Lactic acid bacteria for nutraceuticals and added value

Lactic acid bacteria for nutraceuticals and added value products production Enrica PESSIONE Department of life Sciences and Systems Biology. University of Torino- Italy London, August 18 th 2016

In spite of the long established use of LAB , their biotechnological potential nutraceutical industry is still: • Underexplored • Underexploited for the food and
LACTIC ACID BACTERIA Biocontrol agents Probiotics Food starters Cell factories
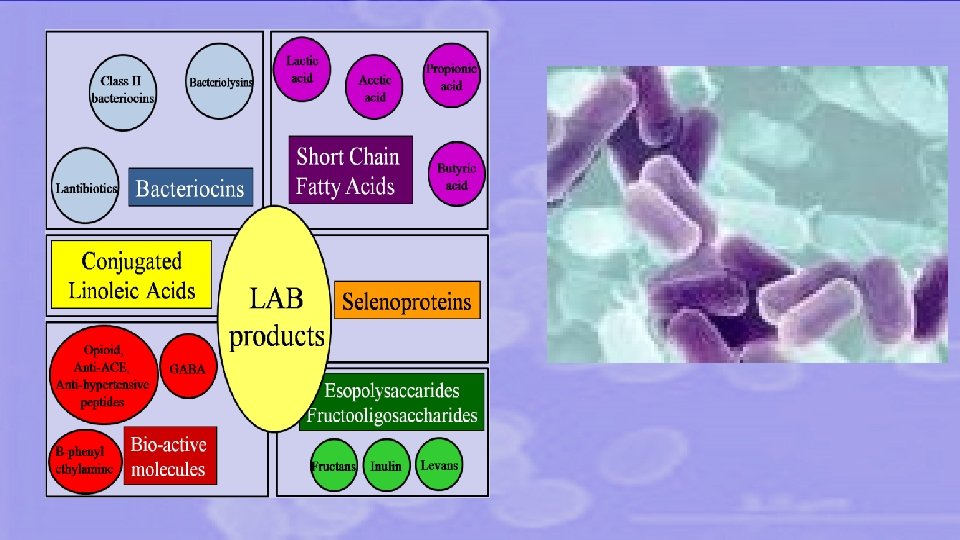
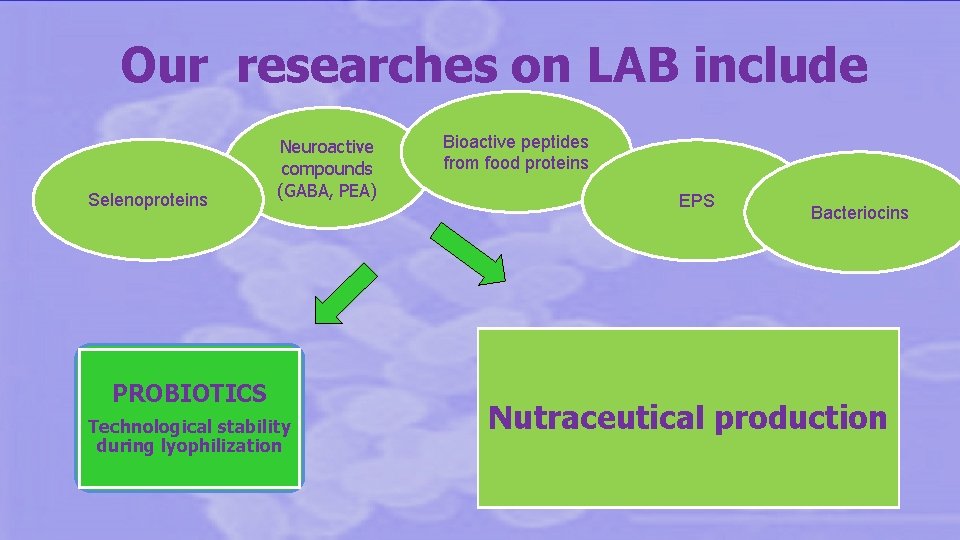
Our researches on LAB include Selenoproteins Neuroactive compounds (GABA, PEA) PROBIOTICS Technological stability during lyophilization Bioactive peptides from food proteins EPS Bacteriocins Nutraceutical production
PROTEOMICS Reconstructing metabolic pathways Evaluating stress resistance Optimizing the technological operative conditions

Detection of enzymes involved generation of desired metabolites: • Proteases • Glycosyltransferases • Amino acid decarboxylases • Selenocysteine lyase in

Detection of proteins controlling stress: • Acid stress • Oxidative stress • Starvation • Osmotic stress • Competition stress ( co-cultures) • Heat/cold stress
SUBPROTEOMES

Probiotic LAB releasing seleno proteins

Selenium plays an important role in: • Antioxidant activity • tyroid function • healthy immune response • viral infections • regulation of inflammatory pathways • protection against some forms of cancers • ageing

Selenium in nature

Selenium chemical forms • Selenium can be present in different chemical forms: • Selenite • Selenate • Elemental selenium • Selenomethionine • Selenocysteine Some form are toxic or not bioavailable for humans

SELENIUM-FIXING LAB some Lactobacillus strains ( L. reuteri Lb 2 BM) are able to fix Selenium depending on the oxidation state of Se: • when it is added to the medium as selenate only a very low amount of Se was intracellularly detected • conversely the highest Se concentration in the biomass was obtained (Na 2 Se. O 3= 4. 38 mg/L) with sodium selenite

