Laserinduced Coulomb explosion imaging of molecular dynamics 1

Laser-induced Coulomb explosion imaging of molecular dynamics 1. Introduction 2. Structure determination with CEI 3. Dynamics studies with CEI
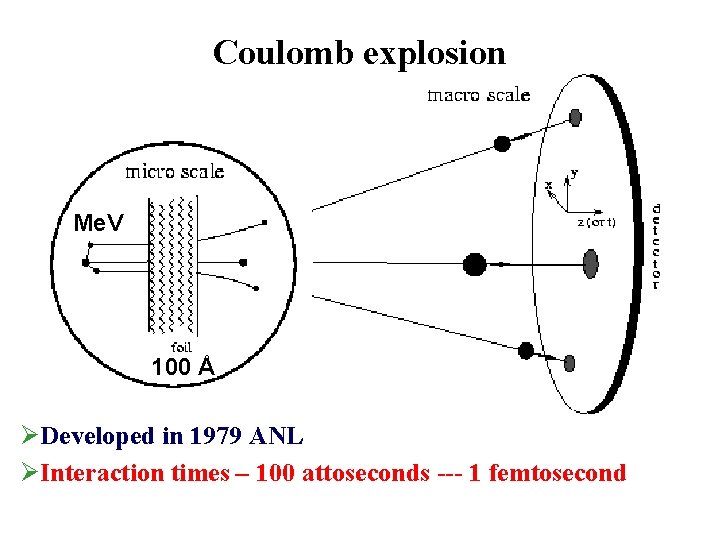
Coulomb explosion Me. V 100 Å ØDeveloped in 1979 ANL ØInteraction times – 100 attoseconds --- 1 femtosecond
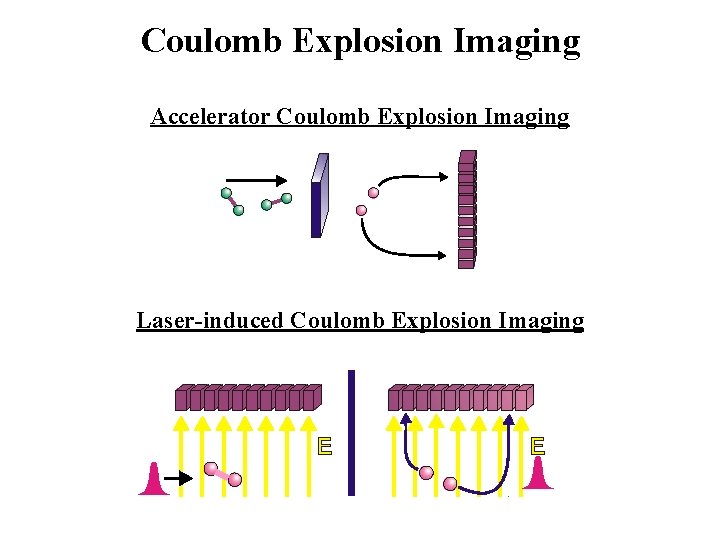
Coulomb Explosion Imaging Accelerator Coulomb Explosion Imaging Me. V beam Exploding molecule ANL 1979 Ultrathin foil Detector Laser-induced Coulomb Explosion Imaging Detector
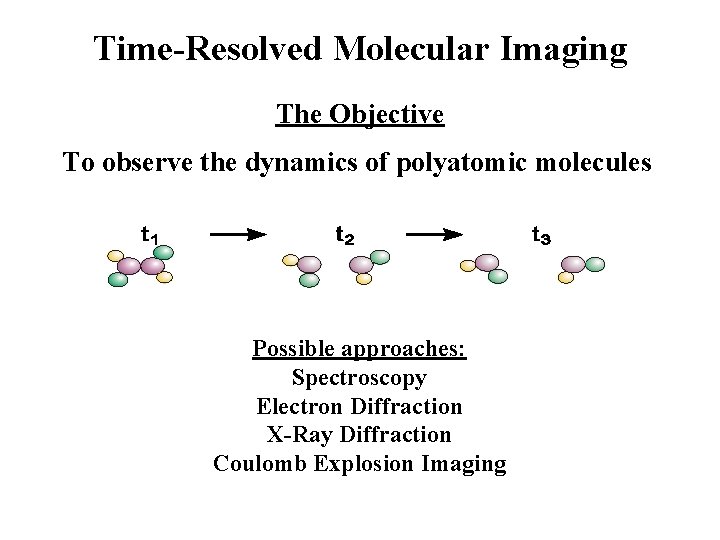
Time-Resolved Molecular Imaging The Objective To observe the dynamics of polyatomic molecules Possible approaches: Spectroscopy Electron Diffraction X-Ray Diffraction Coulomb Explosion Imaging
