Layer Silicate Clay Structure basic building blocks 1
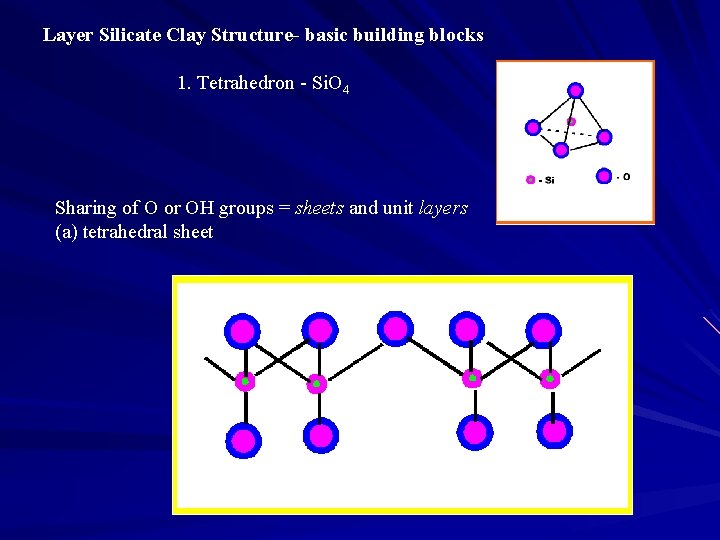
Layer Silicate Clay Structure- basic building blocks 1. Tetrahedron - Si. O 4 Sharing of O or OH groups = sheets and unit layers (a) tetrahedral sheet
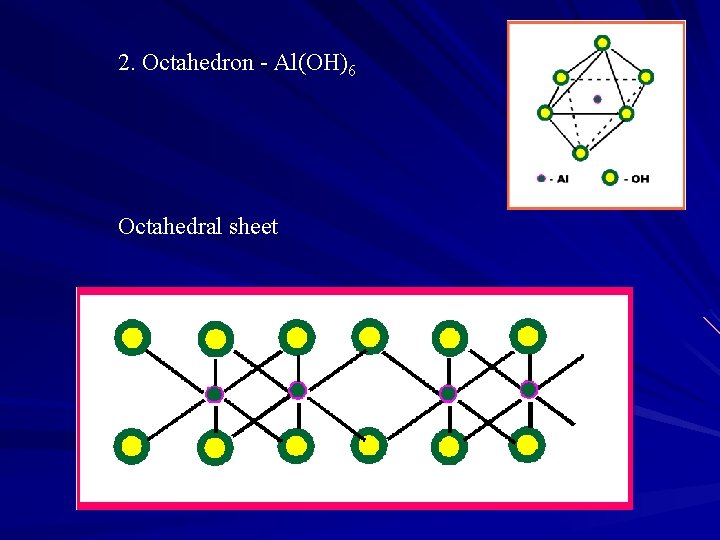
2. Octahedron - Al(OH)6 Octahedral sheet
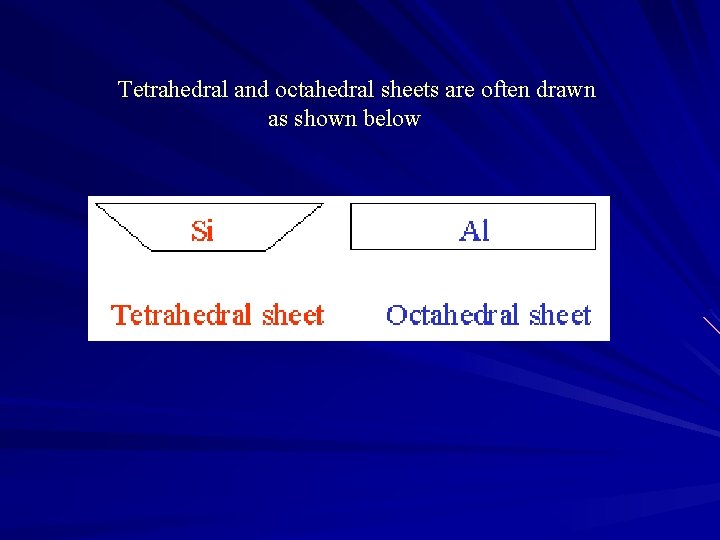
Tetrahedral and octahedral sheets are often drawn as shown below
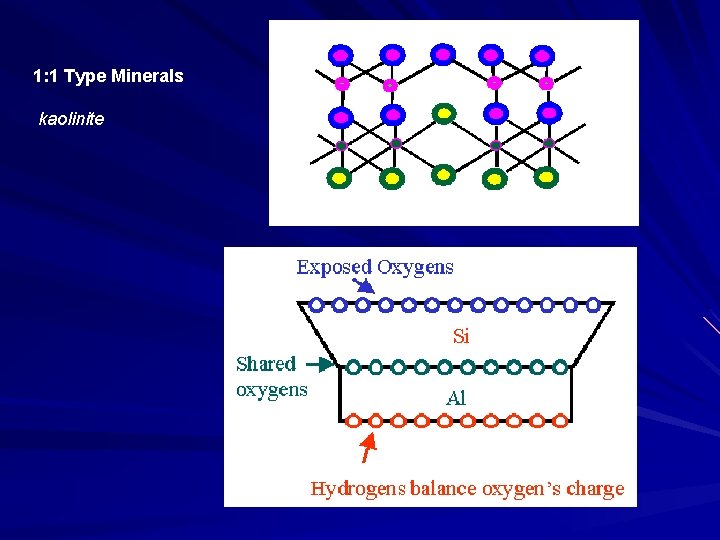
1: 1 Type Minerals kaolinite
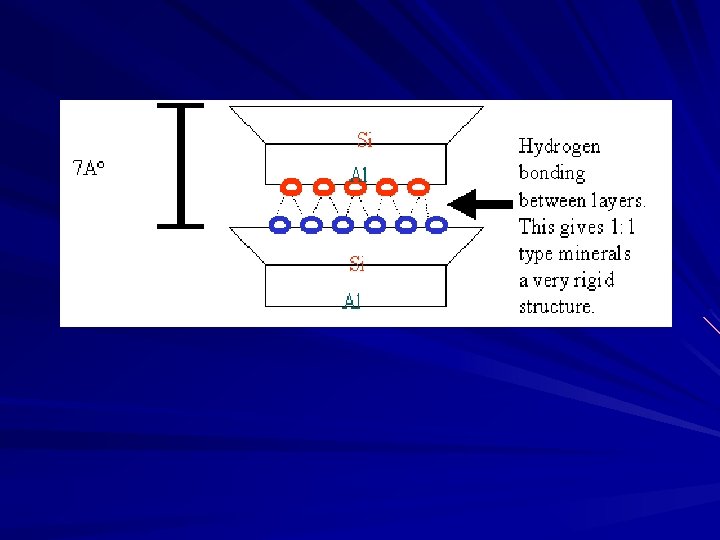
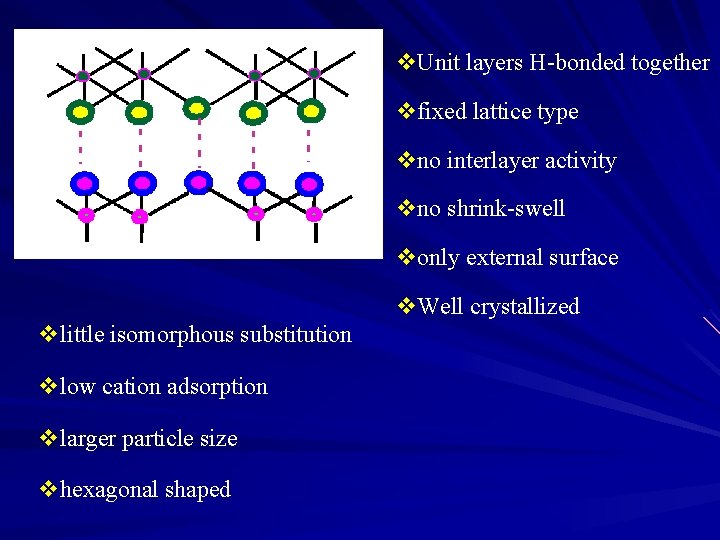
v. Unit layers H-bonded together vfixed lattice type vno interlayer activity vno shrink-swell vonly external surface v. Well crystallized vlittle isomorphous substitution vlow cation adsorption vlarger particle size vhexagonal shaped
