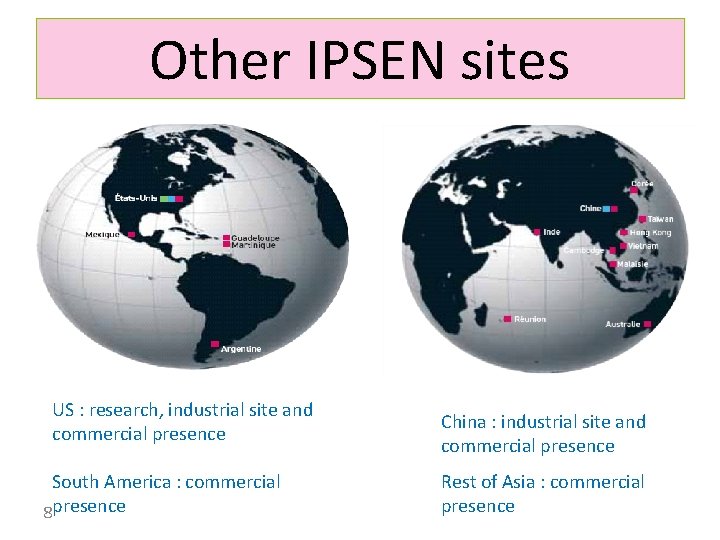
Other IPSEN sites US : research, industrial site and commercial presence South America : commercial 8 presence China : industrial site and commercial presence Rest of Asia : commercial presence
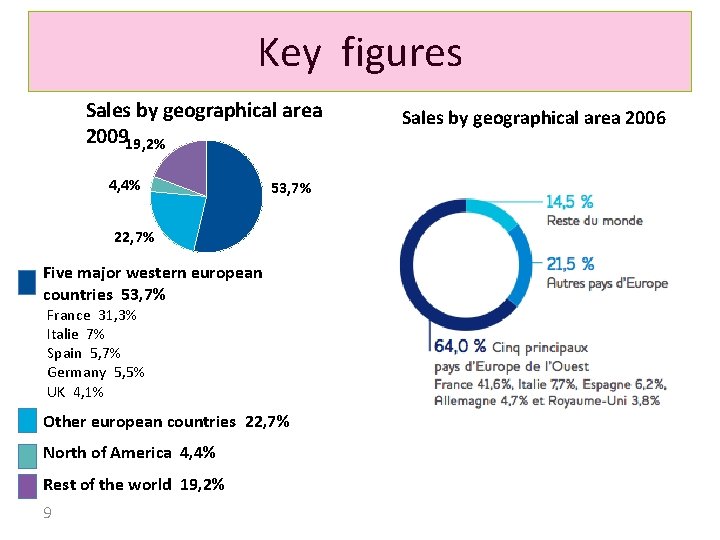
Key figures Sales by geographical area 200919, 2% 4, 4% 53, 7% 22, 7% Five major western european countries 53, 7% France 31, 3% Italie 7% Spain 5, 7% Germany 5, 5% UK 4, 1% Other european countries 22, 7% North of America 4, 4% Rest of the world 19, 2% 9 Sales by geographical area 2006

BACKGROUND 10

IPSEN background 1929 Beaufour Laboratories • Founded by Dr Henri Beaufour (pharmacist) in Dreux • Launch of Romarène® digestive disorders • Launch of Citrate de bétaïne® Beaufour dyspepsia • Integration of Henri’s sons, Gerard and Albert, in Beaufour Laboratories 1963 11 • Launch of Actapulgite® antacid, gastric demulcent and antidiarrhoeal 1954

70’s IPSEN background 70’s Start marketing products of natural origin. 1970 « IPSEN » creation Subsidiary company of « Beaufour Laboratories » = Institut des Produits de Synthèse et d’Extraction Naturelle Gingko biloba 12 Primary care

70’s IPSEN background 70’s Start marketing products of natural origin. 1972 Ginkor® Indications: Cardiovascular Active substances: Ginkgo biloba extract chronic venous insufficiency Heptaminol chlorhydrate acute haemorrhoid episodes Troxérutine superficial phlebitis 1987: Ginkor Procto® 1988: Ginkor Fort® 13 Primary care

70’s IPSEN background 70’s Start marketing products of natural origin. 1972 Tanakan® Active substances: EGb 761® (Ginkgo biloba standardized extract) Indications: Cognitive disorders Age-related cognitive disorders (excepted Alzheimer’s disease) Anti-ischemic (atreritis, Raynaud’s phenomenon) cochleovestibular disorders 14 Primary care

70’s IPSEN background 70’s Start marketing products of natural origin. 1977 Smecta® Active substances: Indications: Gastroenterology diosmectite (clay) Digestive coating (pain of œsopagus, stomach, duodenum and colon) Acute and chronic diarrhea 1980 Active substances: montmorillonite beidellitique (clay) 15 Bedelix® Indications: Gastroenterology Digestive coating Symptomatic treatment of functionnal colopathies Primary care

90’s IPSEN background 1996 90’s Forlax® Active substances: Macrogol 4000 (polyethylène glycol 4000) Indications: Gastrointestinal constipation (adults and children) 16 Primary care

Start in research • Albert beaufour : biotechnology research 1976 Biomeasure Boston A. Beaufour research institute : peptide focused engineering • Research relations with american universities. 17