SELENIUM-FIXING LAB • the excess of sodium selenite not used for insertion into proteins is reduced to elemental selenium Se(0). • This is a non-toxic form of Se that produces deposits at surface level giving a red color to the biomass (Lamberti et al. 2011).
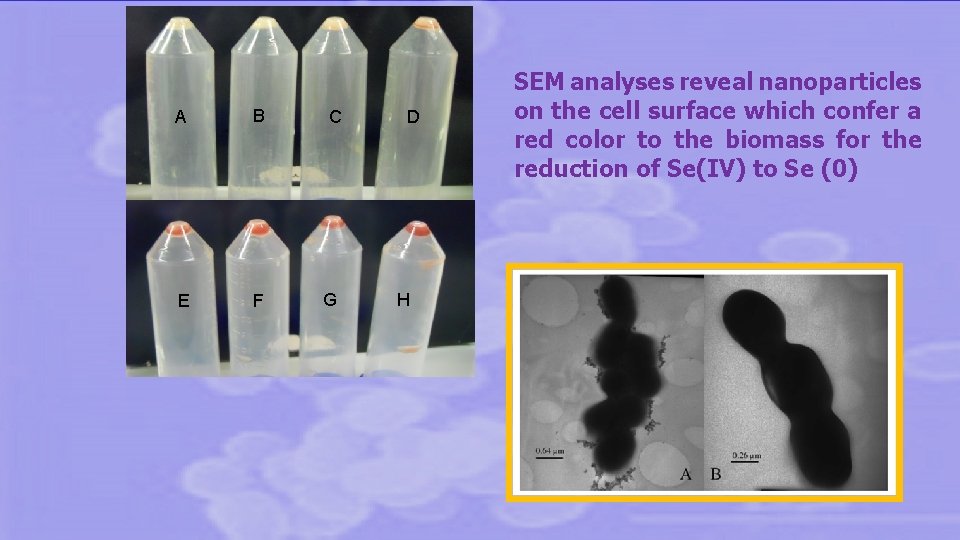
A B C E F G D H SEM analyses reveal nanoparticles on the cell surface which confer a red color to the biomass for the reduction of Se(IV) to Se (0)
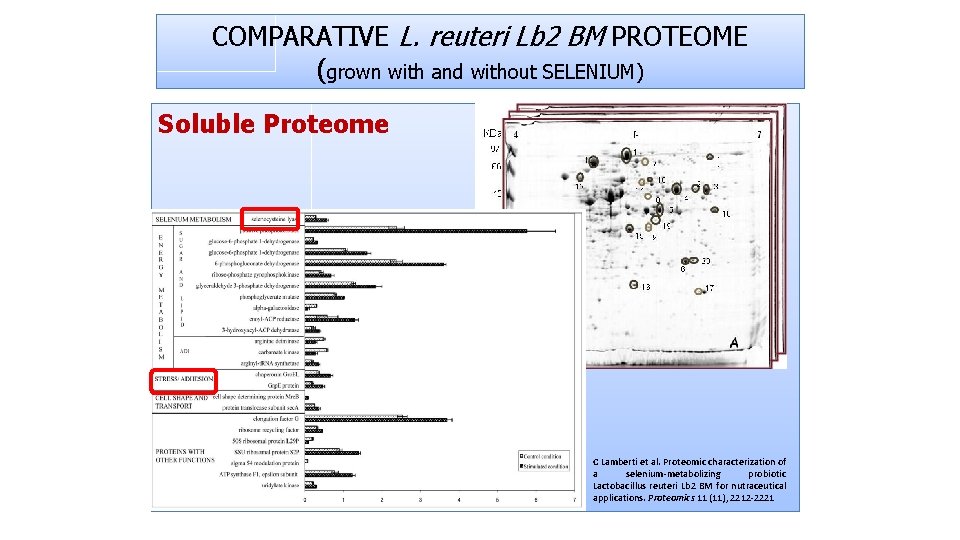
COMPARATIVE L. reuteri Lb 2 BM PROTEOME (grown with and without SELENIUM) Soluble Proteome C Lamberti et al. Proteomic characterization of a selenium‐metabolizing probiotic Lactobacillus reuteri Lb 2 BM for nutraceutical applications. Proteomics 11 (11), 2212‐ 2221
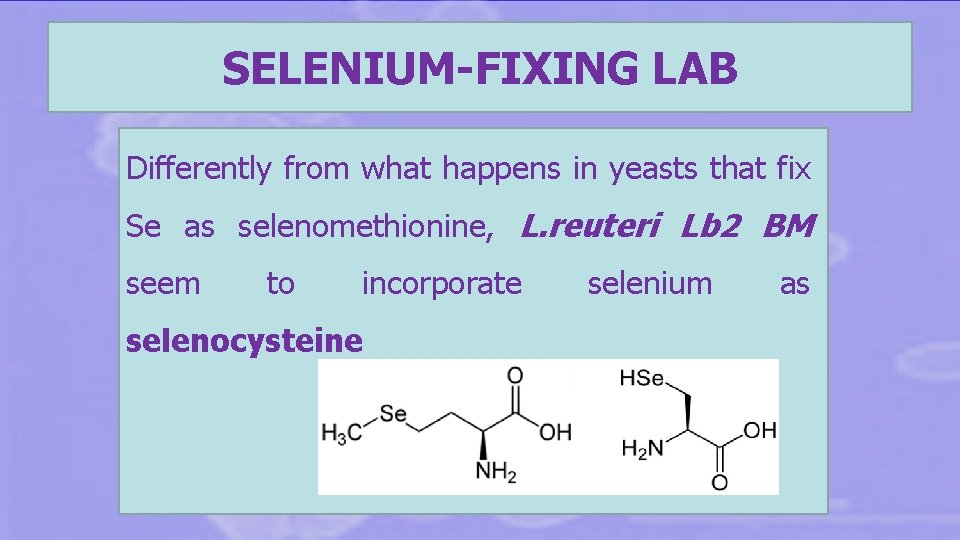
SELENIUM-FIXING LAB Differently from what happens in yeasts that fix Se as selenomethionine, L. reuteri Lb 2 BM seem to incorporate selenocysteine selenium as
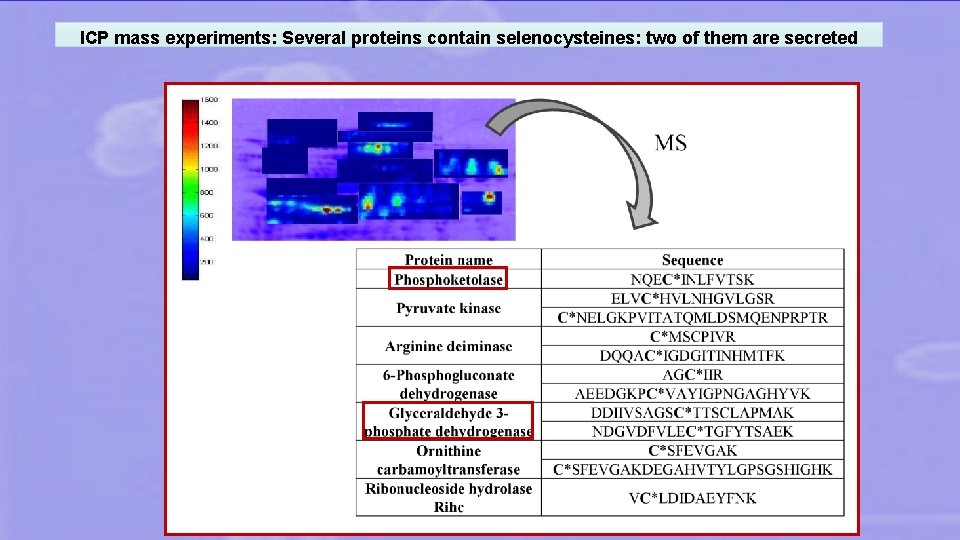
ICP mass experiments: Several proteins contain selenocysteines: two of them are secreted

