Laws of Matter Daltons Atomic Theory Reviving the














- Slides: 17

Laws of Matter & Dalton’s Atomic Theory Reviving the Ancient Idea

Review of Lavoisier n n n Defined what an element was. Proposed program for chemistry. Discovered conservation of mass.

What’s Next? Quite clear that Aristotle was incorrect. n But, what then was the world made up of? n Elements? n But, what then were elements made up of? n

n Fortunately, advancements in instrumentation led to more precise balances and …

Other Laws Discovered Law of Definite Proportions n Law of Multiple Proportions n In addition to the Gas Laws and Conservation of Mass n

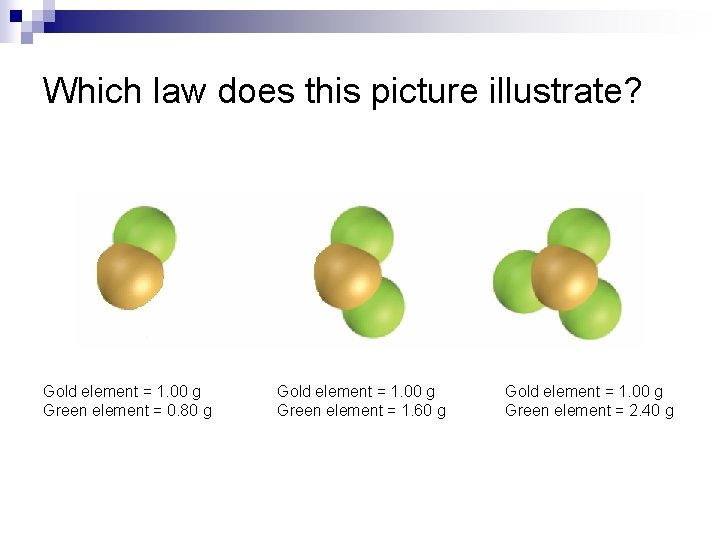
Which law does this picture illustrate? Gold element = 1. 00 g Green element = 0. 80 g Gold element = 1. 00 g Green element = 1. 60 g Gold element = 1. 00 g Green element = 2. 40 g

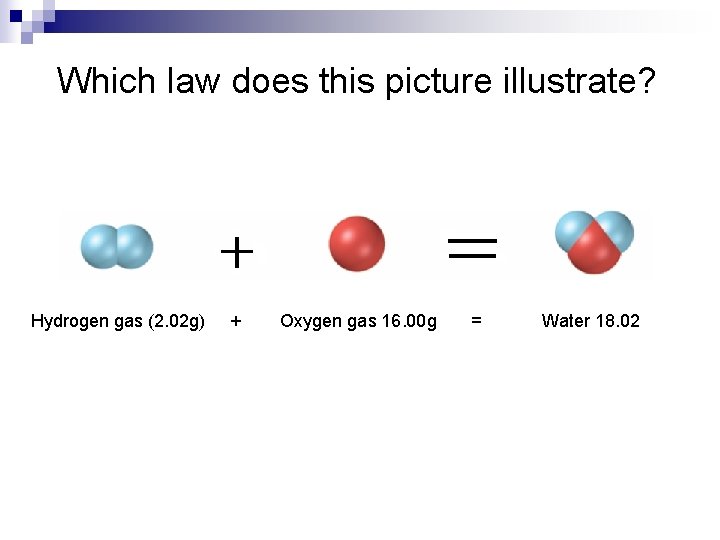
Which law does this picture illustrate? Hydrogen gas (2. 02 g) + Oxygen gas 16. 00 g = Water 18. 02

Which law does this picture illustrate? • Manufactured in a lab • Mined out of the ground 50% oxygen by mass 50% sulfur by mass

Which law does this picture illustrate? • Balloon at room temperature • Balloon heated up

The Atom becomes a theory n These developments led John Dalton to revive Democritus’ idea of the atom and create a theory of his own.

Dalton’s Atomic Theory 1. All matter is made of extremely small particles called atoms (in constant motion). 2. Atoms of a given element are identical in size, mass, and other properties; atoms of different elements differ in size, mass, and other properties. 3. Atoms cannot be subdivided, created, or destroyed. 4. Atoms of different elements combine in simple whole-number ratios to form chemical compounds. 5. In chemical reactions, atoms are combined, separated, or rearranged.

Conservation of mass Point 3: Atoms cannot be subdivided, created, or destroyed. n Point 4: In chemical reactions, atoms are combined, separated, or rearranged. n

Gas Laws n Point 1: All matter is made of extremely small particles called atoms (in constant motion).

Law of definite proportions n Point 2: Atoms of a given element are identical in size, mass, and other properties; atoms of different elements differ in size, mass, and other properties.

Law of multiple proportions n Point 4: Atoms of different elements combine in simple whole-number ratios to form chemical compounds.

Theory Accepted Dalton’s theory was accepted because it explained what was seen. n But, it did not give a way to determine molecule formulas. n ¨ How two atoms came together and in what ratios to form compounds. ¨ Dalton knew water was a 1 to 8 ratio by mass but thought the molecular formula was HO.

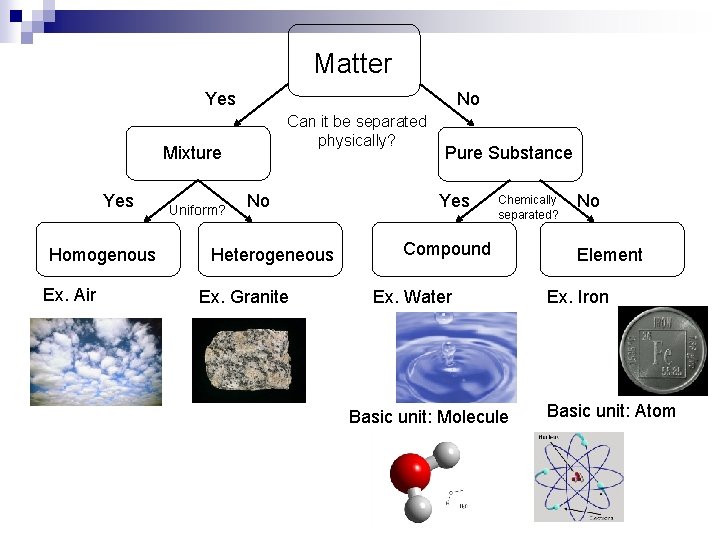
Matter Yes No Can it be separated physically? Mixture Yes Homogenous Ex. Air Uniform? No Heterogeneous Ex. Granite Pure Substance Yes Chemically separated? Compound Ex. Water Basic unit: Molecule No Element Ex. Iron Basic unit: Atom