80’s IPSEN background 80’s Interest in specialized therapies 1980 Partnership with • International development • Launch of Decapeptyl ®(1986) Active substances: triptoreline (Gn. RH agonist) Indications: Oncology Prostate cancer Uterin fibroids Endometriosis Early-onset puberty Female infertility 18 Decapeptyl® 1 mois (1995) Decapeptyl® 3 mois (1996) Decapeptyl® 6 mois (2009, november) Targeted therapy
90’s IPSEN background 90’s Interest in specialized therapies 1994 Dysport® Purchase of KEYWOOD (UK) Active substances: botilium neurotoxin, type A toxin-haemagglutinin complex. Indications: Neuromuscular > 75 countries hemifacial spasticity Cervical dystonia blepharospasm (glabellaires wrinkles) 19 Targeted therapy
90’s IPSEN background 90’s Interest in specialized therapies 1995 Somatuline® Active substance: Lanréotide = an octapeptide somatostatin analog Inhibition of Insulin-like growth factor-1 (IGF-1) and growth hormone (GH). Indications: Endocrinology Acromegaly Neuroendocrine tumours Somatuline Depot ® 1 st product from IPSEN R&D approved by the FDA and available at international level 20 Targeted therapy

90’s IPSEN background 90’s Family business Disagreements beetwen Albert and Gerard, at last Albert bought Gerard’s parts (50%) 1996 Still an independent family company Progressively, Stephane Francois, son-in-law of Albert, has more importance. 21

90’s 2001 IPSEN background 90’s Death of Albert. Stephane Francois is renowned CEO, but Anne and Henri Beaufour don’t agree with that. Stephane Francois is fired. 2002 Welcome to Jean-Luc Bélingard ! 22

New team J. L Bélingard Anne Beaufour Claire Giraut • Chairman and Chief Executive Offi cer. • Delegate General and spokesman for G 5 2003: Director Chief Financial Offi cer 1999 -2001, he was a member of the Executive Board and CEO of Bio. Mérieux. Pierre Fabre. bachelor’s member degree of thein. Executive geology. director Board ofof the Company Technip since Group 1998. Eric Drapé Henri Beaufour Executive Seniorof. Vice Director Vice-President, Bachelor arts. President degree. of the company’s Manufacturing and Supply. Since Diabetes Finishedof. Products at Novo(LXB) 2003, manager Camilia Holding Organisation. Nordisk Jacques-Pierre Moreau Executive Vice-President, Chief Scientifi c Offi cer founded Biomeasure Incorporated, based near Boston Frédéric Babin Executive Vice-President, Human Resources Master of Business Law Christophe Jean Group Vice-President Operations Chairman and CEO of Pierre Fabre Médicaments. 23
NEW BOSS. . NEW STRATEGY 24 1 • Optimize its primary care portfolio 2 • Geographic expansion 3 • Focus on targeted therapeutic areas
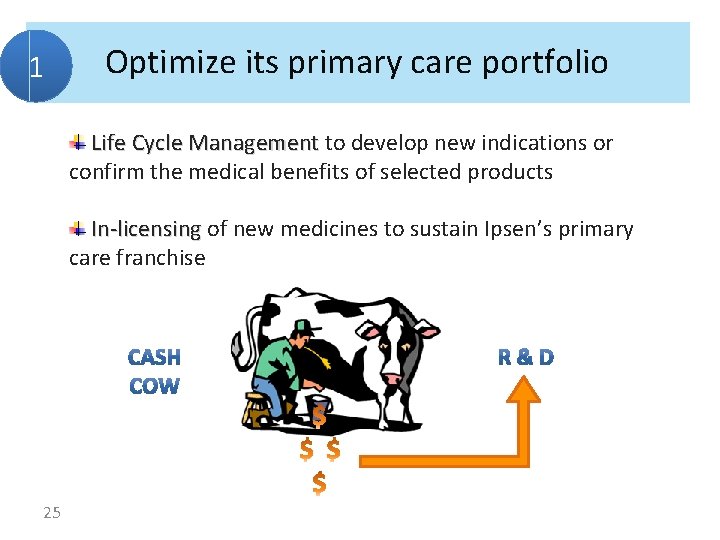
1 Optimize its primary care portfolio Life Cycle Management to develop new indications or confirm the medical benefits of selected products In-licensing of new medicines to sustain Ipsen’s primary care franchise 25
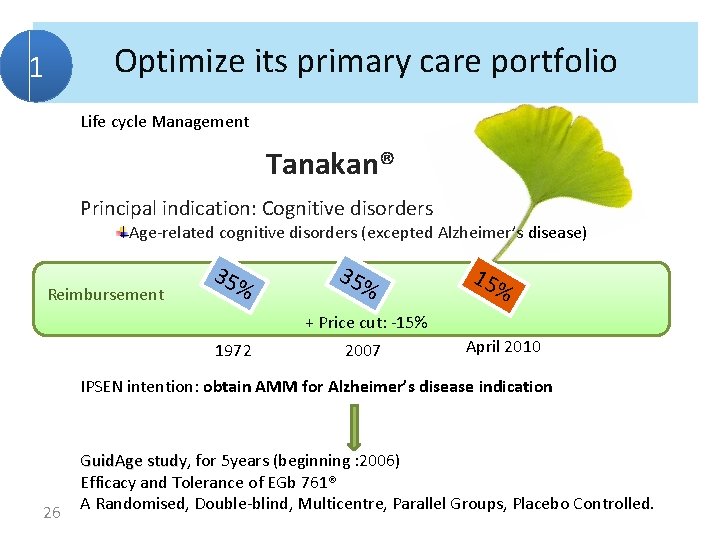
Optimize its primary care portfolio 1 Life cycle Management Tanakan® Principal indication: Cognitive disorders Age-related cognitive disorders (excepted Alzheimer’s disease) Reimbursement 35% 15 % + Price cut: -15% 1972 2007 April 2010 IPSEN intention: obtain AMM for Alzheimer’s disease indication 26 Guid. Age study, study for 5 years (beginning : 2006) Efficacy and Tolerance of EGb 761® A Randomised, Double-blind, Multicentre, Parallel Groups, Placebo Controlled.