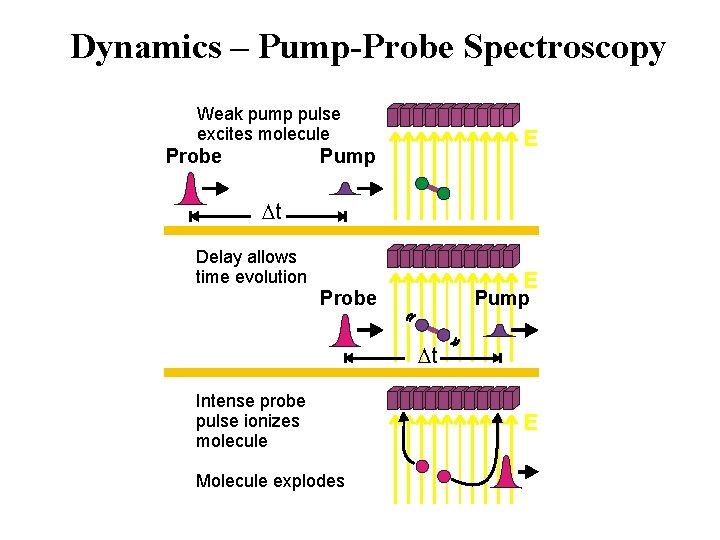
Dynamics – Pump-Probe Spectroscopy Weak pump pulse excites molecule Probe E Pump t Delay allows time evolution E Probe Pump t Intense probe pulse ionizes molecule Molecule explodes E
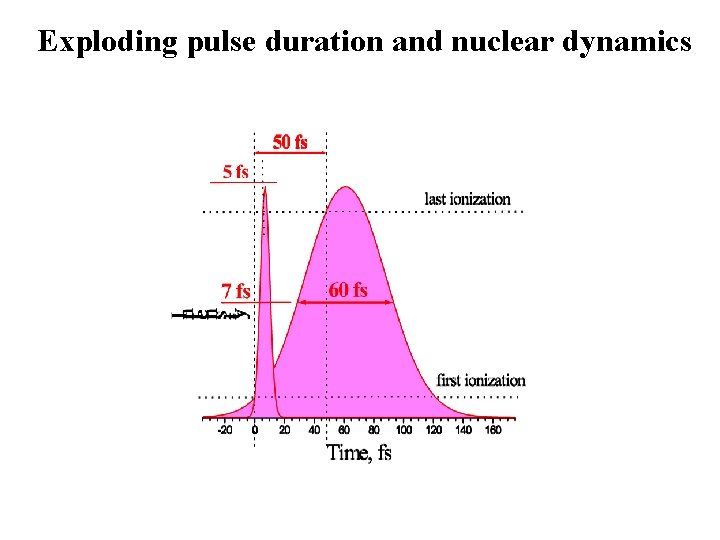
Exploding pulse duration and nuclear dynamics
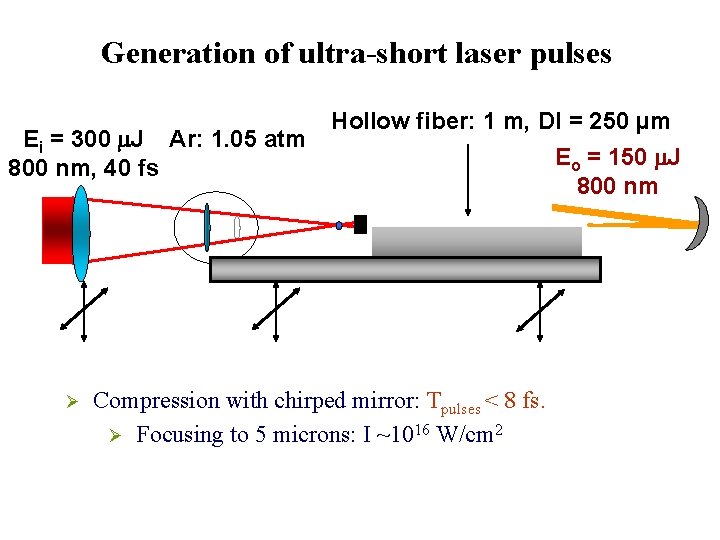
Generation of ultra-short laser pulses Ei = 300 m. J Ar: 1. 05 atm 800 nm, 40 fs Ø Hollow fiber: 1 m, DI = 250 µm Compression with chirped mirror: Tpulses < 8 fs. Ø Focusing to 5 microns: I ~1016 W/cm 2 Eo = 150 m. J 800 nm
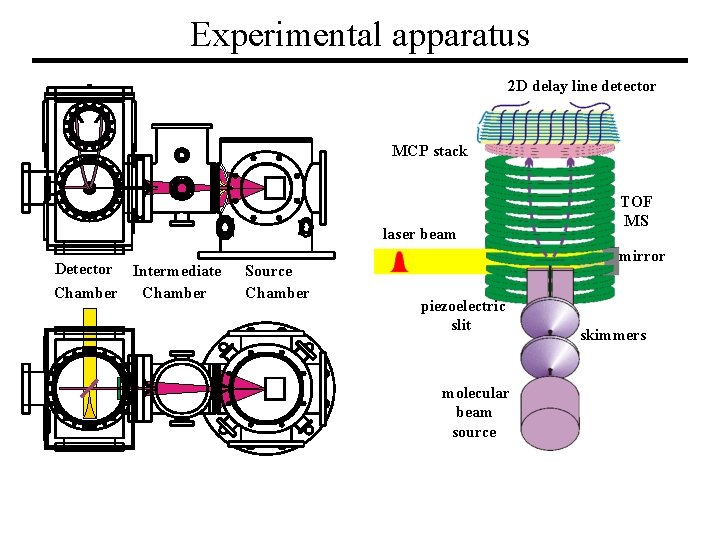
Experimental apparatus 2 D delay line detector MCP stack laser beam Detector Intermediate Chamber Source Chamber TOF MS mirror piezoelectric slit molecular beam source skimmers

Simplest model --Coulomb explosion of D 2