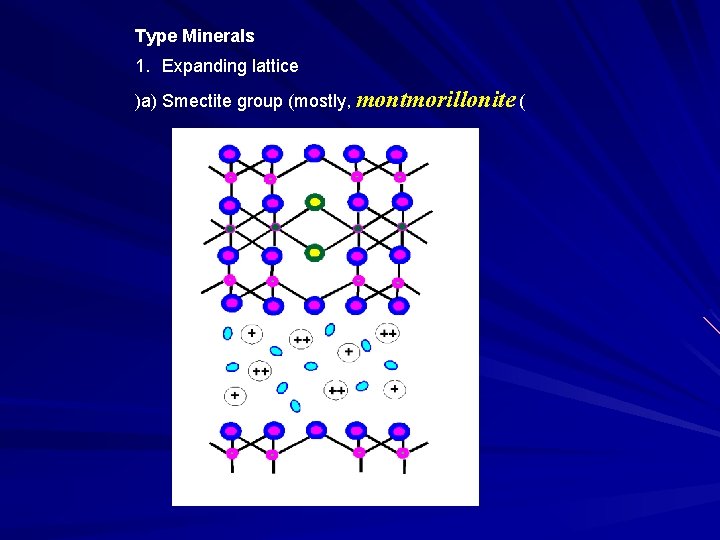
Type Minerals 1. Expanding lattice )a) Smectite group (mostly, montmorillonite (
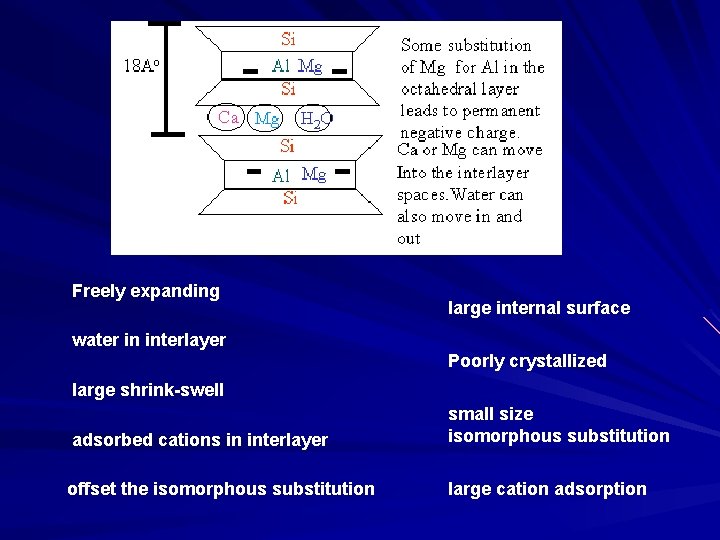
Freely expanding large internal surface water in interlayer Poorly crystallized large shrink-swell adsorbed cations in interlayer small size isomorphous substitution offset the isomorphous substitution large cation adsorption

Vermiculite Similar to smectites Except Al+3 for Si+4 in tetrahedral layer Interlayer ions are more structured (Mg+2 + H 2 O) Limited expansion Large cation adsorption
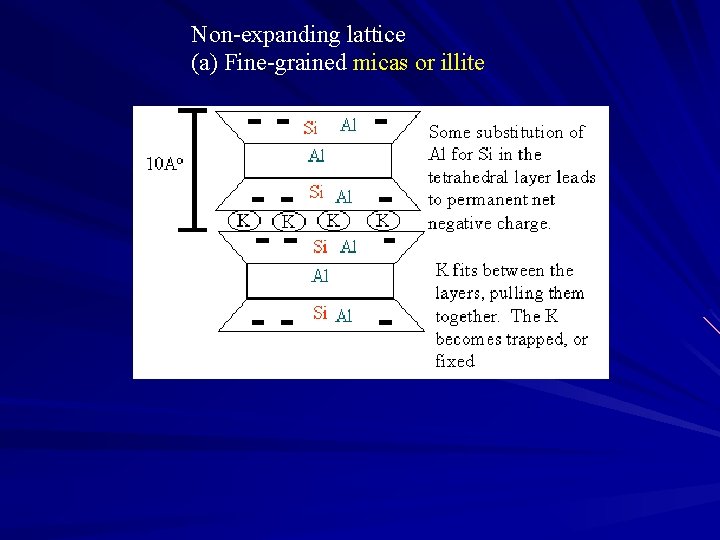
Non-expanding lattice (a) Fine-grained micas or illite