COMPARATIVE EXTRACELLULAR PROTEOME • Lactobacillus reuteri Lb 2 BM is able to fix Se in 2 extracellular proteins (phosphoketolase and GAP dehydrogenase) as Se. Cys. • In the near future Se-enriched lactobacilli could be used as food supplements to supply Sec-containing proteins.

LAB PRODUCING NEUROMODULATING COMPOUNDS

GABA Gamma amino butyric acid
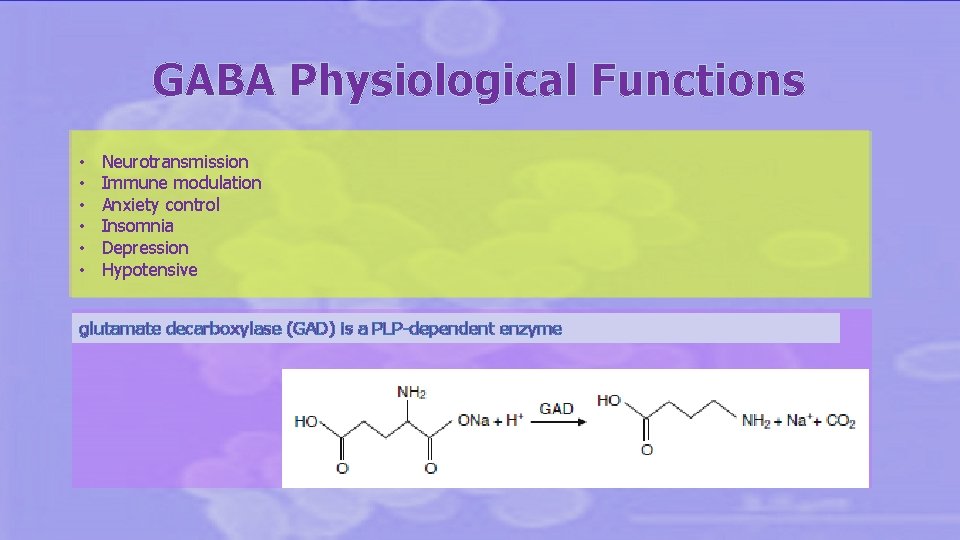
GABA Physiological Functions • • • Neurotransmission Immune modulation Anxiety control Insomnia Depression Hypotensive glutamate decarboxylase (GAD) is a PLP-dependent enzyme glutamate decarboxylase enzyme (GAD)