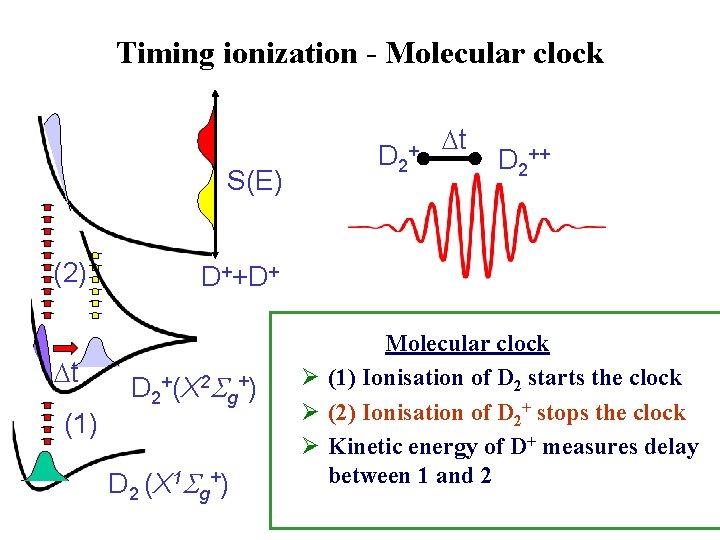
Timing ionization - Molecular clock S(E) (2) t (1) D 2 + t D 2++ D++D+ D 2+(X 2 Sg+) D 2 (X 1 Sg+) Molecular clock Ø (1) Ionisation of D 2 starts the clock Ø (2) Ionisation of D 2+ stops the clock Ø Kinetic energy of D+ measures delay between 1 and 2
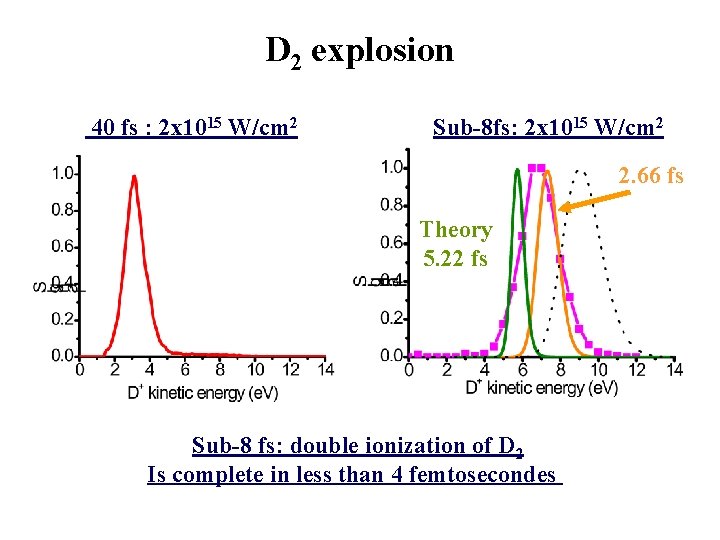
D 2 explosion 40 fs : 2 x 1015 W/cm 2 Sub-8 fs: 2 x 1015 W/cm 2 2. 66 fs Theory 5. 22 fs Sub-8 fs: double ionization of D 2 Is complete in less than 4 femtosecondes
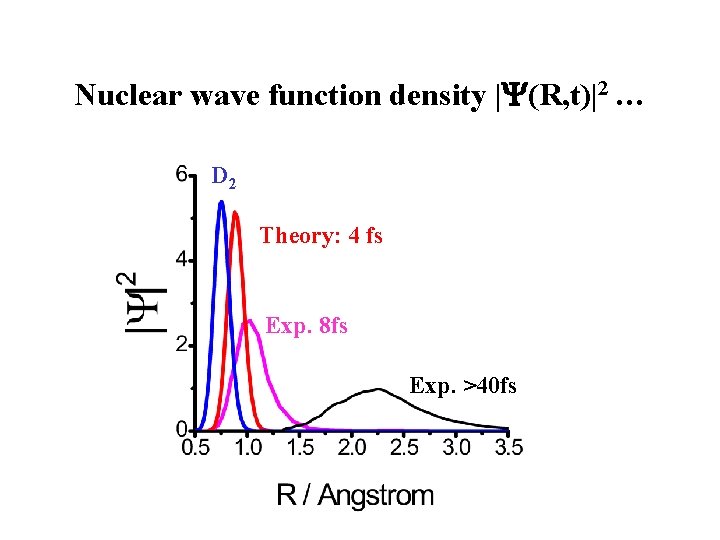
Nuclear wave function density | (R, t)|2 … D 2 Theory: 4 fs Exp. 8 fs Exp. >40 fs
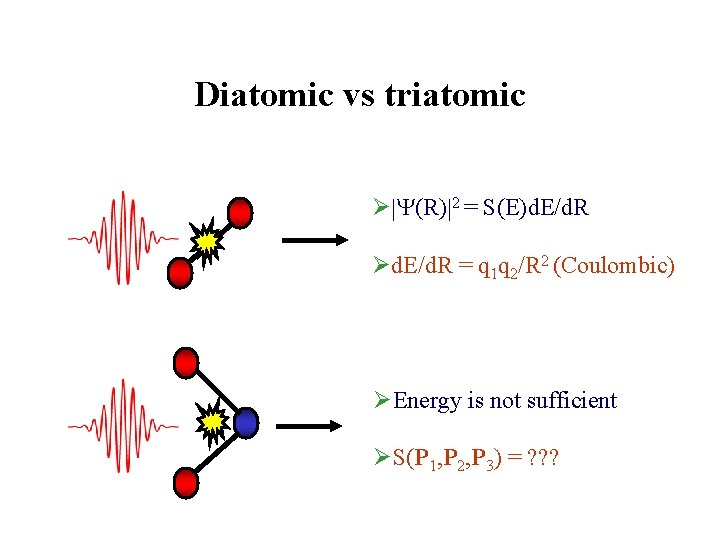
Diatomic vs triatomic Ø (R) 2 = S(E)d. E/d. R Ød. E/d. R = q 1 q 2/R 2 (Coulombic) ØEnergy is not sufficient ØS(P 1, P 2, P 3) = ? ? ?
