Lasers and effects of magnetic field Physics 123












- Slides: 19

Lasers and effects of magnetic field Physics 123 9/30/2020 Lecture XXI 1

Light emission: classical case • Light bulb: current heats up atoms, they collide with each other and emit EM waves – light • Incoherent source of light – a continuous spectrum, isotropic in direction, no correlation in phase 9/30/2020 Lecture XXI 2

Light emission: quantum transitions Energy transition light emission fixed photon energy (frequency and wave length) Atoms are usually in the ground state (no emission) When excited (e. g. from temperature collisions) they go back to the ground state through spontaneous incoherent light emission (frequency=color). 9/30/2020 Lecture XXI 3

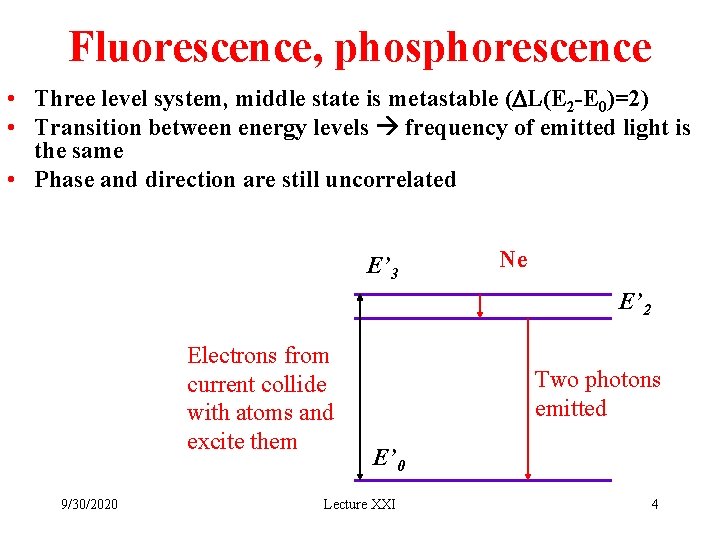
Fluorescence, phosphorescence • Three level system, middle state is metastable (DL(E 2 -E 0)=2) • Transition between energy levels frequency of emitted light is the same • Phase and direction are still uncorrelated E’ 3 Ne E’ 2 Electrons from current collide with atoms and excite them 9/30/2020 Two photons emitted E’ 0 Lecture XXI 4

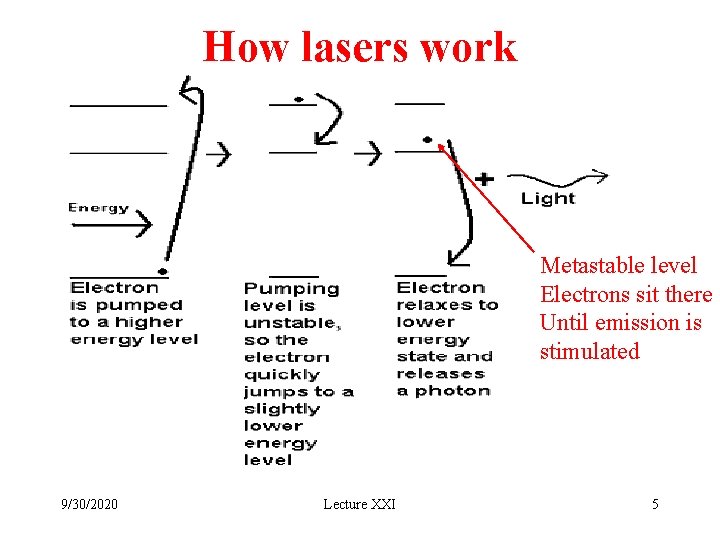
How lasers work Metastable level Electrons sit there Until emission is stimulated 9/30/2020 Lecture XXI 5

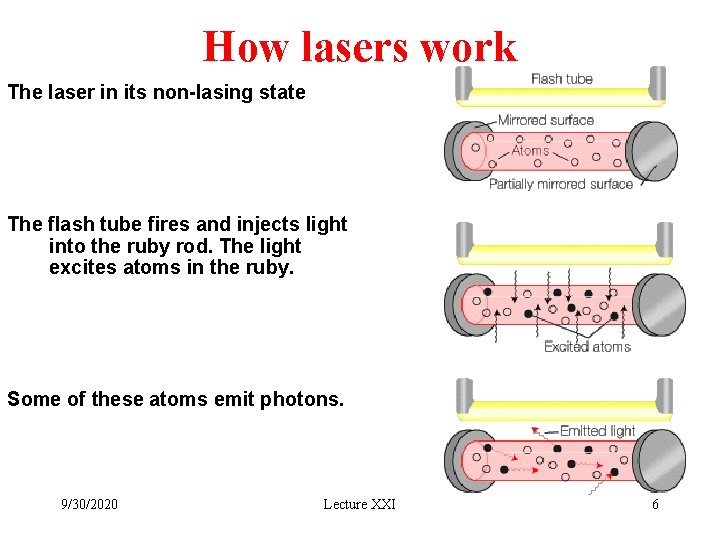
How lasers work The laser in its non-lasing state The flash tube fires and injects light into the ruby rod. The light excites atoms in the ruby. Some of these atoms emit photons. 9/30/2020 Lecture XXI 6