Optimize its primary care portfolio 1 Life cycle Management Reimbursement Ginkor Fort® 35% 15 0% % + Price cut: -15% 1972 Turnover decrease 2005 2006 2007 2008 39. 7% Marketing authorizations in France, Andorra and Monaco relinquished to in august 2007 27
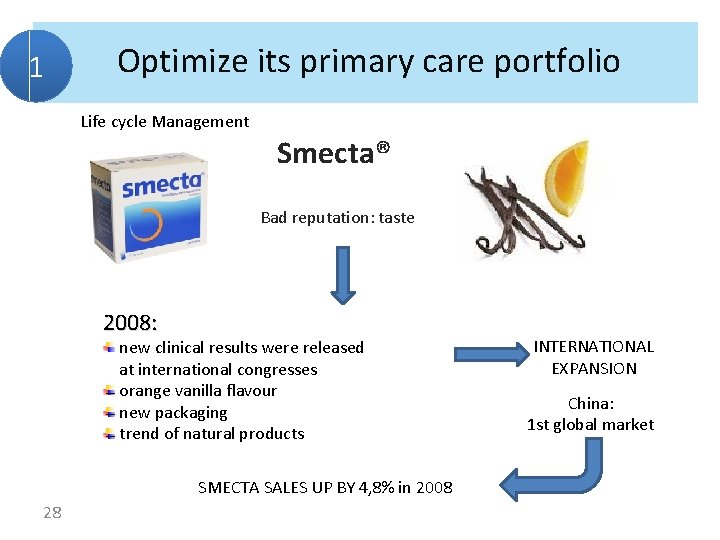
1 Optimize its primary care portfolio Life cycle Management Smecta® Bad reputation: taste 2008: new clinical results were released at international congresses orange vanilla flavour new packaging trend of natural products SMECTA SALES UP BY 4, 8% in 2008 28 INTERNATIONAL EXPANSION China: 1 st global market

Optimize its primary care portfolio 1 In-licensing of new medicines Co-promotion agreements Hypertension 2003 Nisis® and Nisisco ® 2009 Exforge® Non steroidal anti-inflammatory drug 2006 Artotec® Post-menopausal osteoporosis 2007 Adrovance® Hyperuricemia 29 2008 Adenuric ® France Europe
NEW BOSS. . NEW STRATEGY 30 1 • Optimize its primary care portfolio 2 • Geographic expansion 3 • Focus on targeted therapeutic areas
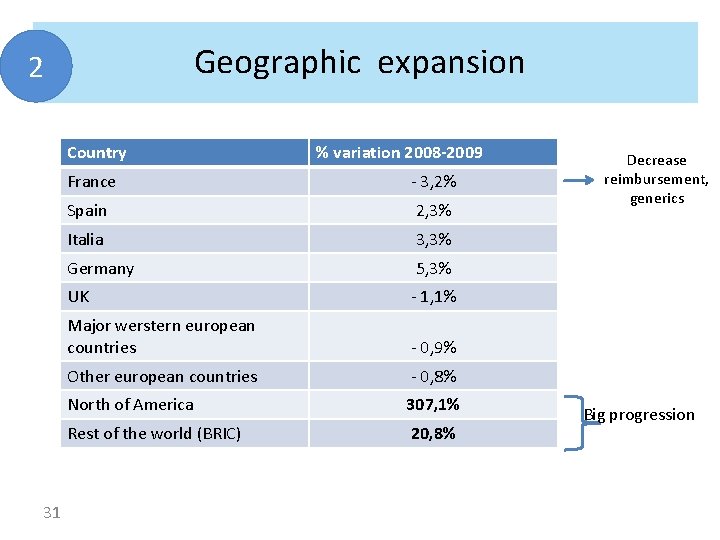
Geographic expansion 2 Country 31 % variation 2008 -2009 France - 3, 2% Spain 2, 3% Italia 3, 3% Germany 5, 3% UK - 1, 1% Major werstern european countries - 0, 9% Other european countries - 0, 8% North of America 307, 1% Rest of the world (BRIC) 20, 8% Decrease reimbursement, generics Big progression