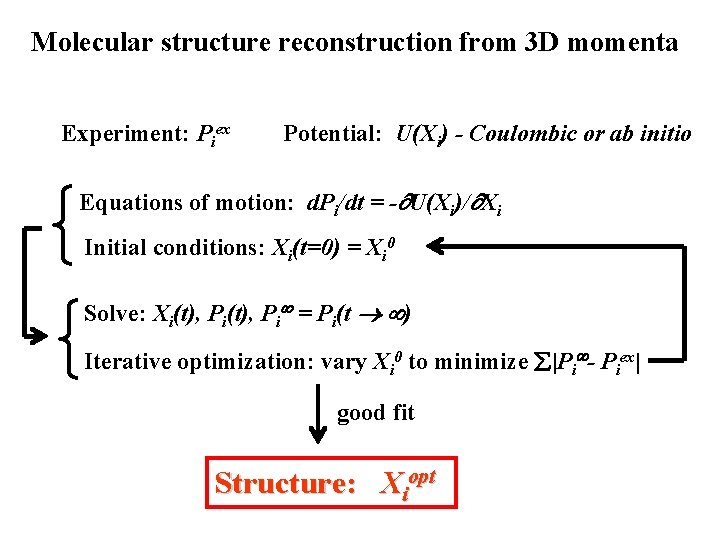
Molecular structure reconstruction from 3 D momenta Experiment: Piex Potential: U(Xi) - Coulombic or ab initio Equations of motion: d. Pi/dt = - U(Xi)/ Xi Initial conditions: Xi(t=0) = Xi 0 Solve: Xi(t), Pi = Pi(t ) Iterative optimization: vary Xi 0 to minimize |Pi - Piex| good fit Structure: Xiopt
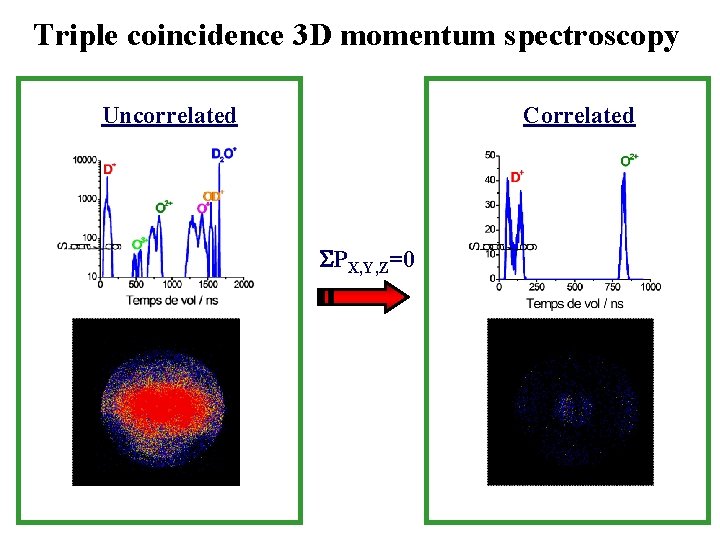
Triple coincidence 3 D momentum spectroscopy Correlated Uncorrelated PX, Y, Z=0
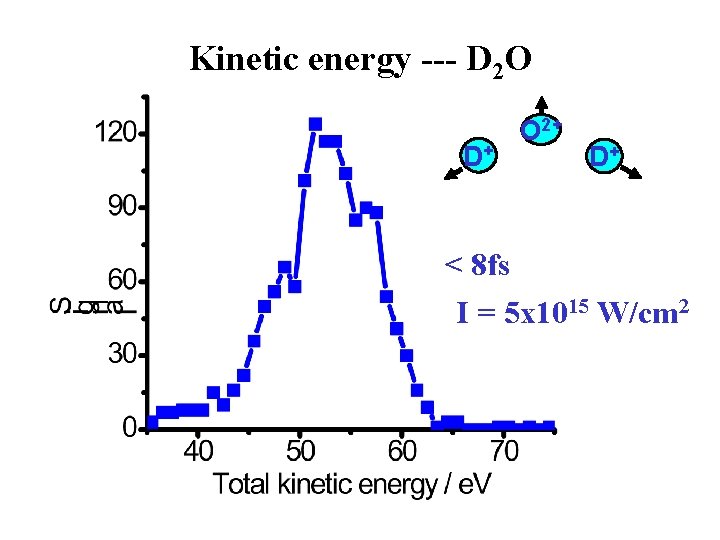
Kinetic energy --- D 2 O D+ O 2+ D+ < 8 fs I = 5 x 1015 W/cm 2
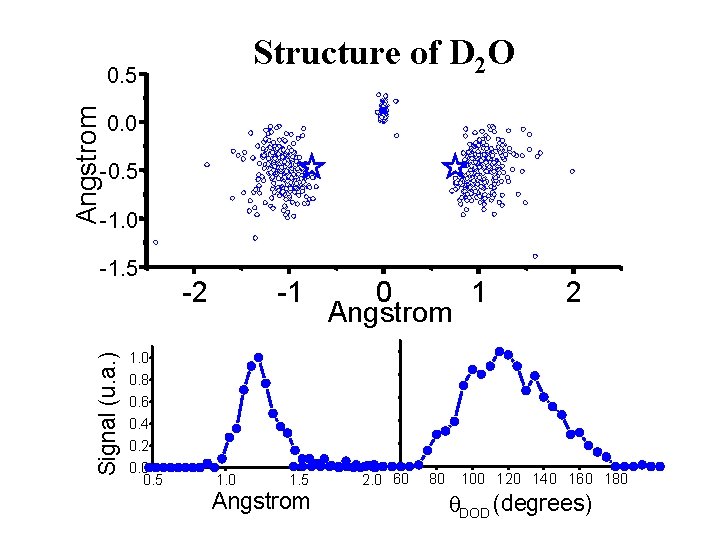