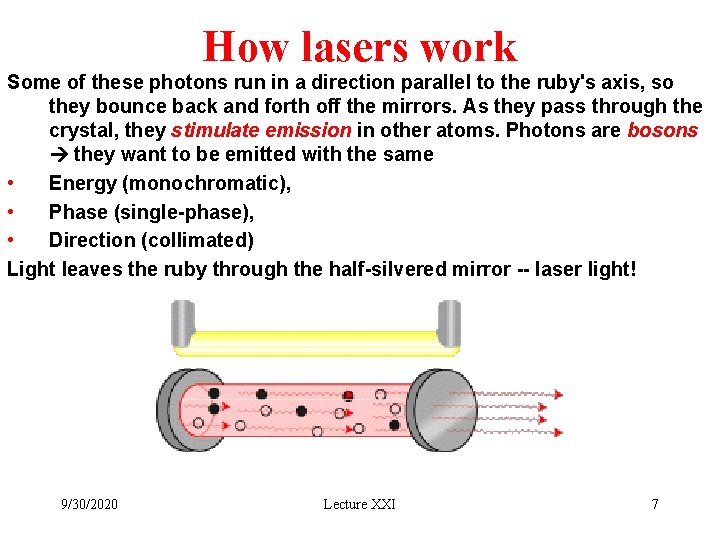
How lasers work Some of these photons run in a direction parallel to the ruby's axis, so they bounce back and forth off the mirrors. As they pass through the crystal, they stimulate emission in other atoms. Photons are bosons they want to be emitted with the same • Energy (monochromatic), • Phase (single-phase), • Direction (collimated) Light leaves the ruby through the half-silvered mirror -- laser light! 9/30/2020 Lecture XXI 7

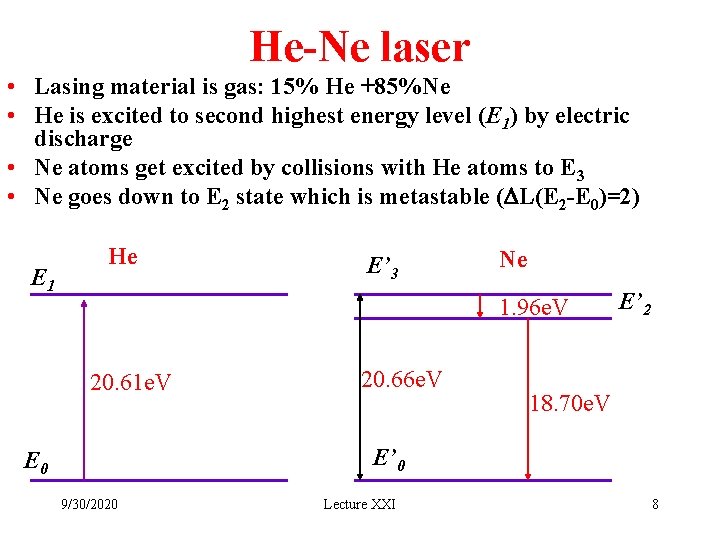
He-Ne laser • Lasing material is gas: 15% He +85%Ne • He is excited to second highest energy level (E 1) by electric discharge • Ne atoms get excited by collisions with He atoms to E 3 • Ne goes down to E 2 state which is metastable (DL(E 2 -E 0)=2) E 1 He E’ 3 Ne 1. 96 e. V 20. 61 e. V 20. 66 e. V E’ 2 18. 70 e. V E’ 0 E 0 9/30/2020 Lecture XXI 8

Lasers • Angular spread is very small, determined by diffraction on end mirror: – Q~l/D D Main advantage of lasers – energy is concentrated in one spot precision. Applications – laser surgery; - optical alignment; - drilling precision holes 9/30/2020 Lecture XXI 9

9/30/2020 Lecture XXI 10

Electron quantum state • Orbital quantum number is a vector length l • Orbital angular momentum: z • Its projection on z axis is Lz =mlh another q. n. – magnetic quantum number ml • ml can be only integer 9/30/2020 Lecture XXI 11