Gamma amino butyric acid is produced as the result of the peculiar energy metabolism of LAB mainly based on lactic fermentation. ( No heme, no respiration) Amino acid decarboxylations coupled with antiport systems allow to increase the proton gradient across the membrane together with cytoplasm basification
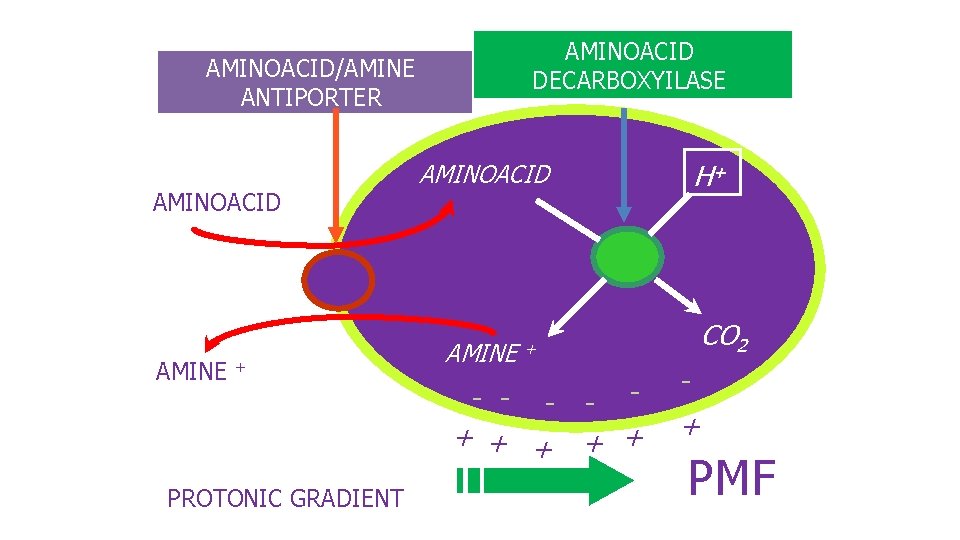
AMINOACID DECARBOXYILASE AMINOACID/AMINE ANTIPORTER AMINOACIDO AMINE + PROTONIC GRADIENT H+ AMINOACID AMINE CO 2 + - - + + + PMF
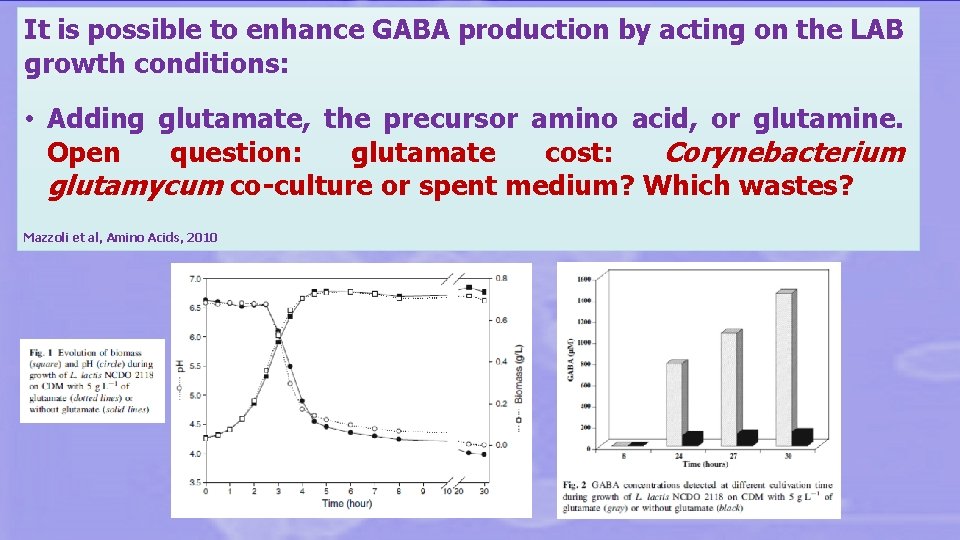
It is possible to enhance GABA production by acting on the LAB growth conditions: • Adding glutamate, the precursor amino acid, or glutamine. Open question: glutamate cost: Corynebacterium glutamycum co-culture or spent medium? Which wastes? Mazzoli et al, Amino Acids, 2010
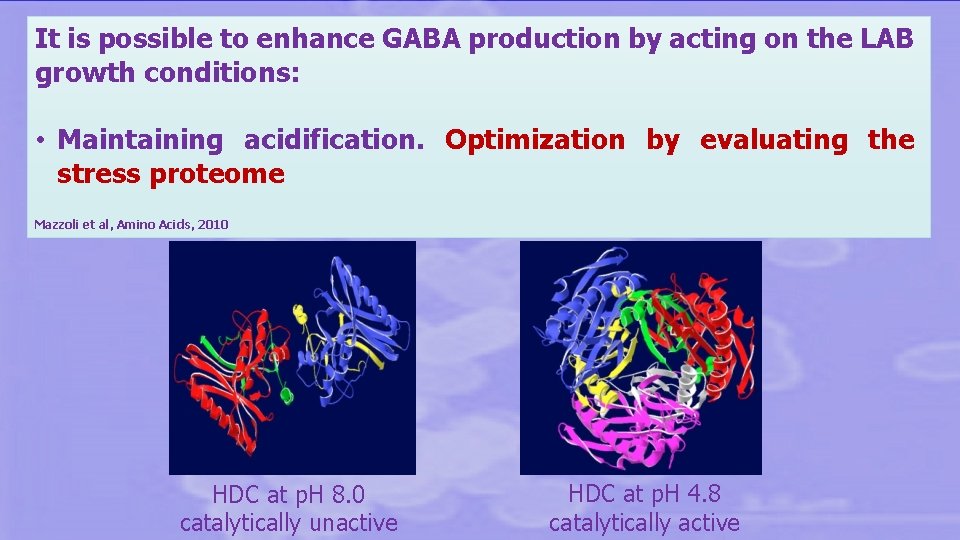
It is possible to enhance GABA production by acting on the LAB growth conditions: • Maintaining acidification. Optimization by evaluating the stress proteome Mazzoli et al, Amino Acids, 2010 HDC at p. H 8. 0 catalytically unactive HDC at p. H 4. 8 catalytically active

It is possible to enhance GABA production by acting on the LAB growth conditions: • Maintaining a high chloride content (transcriptional activator of GAD gene) Optimization by evaluating the stress proteome • Adding arginine to supply an additional NH 3 donor for transaminations to direct all glutamate or glutamine to GABA