2 Example of partnership with Inspiration founded in 2004 • mission: revolutionize treatment for hemophilia • Wide portfolio in hemophilia and haemorrhage • OBI-1 : porcine FVIII • Reminder: Hemophilia A = deficit in FVIII controlled by injections of human FVIII ( 2 – 3 t/ week) treatment of hemophilia A Pb: 1/3 of cases, immune reaction (Ac anti human FVIII) preventing reinjection of human FVIII • So OBI-1 : porcine origin low cross-reactivity to Ac anti h. FVIII therapeutic benefit for these patients IPSEN ambition: engage in a niche sector 32
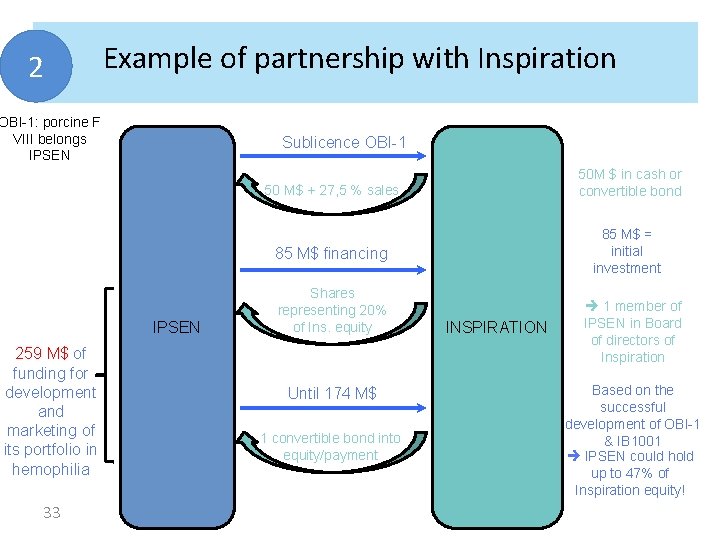
2 Example of partnership with Inspiration OBI-1: porcine F VIII belongs IPSEN Sublicence OBI-1 50 M $ in cash or convertible bond 50 M$ + 27, 5 % sales 85 M$ = initial investment 85 M$ financing IPSEN 259 M$ of funding for development and marketing of its portfolio in hemophilia 33 Shares representing 20% of Ins. equity Until 174 M$ 1 convertible bond into equity/payment INSPIRATION 1 member of IPSEN in Board of directors of Inspiration Based on the successful development of OBI-1 & IB 1001 IPSEN could hold up to 47% of Inspiration equity!
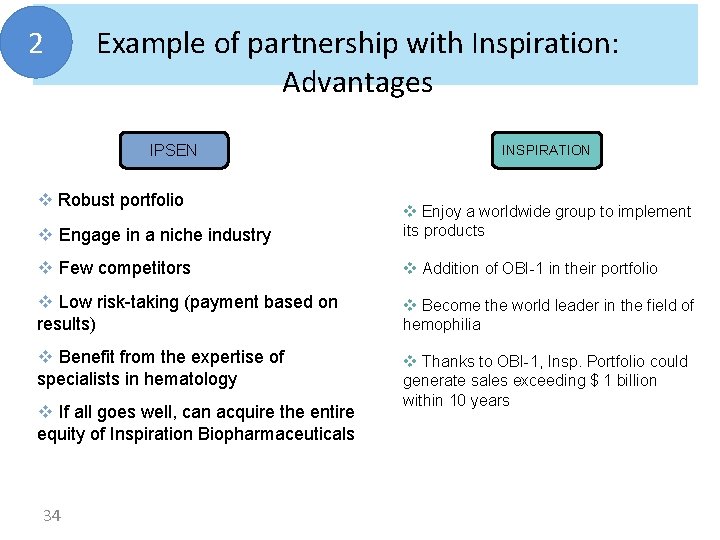
2 Example of partnership with Inspiration: Advantages IPSEN v Robust portfolio INSPIRATION v Engage in a niche industry v Enjoy a worldwide group to implement its products v Few competitors v Addition of OBI-1 in their portfolio v Low risk-taking (payment based on results) v Become the world leader in the field of hemophilia v Benefit from the expertise of specialists in hematology v Thanks to OBI-1, Insp. Portfolio could generate sales exceeding $ 1 billion within 10 years v If all goes well, can acquire the entire equity of Inspiration Biopharmaceuticals 34