Structure of D 2 O Angstrom 0. 5 0. 0 -0. 5 -1. 0 Signal (u. a. ) -1. 5 1. 0 0. 8 0. 6 0. 4 0. 2 0. 0 0. 5 -2 -1 1. 0 1. 5 Angstrom 0 1 Angstrom 2. 0 60 2 80 100 120 140 160 180 q. DOD (degrees)
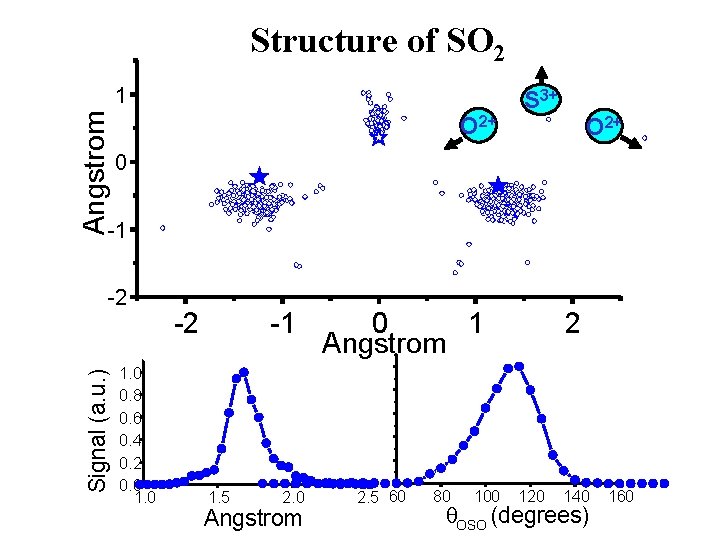
Structure of SO 2 Angstrom 1 O 2+ S 3+ O 2+ 0 -1 Signal (a. u. ) -2 1. 0 0. 8 0. 6 0. 4 0. 2 0. 0 1. 0 -2 -1 1. 5 2. 0 Angstrom 0 1 Angstrom 2. 5 60 80 100 2 120 140 q. OSO (degrees) 160
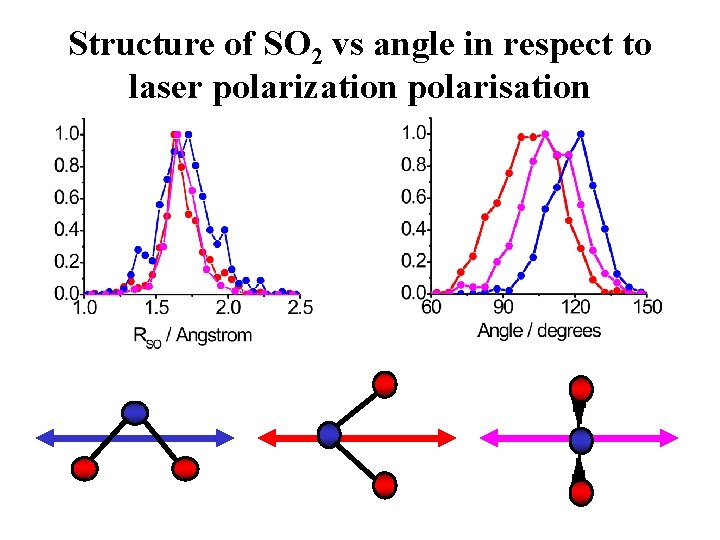
Structure of SO 2 vs angle in respect to laser polarization polarisation

