Lactococcosis in reared rainbow trout in Bulgaria case

Lactococcosis in reared rainbow trout in Bulgaria – case report and diagnostics approaches P. Orozova 1, E. Mileva 1, Sv. Kirova 1, M. Zaharieva 2, H. Naidenski 2 1 National Diagnostic Science and Research Veterinary Medical Institute, Department of Aquaculture, Fish and Bee Diseases, 15 P. Slaveikov Blvd. , Sofia, Bulgaria 2 BAS Institute of Microbiology Deapartment of Infectious Microbiology 26 G. Bonchev Str. , Sofia 1113, Bulgaria

INTRODUCTION Lactococcus garvieae = Enterococcus seriolicida is a causative agent of lactococcosis, a disease leading to significant economic losses in fish farming around the world when the water temperature exceeds 15 ° C. Lactococcus garvieae is considered as one of the most significant trout pathogens especially in countries such as Turkey, Spain and Italy. Pathogenic potential of L. garvieae for human underlines its importance as a zoonotic agent.

Lactococcosis is a disease with high morbidity and mortality that persists for a long time in the affected farms. In the trout cultivation the losses can reach 5080% of the total production. Once appeared, lactococcosis usually occurs again, especially during the summer months. It has been observed in several experimental tests that incubation period of the disease is very short and the microorganism acts with high virulence: symptoms are visible 2– 3 days post-inoculation (Itami et al. , 1996).

TRANSMISSION Lactococcosis could be transmitted from diseased to healthy fish when placed in the same tank.

CASE DESCRIPTION During the summer of 2018 (June-July-August), an outbreak of lactococcosis took place in a rainbow trout farm in Bulgaria. Production of the farm was 100 tones. Clinically lethargie, erratic swimming, darkness in colour were observed. Uni- or bilateral exophthalmia was frequent, with intraocular hemorrhaging and clouding of the eye. Abdominal distension, and hemorrhage around the opercula and anus were observed. Treatment with erythromicin was not effective. When antibiotic therapy finished on 04. 09. 2018 the mortality started again – 40 -90 kg per a day. The mortality rates in affected fish ranged from 30% to 40%.

AIM • Isolation and characterization of Lactococcus garvieae strains from diseased rainbow trouts with signs of lactococcosis via different methods. • Confirmation of the isolates as L. garvieae by molecular methods • Determination of antibiotic susceptibility of disease determinant bacterial strains.

MATERIALS AND METHODS Conventional bacteriological tests Microbiological samples were obtained from fish brains and streaked onto 5% sheep blood agar plates (BAB; Hi. Media). The plates were incubated at 28⁰ C for 18– 20 h and the predominant alpha haemolytic colonies were selected for further phenotypic analyses. The phenotypic characterization was based on presence of α -haemolysis, Gram staining, colony differentiation and standard biochemical tests. For comparison purposes, tree Type Culture Collection (TCC) reference isolates were used: ATCC 43921 type isolate of L. garvieae originating from a clinical case of bovine mastitis (Collins et al. 1983; Schleifer et al. 1985, VP 8066).

MATERIALS AND METHODS primer Sequence (5`-3`) CAU 12 F ACTCGTGCTATCCTT CAU 15 R TGGGTACTCCCAACTTCC ITS Lg 30 F ACTTTATTCAGTTTTGAGGGGTCT ITS Lg 319 R TTTAAAAGAATTCGCAGCTTTACA Target gene Amplicon Ref. size 16 S r. RNA 415 bp Jung, Chang and Kim, 2010 16 S– 23 S r. RNA ITS region 290 bp Dang et al. , 2012