Al+3 and K +substitute for Si+4 )tetrahedral sheet) weathering at edges = release of K+ -very limited expansion - medium cation adsorption - limited internal surface - properties between kaolinite and vermiculite
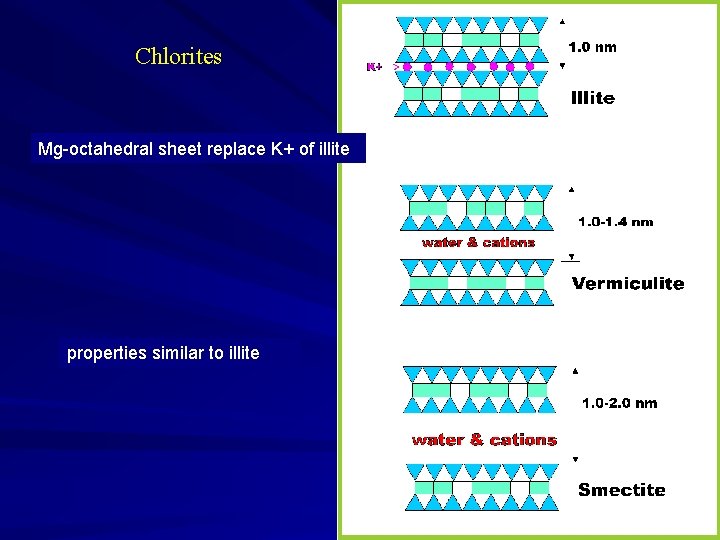
Chlorites Mg-octahedral sheet replace K+ of illite properties similar to illite
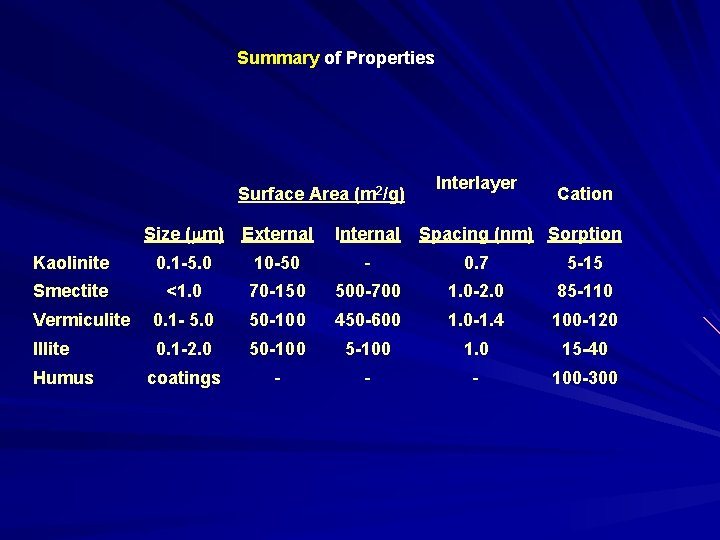
Summary of Properties Surface Area (m 2/g) Interlayer Cation Size (mm) External Internal Kaolinite 0. 1 -5. 0 10 -50 - 0. 7 5 -15 Smectite <1. 0 70 -150 500 -700 1. 0 -2. 0 85 -110 Vermiculite 0. 1 - 5. 0 50 -100 450 -600 1. 0 -1. 4 100 -120 Illite 0. 1 -2. 0 50 -100 5 -100 1. 0 15 -40 coatings - - - 100 -300 Humus Spacing (nm) Sorption


Source of charge on clays- permanent or variable? 