Image distortion Ø Dynamics before the explosion D 2 O – numeric simulation Ø Dependence of ionization rate on nuclear coordinates M 2+ M+ M 0 D 2 –numeric simulation M+ M 0 Ø Angle in respect to the laser field ? ? ? SO 2 Ø Spatial resolution : < 0. 3 Å

Pump-probe spectroscopy --observing motion of protons
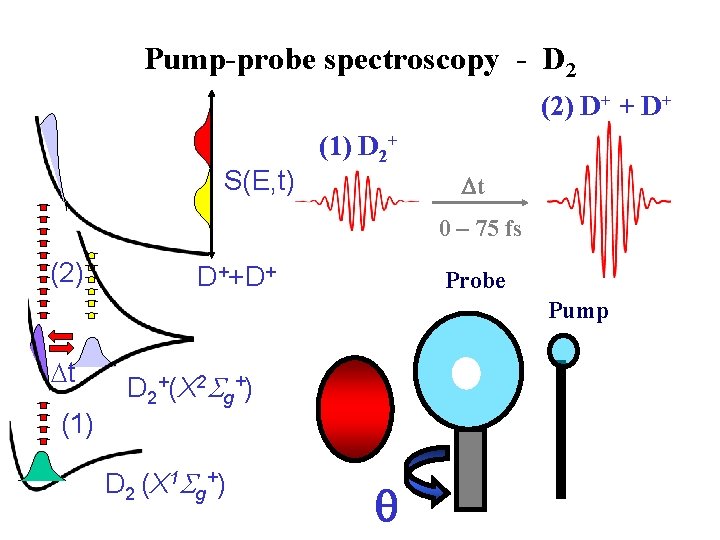
Pump-probe spectroscopy - D 2 (2) D+ + D+ S(E, t) (1) D 2+ t 0 – 75 fs (2) D++D+ Probe Pump t (1) D 2+(X 2 Sg+) D 2 (X 1 Sg+)
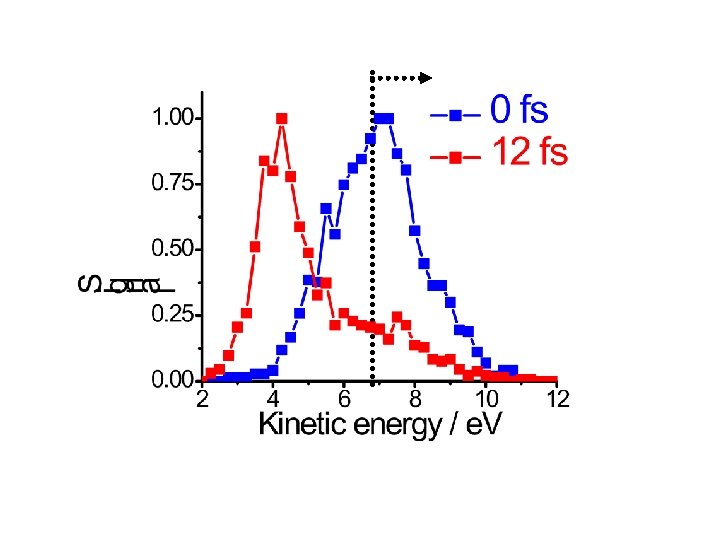
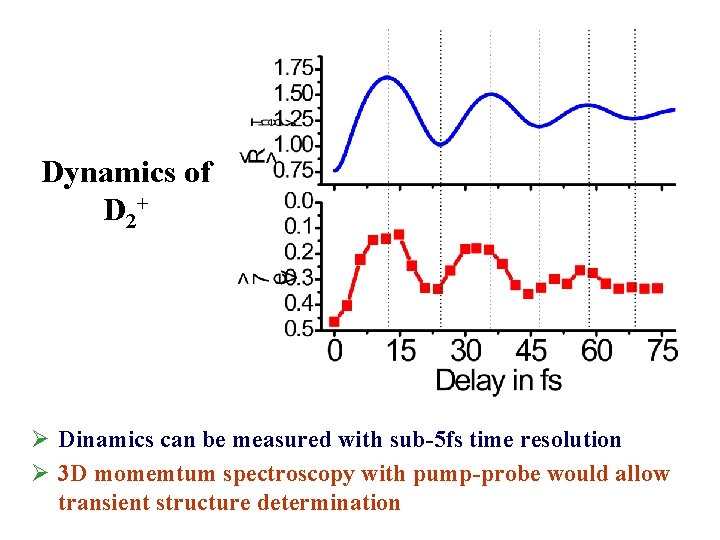