MATERIALS AND METHODS • Antibiotic susceptibility testing by: The Kirby-Bauer disc diffusion method – tested antibiotics: Clyndamicin(2µg), Tetracycline(30µg), Ceftiofur(30µg), Erythromycin(15µg), Nalidixic acid(30µg), Florfenicol(30µg), Ampicillin(10µg), Oxitetracicline (30µg), Oxolinic acid (2µg)
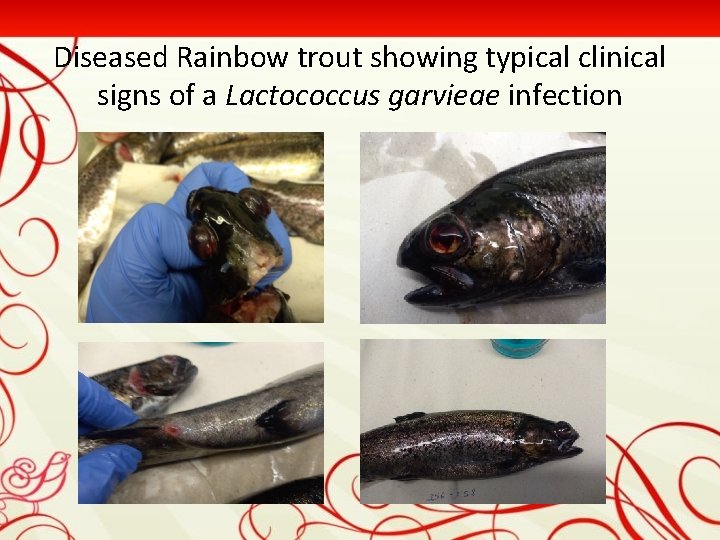
Diseased Rainbow trout showing typical clinical signs of a Lactococcus garvieae infection

DIAGNOSIS Ten fish showing clinical signs of the disease were sampled from the farm and stored at 4°C prior to transport to a laboratory for bacteriological analysis. Swabs of brain and kidney tissue from each fish were streaked onto 5% sheep blood agar, and incubated at 28°C for 48 h. Strains were stored at -80°C in brain/heart infusion broth with 15% glycerol, until use. Brains (fish 1 + fish 2; fish 3 + fish 4 etc. ) of the fish were collected in Eppendorff tubes and stored at -80°C. For the purpose of molecular analysis, the isolates and brains were thawed, streaked onto 5% sheep blood agar, and incubated at 28°C for 24 h.

Bacteriological Findings After incubation for 48 h at 28°C, pure cultures of whitish colonies were observed. Oxidase negative, Gram-positive cocci were inoculated onto MRS agar plates (Hi. Media), and in appropriate media for other biochemical tests. Catalase (Sigma-Aldrich) and 6. 5% salt tolerance was tested also. All isolates were nonmotile, oxidase and catalase negative, and Gram-positive cocci short chain. Based on phenotypic characteristics, the bacterium was identified as L. garvieae.
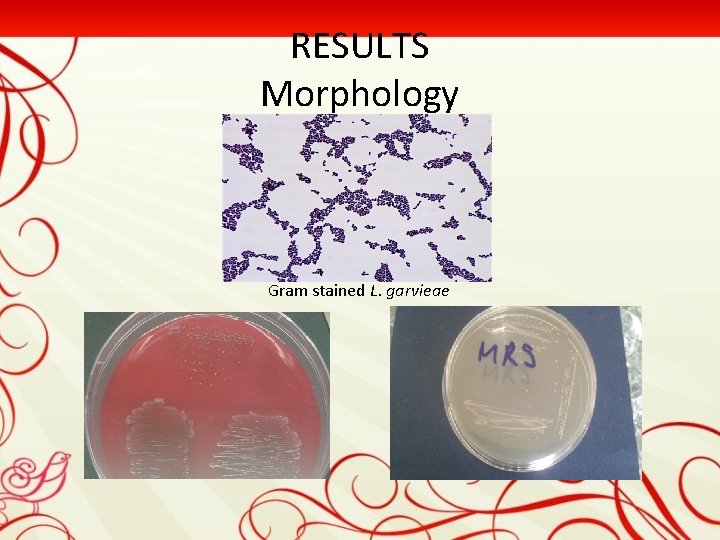
RESULTS Morphology Gram stained L. garvieae

RESULTS For the rapid, primary diagnosis hyperimmune anti-L. garvieae rabbit serum was used.

Biochemical cultural and physiological characteristics of L. garvieae Cell morphology: Gram: Motility: Growth on: 4 °C 20 °C 37 °C p. H 9. 6 6. 5% Na. Cl Hemolysis: Catalase Oxidase VP H 2 S production Ovoid cocci + − + + + α − − + −

Specificity of the L. garvieae PCR assay according to the method of Dang et al. , 2012. A conventional PCR with primers designed to detect 16 Sr. RNA and 23 Sr. RNA spacer region was tested for specificity against comprehensive range of closely related species and genera and compared to previouosly published primers (Dang et al. , 2012). The master mix consists of 1 m. M of each primer ITSLg 30 F (5’TTATTCAGTTTTGAGGGGTCT 3’) and ITSLg 319 R (5’AAAAGAATTCGCAGCTTTACA 3’) , 2. 5 µl reaction buffer, 0. 2 m. M d. NTPs, 1. 5 m. M Mg. Cl 2, 2. 5 U Taq polymerase, and 50 ng DNA in a 25µl reaction volume. Thermocycling is carried out using an initial denaturation step at 94⁰ C for 5 min followed by 30 cycles denaturation at 94 ⁰ C for 30 sec, annealing at 58 ⁰ C for 30 sec, extension at 72 ⁰ C for 40 sec, and a final step using an extension at 72 ⁰ C for 7 min. Amplified product is 290 bp. The limit of detection is 2. 6 pg DNA.