GABA-TEA GABA-RICE YOGURT FERMENTED SOYBEAN

BIOACTIVE PEPTIDES

BIOACTIVE PEPTIDES • • Antioxidant Metal chelating Antimicrobial Immunomodulating • Antithrombotic • Antihypertensive • Cholesterol lowering • Opioid • Anti-opiod Infection Inflammation Ageing Cardiovascular diseases Anxiety Depression Mood
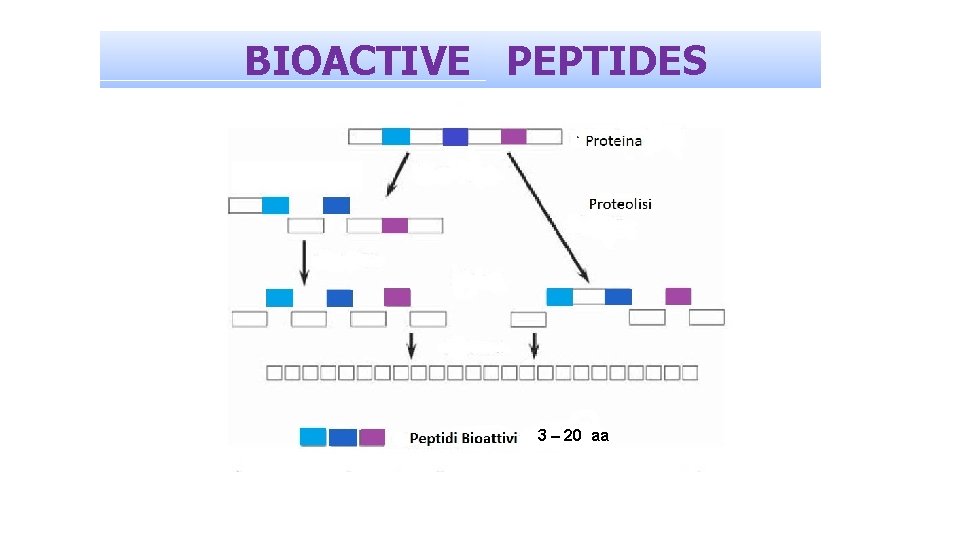
BIOACTIVE PEPTIDES 3 – 20 aa
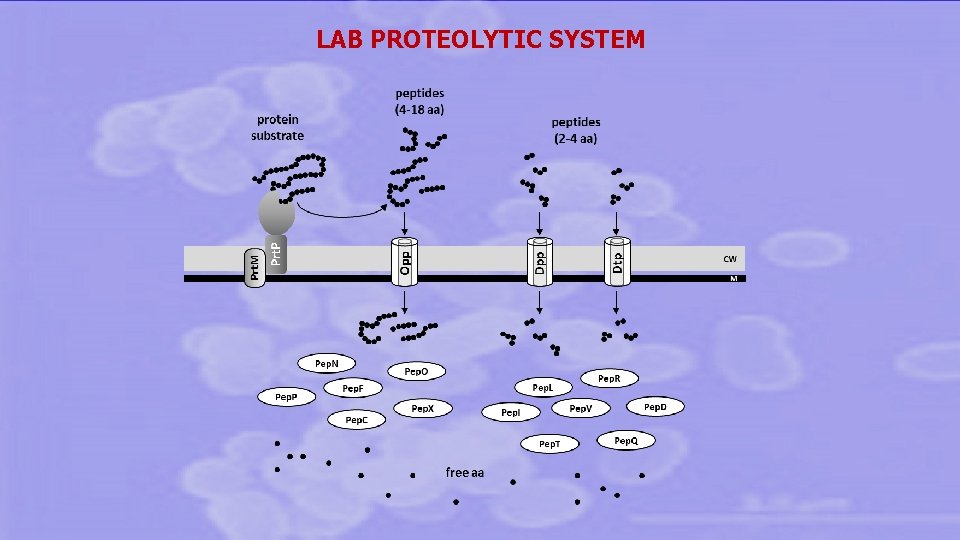
LAB PROTEOLYTIC SYSTEM