2 So … Realize partnership permits to IPSEN: – sell products that are not in its field (little knowledge) – engage with partners only if it can make a profit – eventually buy out his partner / no longer care 35
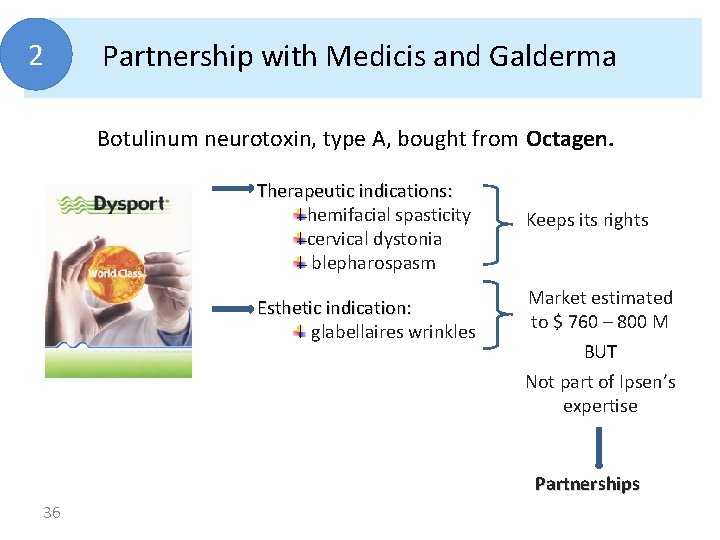
2 Partnership with Medicis and Galderma Botulinum neurotoxin, type A, bought from Octagen. Therapeutic indications: hemifacial spasticity cervical dystonia blepharospasm Esthetic indication: glabellaires wrinkles Keeps its rights Market estimated to $ 760 – 800 M BUT Not part of Ipsen’s expertise Partnerships 36
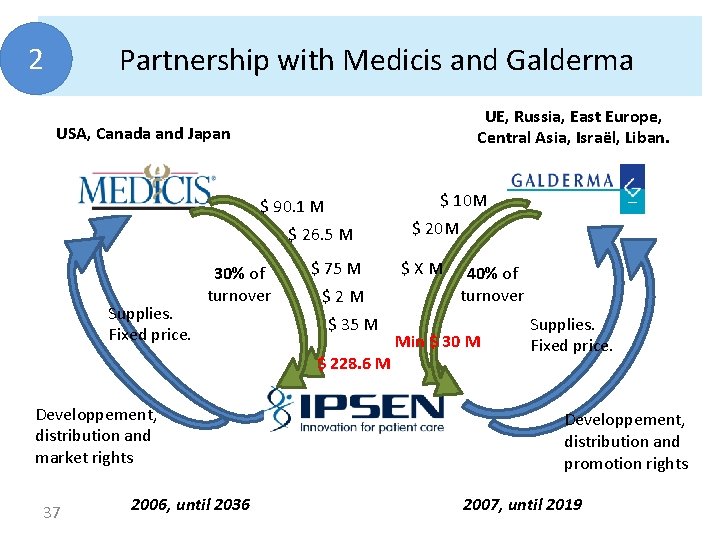
2 Partnership with Medicis and Galderma UE, Russia, East Europe, Central Asia, Israël, Liban. USA, Canada and Japan $ 10 M $ 90. 1 M $ 26. 5 M Supplies. Fixed price. 30% of turnover $ 75 M $2 M $ 35 M $ 228. 6 M Developpement, distribution and market rights 37 2006, until 2036 $ 20 M $XM 40% of turnover Min $ 30 M Supplies. Fixed price. Developpement, distribution and promotion rights 2007, until 2019
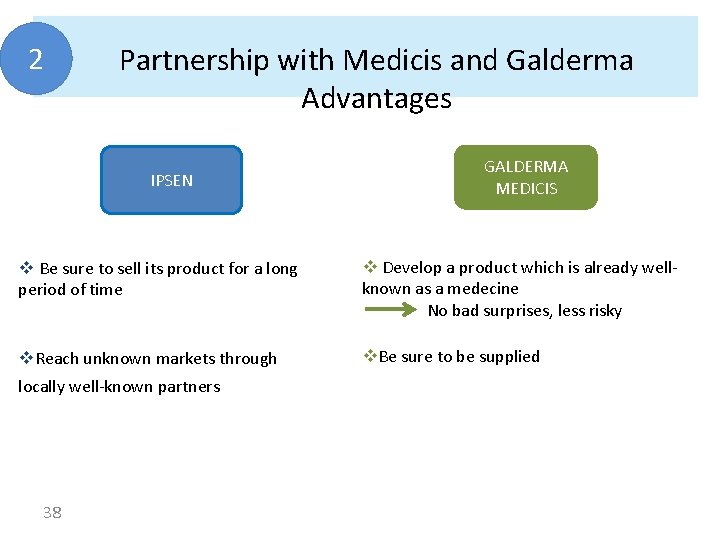
2 Partnership with Medicis and Galderma Advantages IPSEN GALDERMA MEDICIS v Be sure to sell its product for a long period of time v Develop a product which is already wellknown as a medecine No bad surprises, less risky v. Reach unknown markets through v. Be sure to be supplied locally well-known partners 38
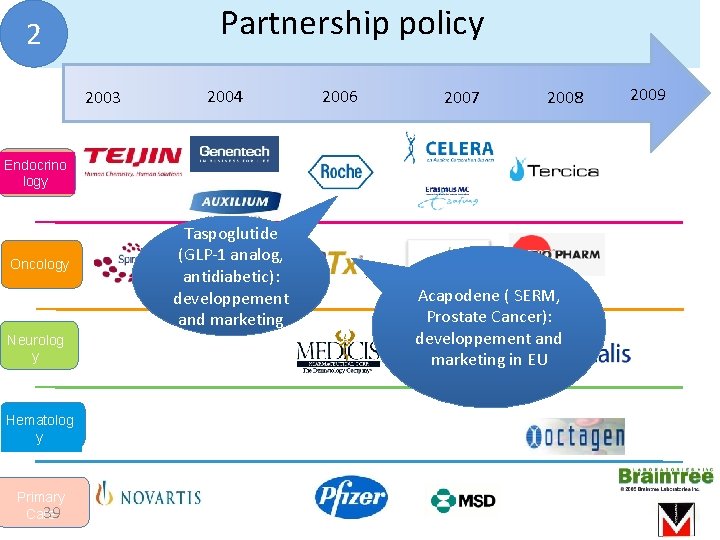
Partnership policy 2 2003 2004 2006 2007 2008 Endocrino logy Oncology Neurolog y Hematolog y Primary 39 Care Taspoglutide (GLP-1 analog, antidiabetic): developpement and marketing Acapodene ( SERM, Prostate Cancer): developpement and marketing in EU 2009
NEW BOSS. . NEW STRATEGY 40 1 • Optimize its primary care portfolio 2 • Geographic expansion 3 • Focus on targeted therapeutic areas
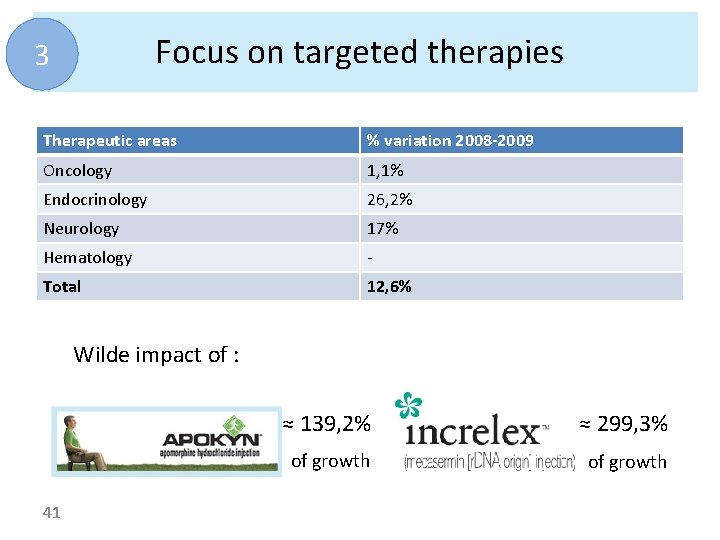
Focus on targeted therapies 3 Therapeutic areas % variation 2008 -2009 Oncology 1, 1% Endocrinology 26, 2% Neurology 17% Hematology - Total 12, 6% Wilde impact of : 41 ≈ 139, 2% ≈ 299, 3% of growth

