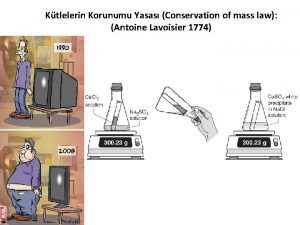
Law of Conservation of Mass Antoine Lavoisier 1775























- Slides: 25

Law of Conservation of Mass Antoine Lavoisier, ~ 1775 Law of Definite Proportions J. L. Proust, 1799

Law of Conservation of Mass In a chemical reaction, the Law of Conservation of Mass states that the Mass of the Reactants must equal the Mass of the Products. A C + B + E Products Reactants Mass A + Mass B + D = Mass ( C + D + E )

Law of Definite Proportions Any pure compound only contains the same elements in the same proportion by mass. H 2 O Define proportion: the ratio that relates one part to another part, or relates one part to the whole. Example: A large proportion of the people present in this classroom are students.

Acids n n n Vinegar is an Acid Chemical name is Acetic Acid Chemical formula: CH 3 CO 2 H

Bases n n n Baking Soda is a Base Chemical name is Sodium Bicarbonate Chemical formula: Na. HCO 3

Acids React with Bases Reactants = Acid + Base Vinegar + Baking Soda Mass of Reactants Product A Salt Water = Gas (sometimes) Sodium Acetate = Water (H 2 O) Carbon Dioxide = Mass of Products

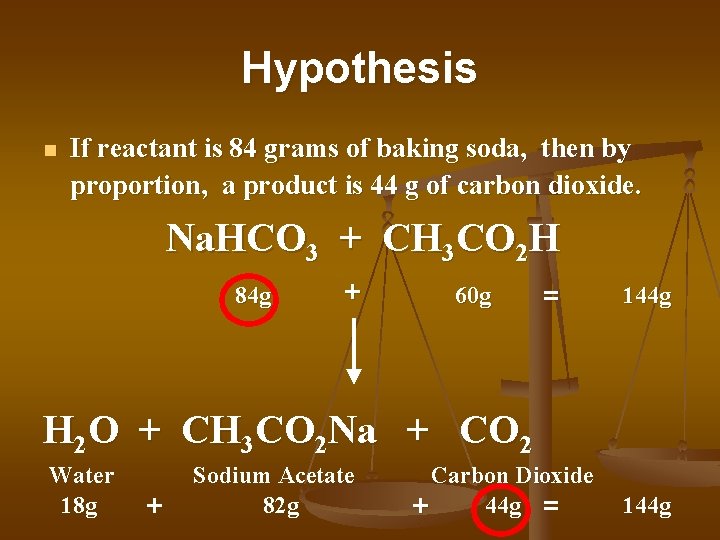
Hypothesis n If reactant is 84 grams of baking soda, then by proportion, a product is 44 g of carbon dioxide. Na. HCO 3 + CH 3 CO 2 H 84 g + 60 g = 144 g H 2 O + CH 3 CO 2 Na + CO 2 Water 18 g + Sodium Acetate 82 g Carbon Dioxide 44 g = + 144 g

Law of Definite Proportions Calculating Mass of Molecule A Atom Mass (g) Baking Soda Sodium Bicarbonate Na Sodium H Hydrogen C Carbon O Oxygen 23 g 1 g 12 g 16 g Na x 1 23 g Hx 1 1 g Cx 1 12 g OX 3 16(3) = 48 g Na. HCO 3 84 g

Law of Definite Proportions Calculating Mass of Molecule B Atom Mass (g) Vinegar Acetic Acid H Hydrogen C Carbon O Oxygen 1 g Hx 4 4 g 12 g Cx 2 24 g O 2 x 16 32 g CH 3 CO 2 H 60 g 16 g

Law of Definite Proportions Calculating Mass of Molecule B Atom Mass (g) Vinegar Acetic Acid H Hydrogen C Carbon O Oxygen 1 g Hx 4 1(4) = 4 g 12 g Cx 2 12(2) = 24 g OX 2 16(2) = 32 g 16 g CH 3 CO 2 H 60 g

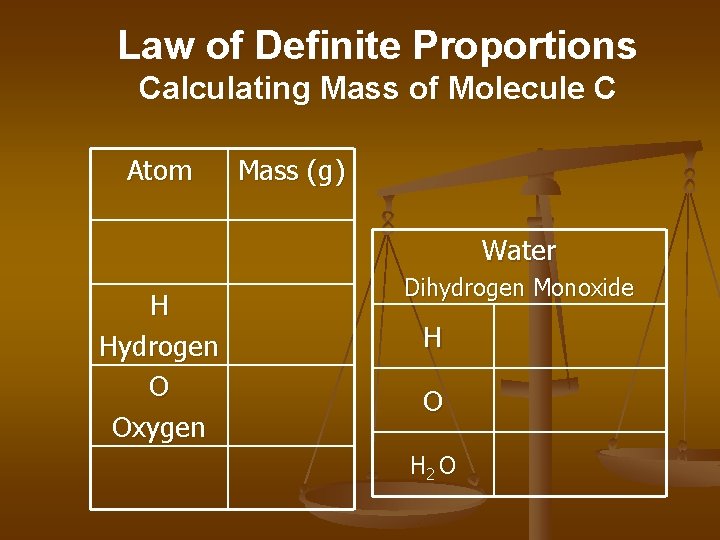
Law of Definite Proportions Calculating Mass of Molecule C Atom Mass (g) Water H Hydrogen O Oxygen Dihydrogen Monoxide H O H 2 O