Dynamics of D 2+ Ø Dinamics can be measured with sub-5 fs time resolution Ø 3 D momemtum spectroscopy with pump-probe would allow transient structure determination
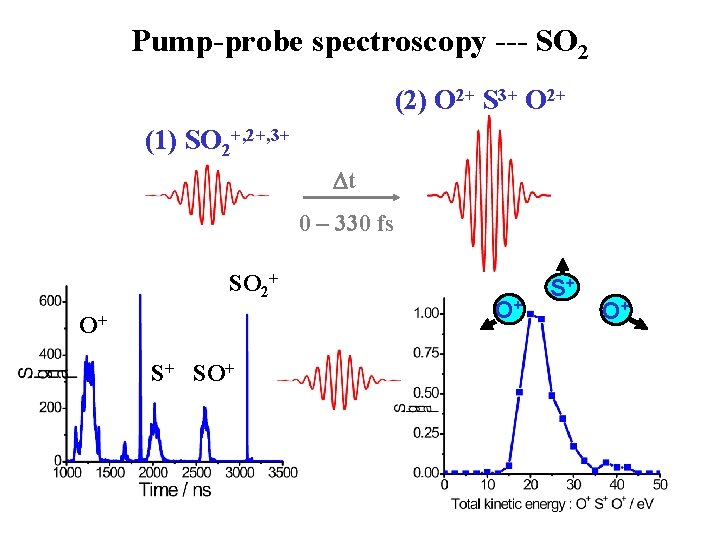
Pump-probe spectroscopy --- SO 2 (2) O 2+ S 3+ O 2+ (1) SO 2+, 3+ t 0 – 330 fs SO 2+ O+ S+ SO+ O+ S+ O+
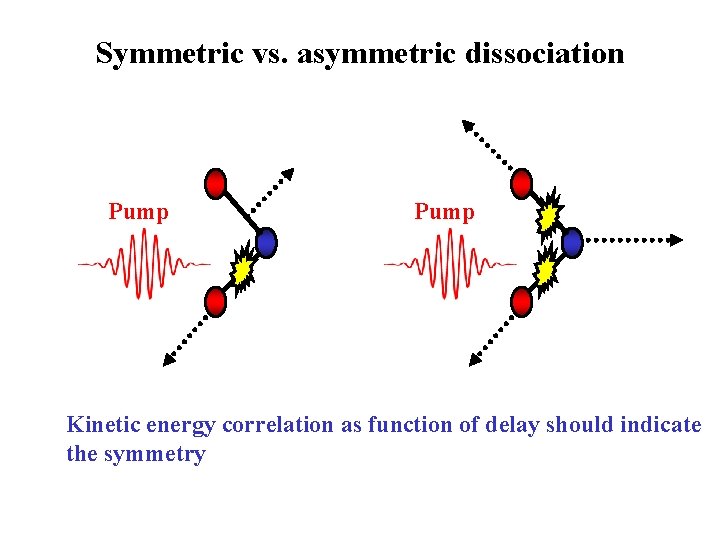
Symmetric vs. asymmetric dissociation Pump Kinetic energy correlation as function of delay should indicate the symmetry
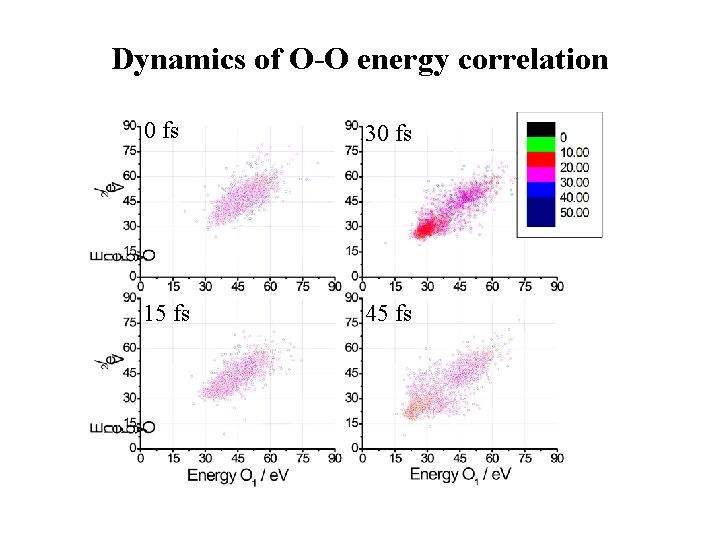
Dynamics of O-O energy correlation 0 fs 30 fs 15 fs 45 fs
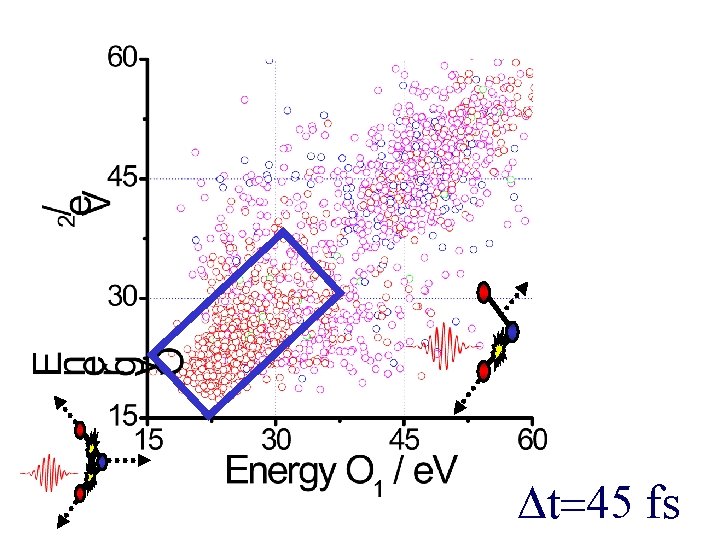
t=45 fs
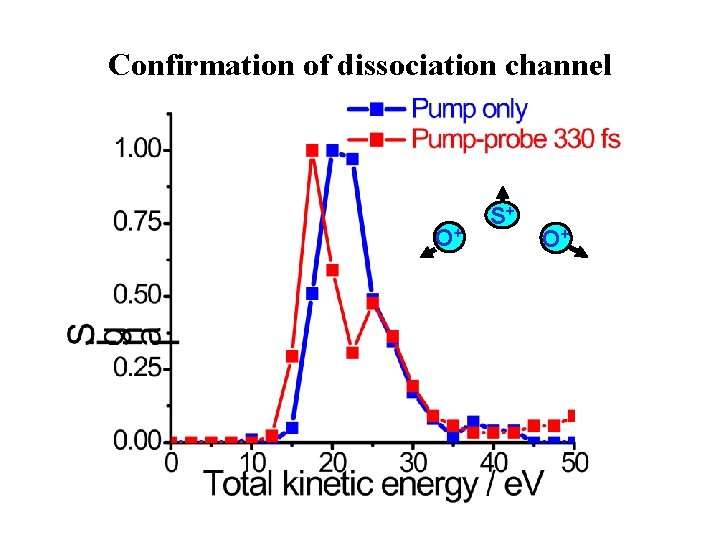