3 R&D q Early commitment, since 1969 q More 800 personnes in R&D q About 20% of CA for R&D every year q Currently 20 programs 42

3 International knowledge 4 research centers: q. Paris : medicinal chemistry and development q. Boston : engineering of peptides and proteins q. Londres : synthesis of steroid hormones q. Barcelona : drug formulation 43
3 Partnership agreements q Salk institute (USA) : « the Ipsen Life Sciences Program » for proliferative and degenerative diseases. q CEA (France) : for Alzheimer and Parkinson diseases. q ERINE (Netherland) : therapeutic concepts of endocrinology, diabetes and metabolism disorders. q INSERM : breast, prostate tumors 44
Pipeline 3 Their identity 45

3 Preclinical Oncology : q Angiomates : anticancer agent, anti-angiogenic acquisition of Sterix q BIM 46187 : Cell signalisation, G-protein signal for prostate and lung cancer q Cd 25 inhibitors phosphatases : regulation of cell cycle Licencied out to Debiopharm for world exclusive right 46
3 Preclinical Endocrinology : q MSH agonists for the MC 4 receptor : metabolic disorders like obesity q Ghrelin agonists (BIM 28131) : regulation of food intake and gastro intestinal function and treatment of cachexia q Inhibitor of 11βHSB : for metabolic disorders Growing markets q Nutropin. Ap® : treatment of growth failure, new formulation for prolonged release Life cycle management 47
3 Phase 1 Oncology : q Elomotecan : cytotoxic agent, advanced metastasic cancer. Partnership with Roche in dec 2002, but Roche quits in may 2005. 48
3 Phase 2 Oncology : q Diflomotecan : topoisomerase inhibitor less SE than other q BN 83495 : breast cancer expressing oestrogen receptors partnership with Biomérieux for diagnosis Endocrinology : q IGF-1 and growth hormone : combination therapy for the treatment of idiopathic short stature. Combo q BIM 23 A 760 : somatostatine analog and dopamine agonist for acromegaly and neuroendocrine tumors 49 Better wide activity with synergy
3 Phase 3 Oncology : q Decapeptyl® : combined hormone therapy for premenopausal breast cancer wirh Aromasin® Pfizer Combo q Toremifene® : GTX licence buyout Hematology : q OBI-1 : hemophilia 50 Pig fact VIII Development of new indications
Phase 3 3 Endocrinology : q Somatuline® Autogel® Life cycle management q Somatuline® Depot® q Increlex® : primary deficit in IGF-1 Global actor in the growth disorders q Taspoglutide : type 2 diabetes (partnership with Roche) GLP 1, third on the market 51

3 Neurology : Research program : new formulation for botulique toxin. Implant on the world market of Dysport® with Galderma, and Medicis and of Apokin® with Vernalis buyout 52

3 New authorization in 2009 • Décapeptyl® 6 months : launch in France. • Dysport® : launch in USA for cosmetic and therapeutic indications. • Azzalure® : marketing authorization in Spain, Germany, France and UK, and launch by Galderma. 53