Law of Definite Proportions Calculating Mass of Molecule C Atom Mass (g) Water H Hydrogen O Oxygen Dihydrogen Monoxide 1 g Hx 2 = 2 g 16 g OX 1 16 g H 2 O 18 g

Law of Definite Proportions Calculating Mass of Molecule D Atom Na Sodium H Hydrogen O Oxygen C Carbon Mass (g) A Salt Sodium Acetate 23 g Na x 1 23 g 1 g Hx 3 1(3) = 3 g OX 2 16(2) = 32 g Cx 2 12(2) = 24 g 16 g 12 g CH 3 CO 2 Na 82 g

Law of Definite Proportions Calculating Mass of Molecule E Atom C Carbon O Oxygen Mass (g) Gas Carbon Dioxide C O CO 2

Law of Definite Proportions Calculating Mass of Molecule E Atom C Carbon O Oxygen Mass (g) 12 g 16 g Gas Carbon Dioxide Cx 1 12 g OX 2 16(2) = 32 g CO 2 44 g

Mass Reactants = Mass Products Mass of 6 atoms Mass of 8 atoms Na. HCO 3 + CH 3 CO 2 H 84 g + 60 g Reactants 14 atoms = 144 g H 2 O + CH 3 CO 2 Na + CO 2 Water 18 g + Mass of 3 atoms Sodium Acetate 82 g Mass of 8 atoms Carbon Dioxide 44 g = + 144 g Mass of 3 atoms Products 14 atoms

Test Hypothesis To shorten the reaction time, we want to use only a small amount of baking soda. n n If reactant is 84 grams of baking soda, then we would get 44 grams of carbon dioxide. But if we use only 5 grams of baking soda, then by proportion, the product is 2. 6 grams of carbon dioxide. 5 g Sodium Bicarbonate ? g CO 2 5 g x 44 g = 2. 6 g CO 2 84 g

How can we measure the mass of gas produced? n Subtract the mass of the bottle + cap after the gas is released from the mass of the bottle + cap before the CO 2 is released. The value should less than 2. 6 g because about 10% of the CO 2 remains dissolved in the water solution.

How do we Measure the Volume of a Gas? n If we can measure the circumference of a sphere that traps the gas, such as a balloon, then we can calculate the volume of the gas.

Volume Calculation n n What is the volume of 2. 6 grams of CO 2? The density of CO 2 is 0. 001975 g/cm 3 V=m d V = 2. 6 g 0. 001975 g/cm 3 V = 1, 316 cm 3

Circumference Calculation n What should be the circumference of the balloon, if it holds 1, 316 cm 3 of CO 2? V = C 3 6π2 where C = Circumference V 6π2 = C 3 1, 316 cm 3 x 6 (3. 1415 x 3. 1415) = C 3 42. 7 cm = C

How do I Calculate the Mass of a Gas? n If we can measure the volume of the gas and we know its density, then we use D = m/V: Density (D) = Mass (m) Volume (V) or Volume (V) x Density (D) = Mass (m)

Comparing Our Measurements with Our Calculations n Calculated Circumference: 42. 7 cm n Measured Circumference: n Explain Any Difference

Conclusion n My hypothesis……. was supported by my data because the mass of all the products of this chemical reaction was equal to mass of all the reactants

Conclusion Continued n I know that this reaction obeys the Law of Conservation of Mass because I used the Law of Definite Proportions to predict the mass of carbon dioxide, and my results matched my prediction within the +/- margin of uncertainty caused by the carbon dioxide that remains dissolved in the water.
 Antoine lavoisier law
Antoine lavoisier law Robert boyle antoine lavoisier
Robert boyle antoine lavoisier Antoine lavoisier atom model
Antoine lavoisier atom model Periodic table notes
Periodic table notes Pse tablica
Pse tablica Law of conservation od mass
Law of conservation od mass Law of conservation of mass lab answer key
Law of conservation of mass lab answer key Law of conservation of mass
Law of conservation of mass Law of conservation of mass
Law of conservation of mass Law of conservation of mass
Law of conservation of mass Control volume approach
Control volume approach Law of conservation of mass
Law of conservation of mass Br3cl9 name
Br3cl9 name Law of conservation of mass examples
Law of conservation of mass examples What is law of conservation of mass
What is law of conservation of mass Law of conservation of mass
Law of conservation of mass Example of law of conservation of mass
Example of law of conservation of mass Law of conservation of mass examples
Law of conservation of mass examples The classical period started from 1750 to 1872
The classical period started from 1750 to 1872 Híres magyar matematikusok
Híres magyar matematikusok 1775-mr4
1775-mr4 Lexington and concord winner
Lexington and concord winner Hans gross
Hans gross Chapter 4 section 1 the revolution begins
Chapter 4 section 1 the revolution begins Peter zenger american revolution
Peter zenger american revolution What is the significance of july 4 1776 brainpop
What is the significance of july 4 1776 brainpop