The proteolytic system of three LAB strains was evaluated towards GLUTEN and CASEIN
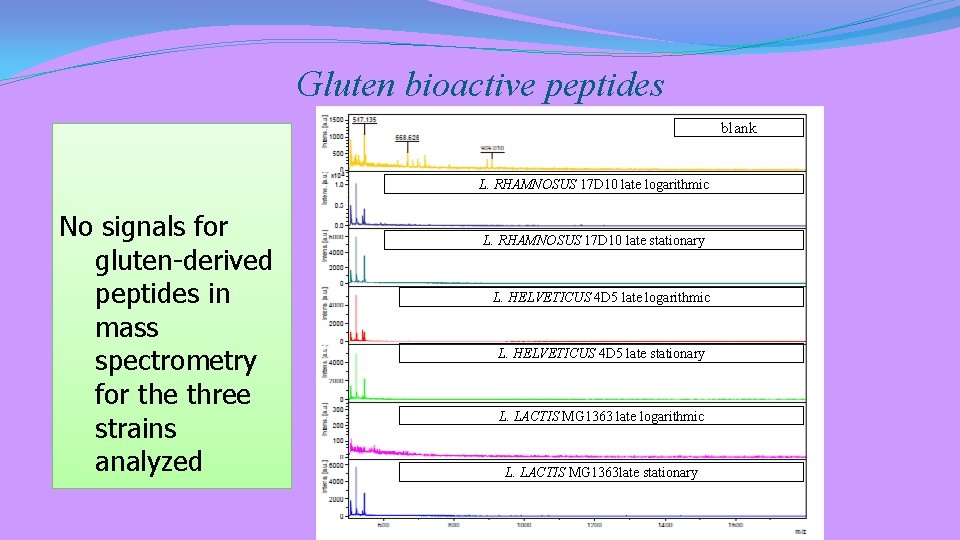
Gluten bioactive peptides blank L. RHAMNOSUS 17 D 10 late logarithmic No signals for gluten-derived peptides in mass spectrometry for the three strains analyzed L. RHAMNOSUS 17 D 10 late stationary L. HELVETICUS 4 D 5 late logarithmic L. HELVETICUS 4 D 5 late stationary L. LACTIS MG 1363 late logarithmic L. LACTIS MG 1363 late stationary
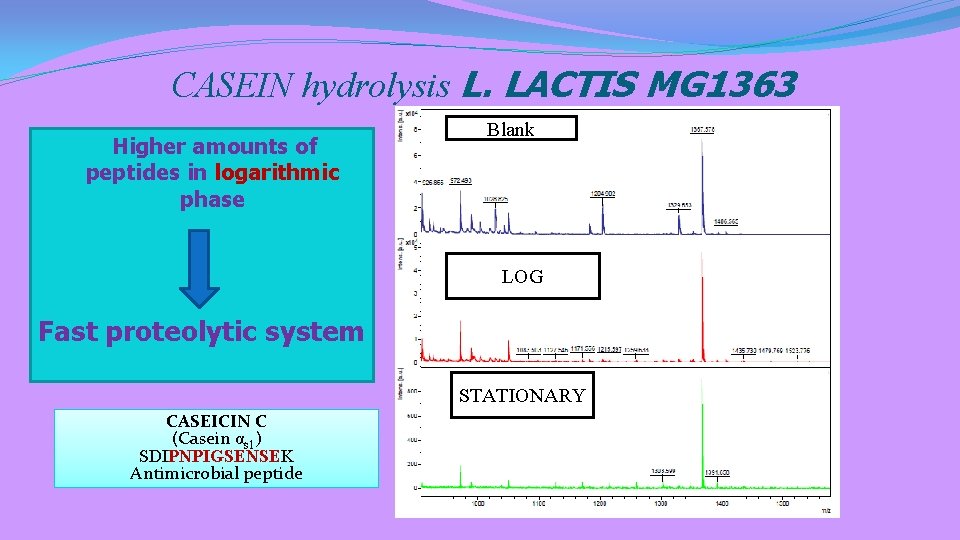
CASEIN hydrolysis L. LACTIS MG 1363 Higher amounts of peptides in logarithmic phase Blank LOG Fast proteolytic system STATIONARY CASEICIN C (Casein αs 1) SDIPNPIGSENSEK Antimicrobial peptide
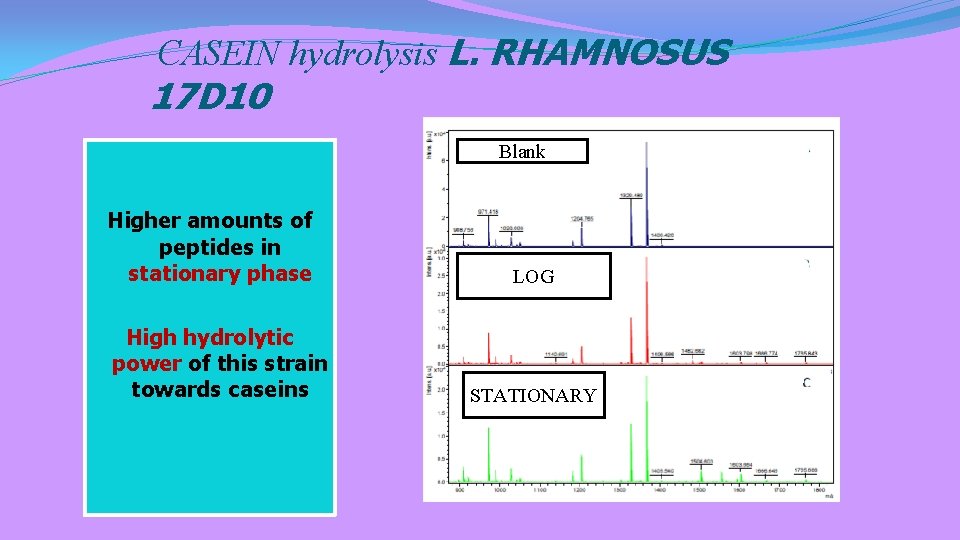
CASEIN hydrolysis L. RHAMNOSUS 17 D 10 Blank Higher amounts of peptides in stationary phase High hydrolytic power of this strain towards caseins LOG STATIONARY

Antimicrobial peptides by L. rhamnosus 17 D 10 ISRACIDIN (Caseina αs 1) RPKHPIKHQGLPQEVLNENLLRF (Hayes et al. , 2007) CASOCIDIN (Caseina αs 2) KTKLTEEEKNRLNFLKKISQRYQKFALPQYLKTVYQHQK

LOW-COST SUBSTRATES Milk whey proteins
BACTERIOCINS

BACTERIOCINS • Antibacterial compounds generally produced under quorum sensing control • Active a very low concentration ( nanomolar) • Small peptides or proteins • Secreted or surface-bound • LAB are good producers: nisin, lactacine, enterocine, reutericine, sakacine, salivaricine, mutacine, gassericine