1. Permanent or constant charge )a) isomorphous substitution for example : Zn+2 for Al+3 in octahedral sheet b) both octahedral and tetrahedral sheets Mg+2, Zn+2, Cu+2, Fe+2 for Al+3 for Si+4 )c) can form + charge - Al+3 for Mg+2 - negative charge dominates

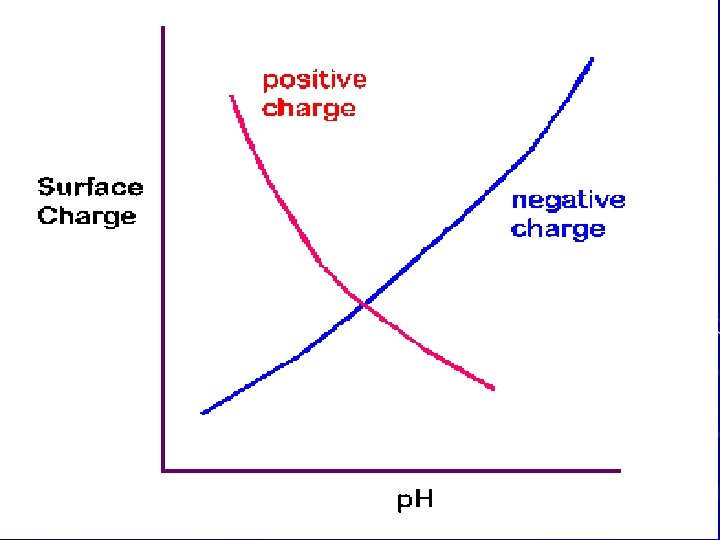
2. Variable or p. H-dependent charge )a) Dissociation of exposed OH groups )b) Occurs with humus, hydrous oxides, and broken edges of silicate clays )c) Increased p. H values = more negative charge d) Protonation of O and OH groups
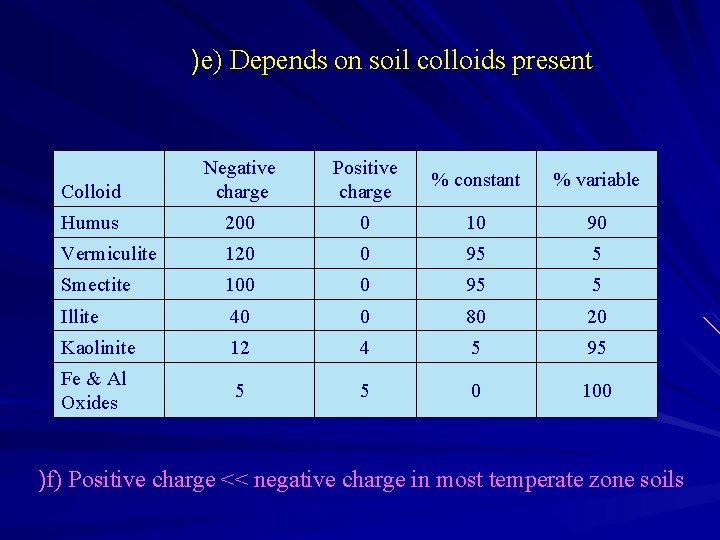
)e) Depends on soil colloids present Colloid Negative charge Positive charge % constant % variable Humus 200 0 10 90 Vermiculite 120 0 95 5 Smectite 100 0 95 5 Illite 40 0 80 20 Kaolinite 12 4 5 95 Fe & Al Oxides 5 5 0 100 )f) Positive charge << negative charge in most temperate zone soils