Specificity of the L. garvieae PCR ay according to the method of Jung, Chang and Kim, 2010 Primers CAU 12 F (5′ ACTCGTGCTATCCTT 3′) and CAU 15 R (5′ TGGGTACTCCCAACTTCC 3′) were used in conventional PCR which consists of 20µl master mix containing 10 pmol of each primer, Tris-HCl (p. H 9), 40 mmol/l KCl, 250µmol/l d. NTPs Taq polymerase, 1. 5 mmol/l Mg. Cl and 1 (10 ng) template DNA in a 20 µl reaction volume. Thermocycling is carried out using an initial denaturation step at 95⁰C for 5 min followed by 35 cycles of denaturation at 95 ⁰ C for 1 min, annealing at 58 ⁰ C for 1 min , extension at 72 ⁰ C for 1 min and a final step using an extension at 72 ⁰ C for 7 min. The amplified product is 415 bp. The sensitivity of the conventional PCR is 1 ng DNA.
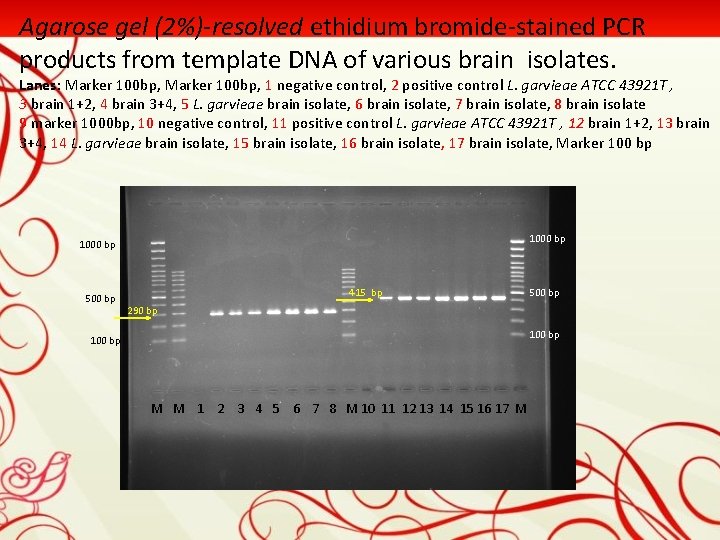
Agarose gel (2%)-resolved ethidium bromide-stained PCR products from template DNA of various brain isolates. Lanes: Marker 100 bp, 1 negative control, 2 positive control L. garvieae ATCC 43921 T , 3 brain 1+2, 4 brain 3+4, 5 L. garvieae brain isolate, 6 brain isolate, 7 brain isolate, 8 brain isolate 9 marker 1000 bp, 10 negative control, 11 positive control L. garvieae ATCC 43921 T , 12 brain 1+2, 13 brain 3+4, 14 L. garvieae brain isolate, 15 brain isolate, 16 brain isolate, 17 brain isolate, Marker 100 bp 1000 bp 500 bp 415 bp 500 bp 290 bp 100 bp M M 1 2 3 4 5 6 7 8 M 10 11 12 13 14 15 16 17 M

Antibiotic susceptibility The most efficient antibiotics tested against L. garvieae isolates were florphenicol, ceftiofur and erythromicin – zone of inhibition 30 and 28 cm

CONCLUSION In conclusion, the bacterium isolated from diseased rainbow trout was identified as Lactococcus garvieae. L. garvieae is possible to be misidentified in microbiology laboratories, due to its similarity with members of the genus Enterococcus, and because of the previous reliance on phenotypic methods. We were unable to identify the isolates when using just main routine biochemical assays, indicating the importance of serological and molecular methods for the correct diagnosis of this pathogen in aquatic animals. The most efficient antibiotics against L. garvieae are erytromicin, florphenicol and cefalosporins.

THANK YOU! CORRESPONDENCE ADDRESS: petyorozova@gmail. com
- Slides: 21