BACTERIOCINS • Promising tools for infection control as antibiotic substitutes (urgent issue!) • During about 50 years use of nisin no resistance spread. • Useful in prolonging shelf-life protecting food from spoilage bacteria, thus reducing the use of preservatives, high salt, high sugar, cold chain. • Useful to control monocytogenes) food-borne infections (Listeria
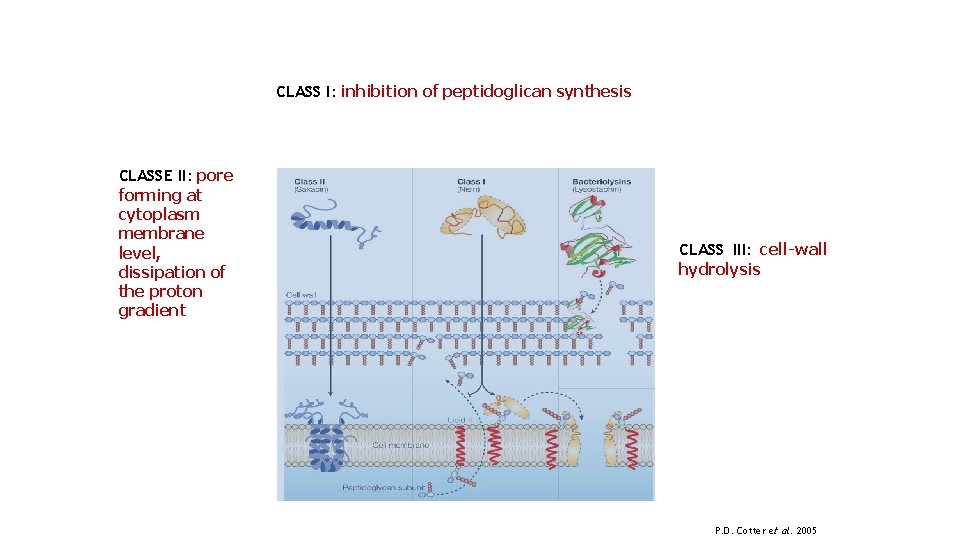
CLASS I: inhibition of peptidoglican synthesis CLASSE II: pore forming at cytoplasm membrane level, dissipation of the proton gradient CLASS III: cell-wall hydrolysis P. D. Cotter et al. 2005
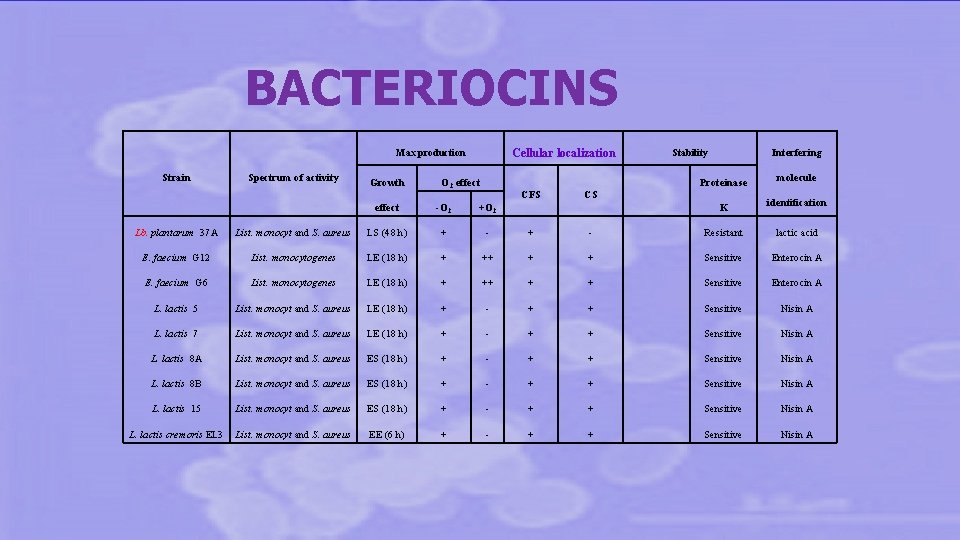
BACTERIOCINS Cellular localization Max production Strain Spectrum of activity Growth O 2 effect CFS effect -O 2 +O 2 Interfering Stability Proteinase molecule K identification CS Lb. plantarum 37 A List. monocyt and S. aureus LS (48 h) + - Resistant lactic acid E. faecium G 12 List. monocytogenes LE (18 h) + ++ + + Sensitive Enterocin A E. faecium G 6 List. monocytogenes LE (18 h) + ++ + + Sensitive Enterocin A L. lactis 5 List. monocyt and S. aureus LE (18 h) + - + + Sensitive Nisin A L. lactis 7 List. monocyt and S. aureus LE (18 h) + - + + Sensitive Nisin A L. lactis 8 A List. monocyt and S. aureus ES (18 h) + - + + Sensitive Nisin A L. lactis 8 B List. monocyt and S. aureus ES (18 h) + - + + Sensitive Nisin A L. lactis 15 List. monocyt and S. aureus ES (18 h) + - + + Sensitive Nisin A L. lactis cremoris EL 3 List. monocyt and S. aureus EE (6 h) + - + + Sensitive Nisin A

BACTERIOCINS: overlapping gel test

BACTERIOCINS • Some limits concern their peptide/protein nature: they are easily degraded by proteases both in the food matrix ( during cheese ripening) and in vivo ( gastrointenstinal system) • A promising strategy is to immobilize them into the food packaging, after suitable enrichement and purification. • The advantage of food packaging-immobilized bacteriocins, « active packaging» results in : a) Limited and controlled diffusion into the food matrix, limited degradation b) Release in a concentration gradient c) Protection from inactivation

BACTERIOCINS: challenges Growing LAB on a lowcost substrate Stimulate production (oxigen stress, co-coltures in membrane fermenter, bacteria hydrolysates) Optimizing recovery (Recover also surfacebound by shaving) Film-bacteriocin compatibility (Enhanced diffusion into polymers) Improve performances conservative modifications to: • Extend the antibacterial spectrum • Enhance protease resistance