54 IPSEN gives itself the means of a strong and sustainable growth

History of stockmarket 55

IPSEN stockmarket 1998 56 Consideration of introduction in stockmarket, year of the opening of the capital of the laboratory to the caisse de dépots and paribas affaires industrielles, which then took a participation of 20% 2003 The conditions of market have not been favorable. Wait until they improve 2005 Initial Public Offering in december
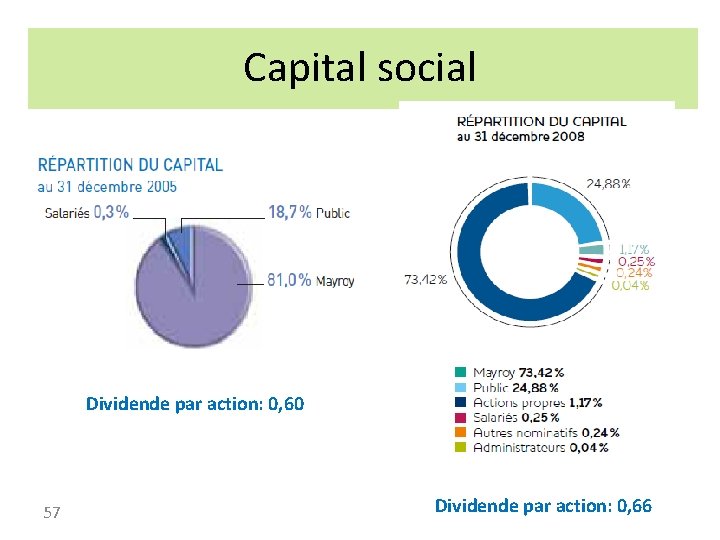
Capital social Dividende par action: 0, 60 57 Dividende par action: 0, 66
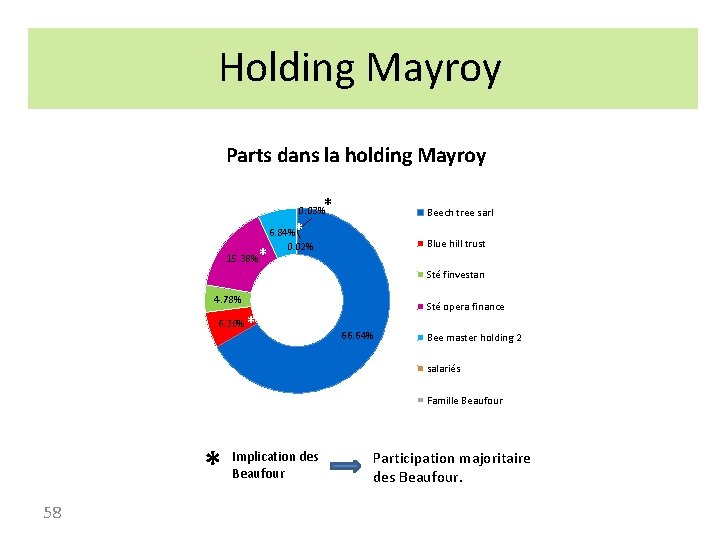
Holding Mayroy Parts dans la holding Mayroy * 0. 03% Beech tree sarl * * 15. 38% 6. 84% 0. 02% Blue hill trust Sté finvestan 4. 78% 6. 30% * Sté opera finance 66. 64% Bee master holding 2 salariés * Famille Beaufour * 58 Implication des Beaufour Participation majoritaire des Beaufour.
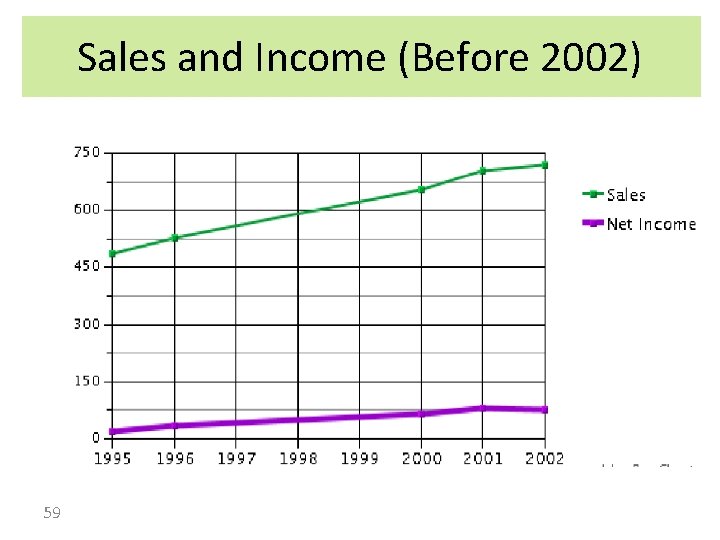
Sales and Income (Before 2002) 59
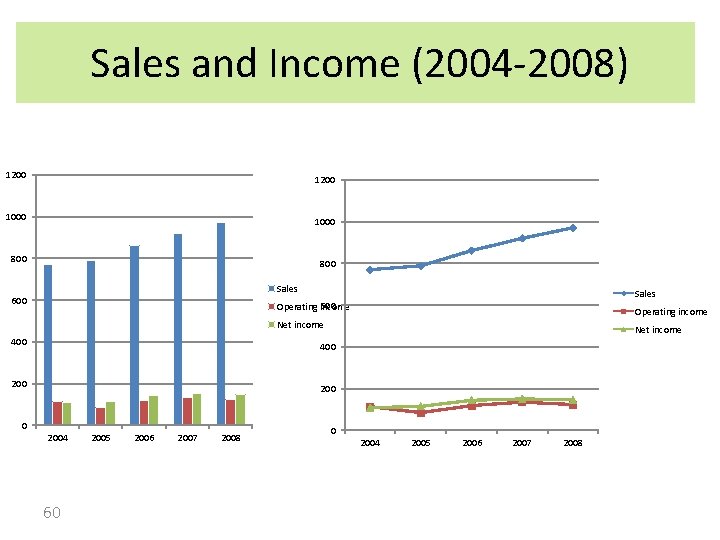
Sales and Income (2004 -2008) 1200 1000 800 Sales 600 Operating income Net income 400 2004 60 2005 2006 2007 2008 0 2004 2005 2006 2007 2008
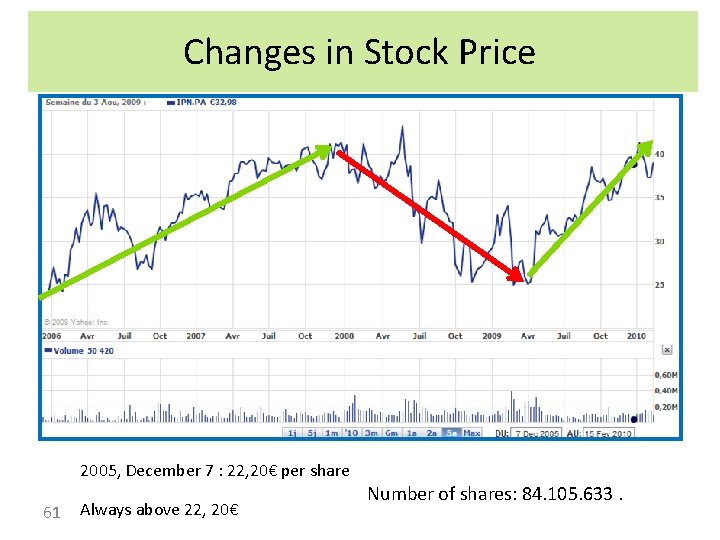
Changes in Stock Price 2005, December 7 : 22, 20€ per share 61 Always above 22, 20€ Number of shares: 84. 105. 633.
Conclusion 62
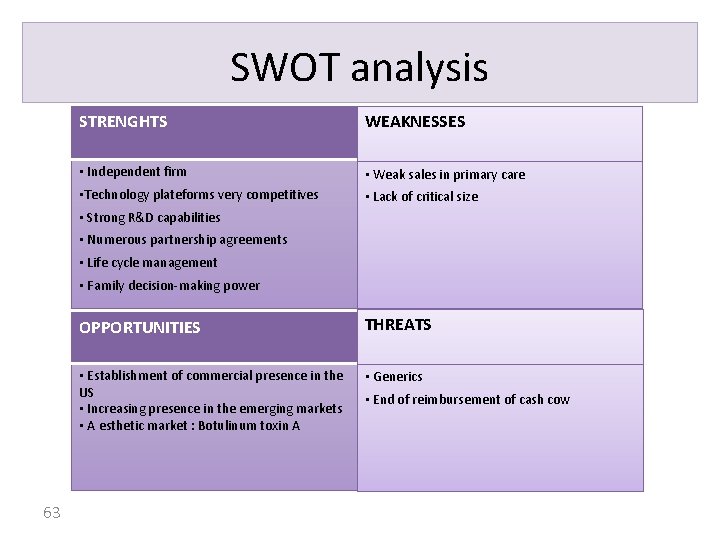
SWOT analysis STRENGHTS WEAKNESSES • Independent firm • Weak sales in primary care • Technology plateforms very competitives • Lack of critical size • Strong R&D capabilities • Numerous partnership agreements • Life cycle management • Family decision-making power 63 OPPORTUNITIES THREATS • Establishment of commercial presence in the US • Increasing presence in the emerging markets • A esthetic market : Botulinum toxin A • Generics • End of reimbursement of cash cow