Confirmation of dissociation channel O+ S+ O+
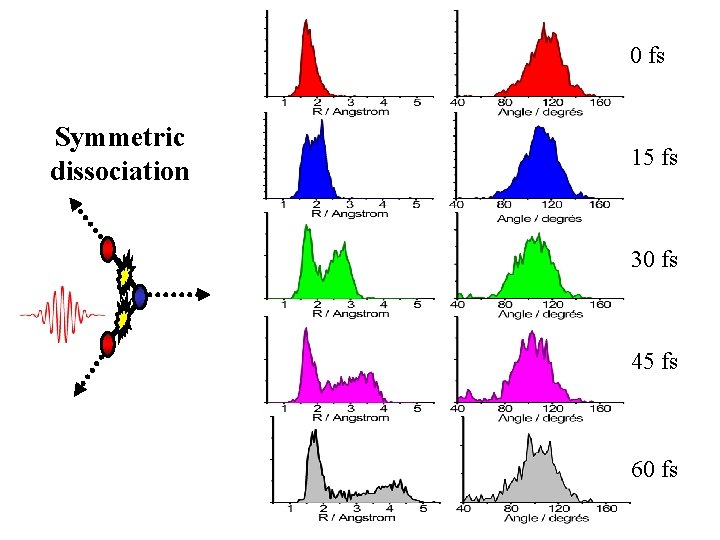
0 fs Symmetric dissociation 15 fs 30 fs 45 fs 60 fs
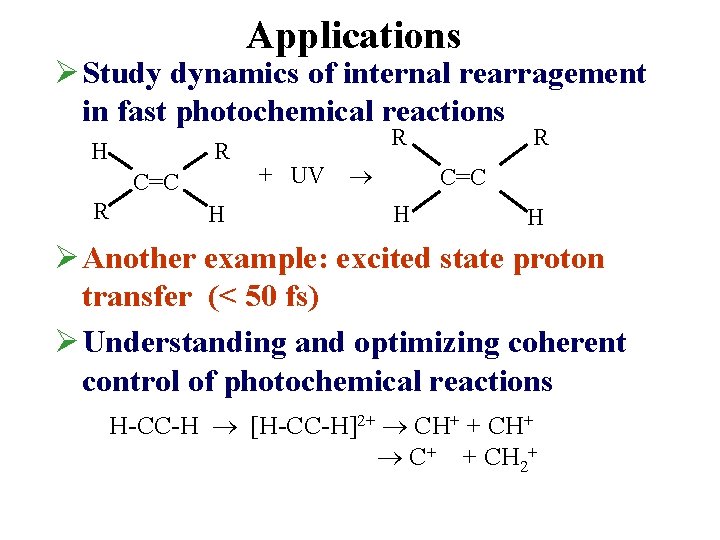
Applications Ø Study dynamics of internal rearragement in fast photochemical reactions H R C=C R H R R + UV C=C H H Ø Another example: excited state proton transfer (< 50 fs) Ø Understanding and optimizing coherent control of photochemical reactions H-CC-H [H-CC-H]2+ CH+ + CH+ C+ + CH 2+

Ø F. Légaré - Université de Sherbrooke Ø K. Lee, P. Dooley - Mc. Master University Ø D. Villeneuve, P. Corkum – NRC Ottawa

- Slides: 33