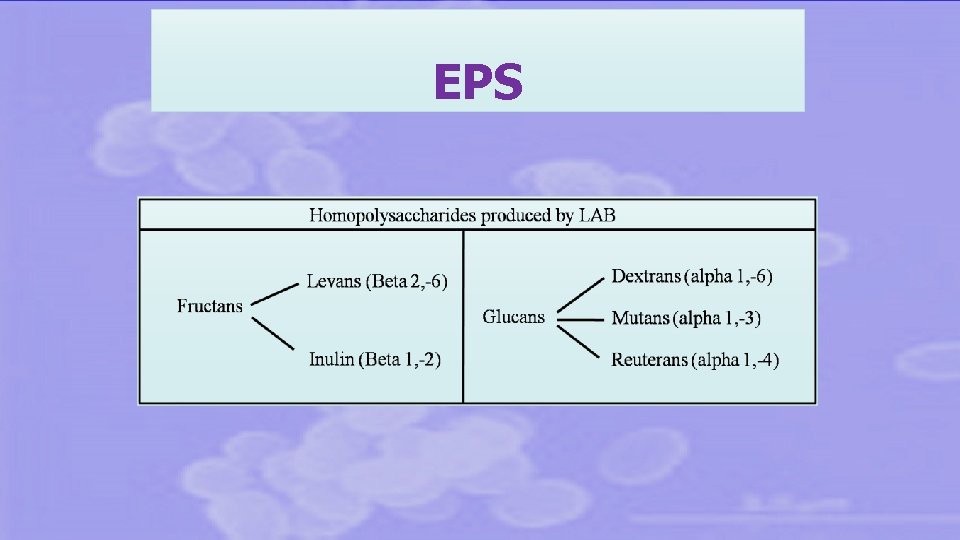
EXOPOLYSACCHARIDES

EXOPOLYSACCHARIDES food industry • Thickeners • Texture improvement • Stabilizers for low-fat products human health • Prebiotics (FOS= low molecular mass EPS) • Immunomodulating • Cholesterol lowering biotechnological industry • Chromatographic media
EPS
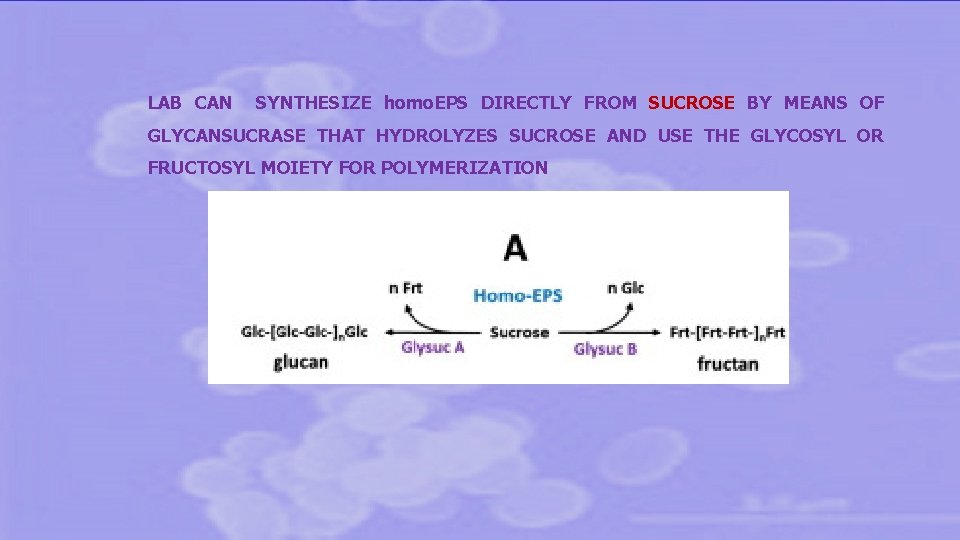
LAB CAN SYNTHESIZE homo. EPS DIRECTLY FROM SUCROSE BY MEANS OF GLYCANSUCRASE THAT HYDROLYZES SUCROSE AND USE THE GLYCOSYL OR FRUCTOSYL MOIETY FOR POLYMERIZATION

Enhancers of homoexopolysaccharide production: • Sucrose • Temperature: the highest the growth temperature the lowest EPS production. Preliminary proteomic results on L. plantarum Q 8. 23 reveal induction of stress proteins during active growth ( 37°) but not under EPS production conditions ( 30°).
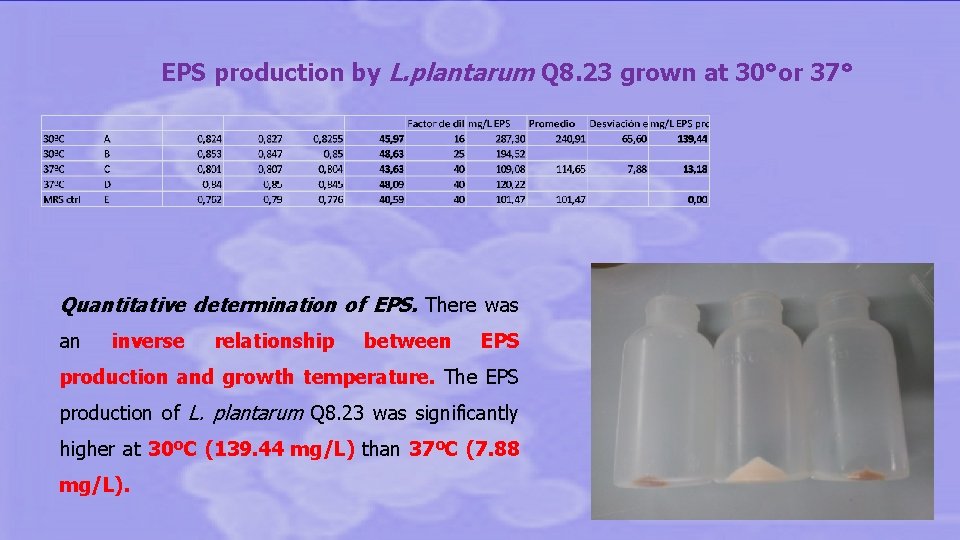
EPS production by L. plantarum Q 8. 23 grown at 30°or 37° Quantitative determination of EPS. There was an inverse relationship between EPS production and growth temperature. The EPS production of L. plantarum Q 8. 23 was significantly higher at 30ºC (139. 44 mg/L) than 37ºC (7. 88 mg/L).

• The results of our researches demonstrate that LAB potential is much higher than expected and that it is possible to exploit them for nutraceuticals and added value chemical production. • It is important to continue to study LAB proteomes to enhance our knowledge on possible future applications in both industrial and medical fields.
THANK YOU!
- Slides: 55