Atom in magnetic field • Magnetic dipole moment associated with orbital angular momentum • Since ml is quantized, z-projection of magnetic dipole moment is quantized as well • Bohr’s magneton • Potential energy of the magnetic dipole in magnetic field (in zdirection 9/30/2020 Lecture XXI 12

Zeeman effect • Potential energy of the magnetic dipole in magnetic field splits into several levels • Transitions between these levels – Zeeman effect • Zeeman effect is due to interaction of external magnetic field with orbital angular momentum of electrons in atoms • Electrons also have internal “angular momentum” - spin 9/30/2020 Lecture XXI 13

Spin • • All electrons have spin=1/2 It is a vector Its projection on z axis is another q. n. – spin ms ms can be only z • Similar to orbital angular momentum we expect • But experimentally measured value turned out to be different Gyromagnetic ratio, g-factor 9/30/2020 Lecture XXI 14

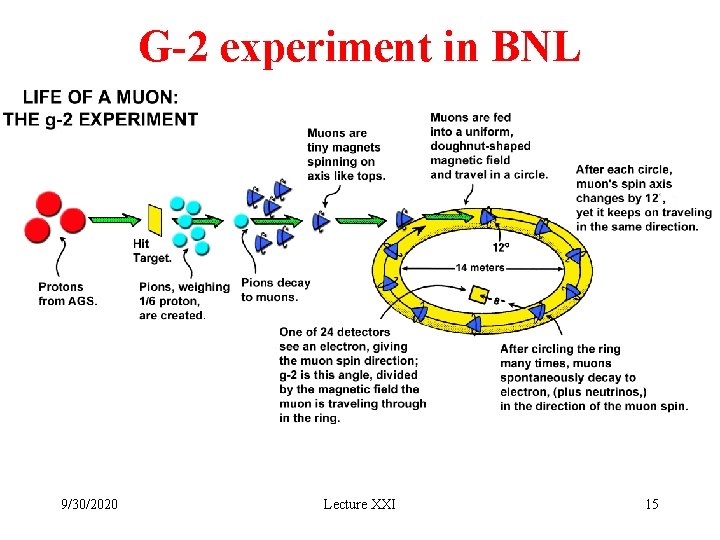
G-2 experiment in BNL 9/30/2020 Lecture XXI 15

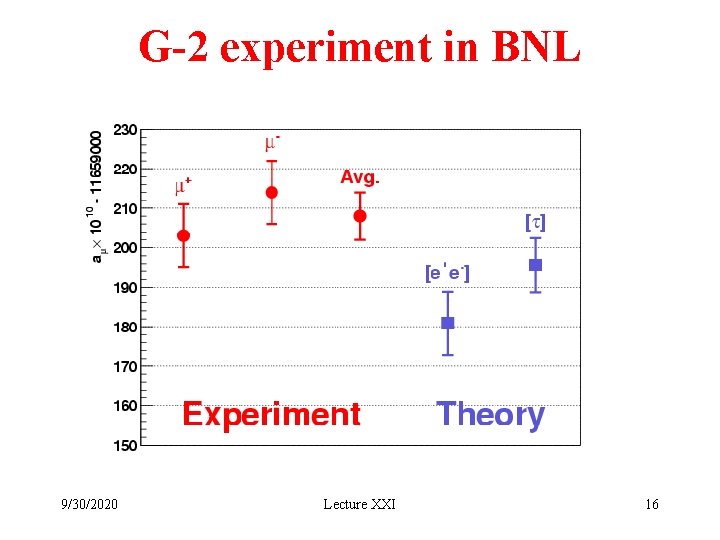
G-2 experiment in BNL 9/30/2020 Lecture XXI 16

Fine structure • In the absence of magnetic field some small splitting of levels in atoms was still observed – interaction of magnetic field created by orbital angular momentum (current loop=nucleus around electron) with electron’s spin • Constant of fine structure (spin-orbit interaction) 9/30/2020 Lecture XXI 17

Spins in magnetic field • Imagine free spin (electron or nucleus) placed inside magnetic field directed along z-axis • Energy levels split by • This system can absorb photons of the corresponding frequency 9/30/2020 Lecture XXI z E 0 DE 18

Nuclear magnetic resonance • Transition between the two states – photons of only a certain energy (frequency) will be absorbed – resonance absorption (NMR) – For H f=42. 58 MHz for B=1. 0 T – This frequency varies slightly for bound (trapped in molecules) H atoms – By mapping f, we can map chemical composition of human body – Precision of NMR – 0. 5 – 1. 0 mm 9/30/2020 Lecture XXI 19