Cation Exchange 1. Process of cation replacement
Cation Exchange Capacity (CEC)

Units of charge per unit weight )a) centimoles of charge per kilogram of soil = cmolc/kg old units of milliequivalents per 100 grams of soil = (meq/100 g( )b) all cations are equal on a charge basis 1 cmolc/kg Na+ = 1 cmolc/kg Ca+2 = 1 cmolc/kg Al+3 )c) conversion of charge to weight 5 cmolc/kg of Ca+2 = ? g Ca+2 5)cmolc/kg soil) (molc/100 cmolc) (mol Ca+2/2 molc) (40 g Ca+2/mol Ca+2( 1 =g Ca+2/kg soil

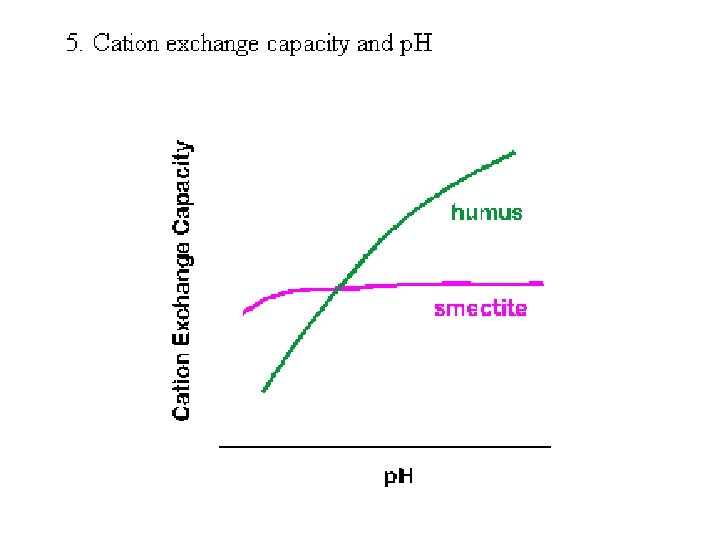
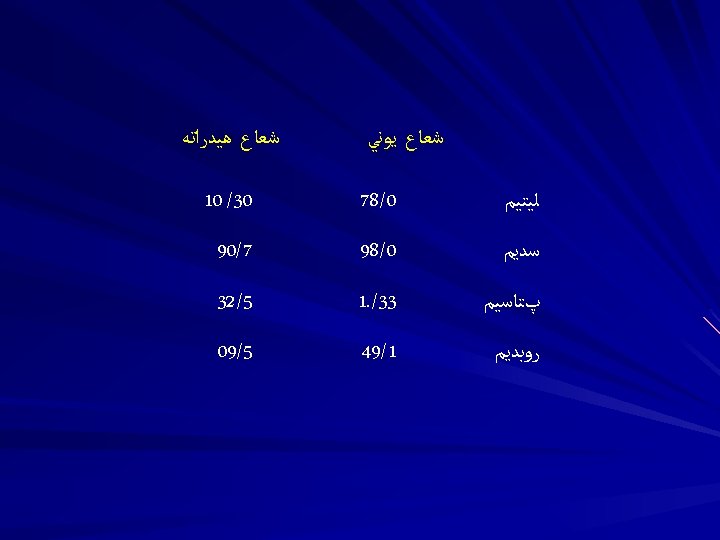
4. Cation exchange capacity of soils (a) related to components humus » 200 cmolc/kg illite » 25 cmolc/kg Fe and Al oxides » 4 cmolc/kg smectites » 100 cmolc/kg kaolinite » 10 cmolc/kg )b) estimate soil cation exchange capacity from composition: % 5 O. M. & 20 % smectite clay 200 x 0. 05 = 10 100 x 0. 20 = 20 total = 30 cmolc/kg % 2 O. M. & 30 % kaolinite clay 200 x 0. 02 = 4 10 x 0. 30 = 3 total = 7 cmolc/kg




10000* 0. 15= 1500 m 3 * 1500 kg/m 3= 2250000 kg 11. 3 meq for 100 g *20 mgr for meq= 226 g ca for 100 g = 0. 226 kg for 100 kg (0. 226 kg/100 kg) * 2250000 kg = 5085 kg ca /hec

Clay Genesis and Distribution . 1 Stages of weathering )a) alkali metals and alkaline earths dissolve )Na+, K+, Ca+2, Mg+2( )b) Si dissolves and leaches )c) continual reforming of new clay minerals Clays reflect weathering processes Young, weakly weathered soils = fine-grained mica, chlorite, vermiculite Intermediate weathering = vermiculite, smectite, kaolinite Strong weathering = kaolinite, hydrous oxides
- Slides: 30