What do you think about IPSEN? Catherine : + strong R&D - leave out of primary care Sophie : + strong R&D + partnership organisation 64 PRODUCTION R&D Gaelle : + partnership organisation + growth DEVPT GALENIQUE Mélodie : + partnership organisation + strategy MARKETING STRATEGIQUE
Thanks for your attention Any questions ? 65
Embauches au sein du groupe 66

Partnership policy Many partnership to enrich R&D portfolio and expand products range → R&D collaboration : Market Ipsen’s products in Japan → R&D agreement and acquisition 20% of CA : new technologies Oct 09 : new agreement SG 2000 (anticancer agent, 1 st class) → Marketing licence for Nisis® & Nisisco® Jan 09: co-promotion agreement Exforge® in France → R&D agreement formulation growth hormone LP → marketing licence for Testim® in France → Development and marketing BIM 51077 67 → development and marketing licence Acapodène® in UE

Partnership policy → development and marketing licence Toxine botulique (dysport®, Azzalure®) → R&D agreement for development of pharmacogenomic tests for growth retardation → R&D agreement for development of innovative products in diabete, endocrinology and metabolism → R&D agreement for development of diagnostic test in breast cancer → co-marketing licence Adrovance® TM in France → definitive merger agreement july 06: licence agreement Somatuline® Autogel® → marketing licence Triptoreline under several brandnames (Salvatyl LP®) 1983: 1 st development and marketing licence Decapeptyl® 68 → final purchase rights Apokyn® (PK)

Partnership policy → purchase agreement, recovery of all actives related with OBI-1 → development and marketing licence BLI-800 : colic washing before coloscopy → exclusive right of licence for Menarini Adenuric® 69
- Slides: 69