lactamase inhibitors Almost all have weak antibacterial activity

β-lactamase inhibitors �Almost all have weak antibacterial activity. �Important in combination with penicillins sensitive to β-lactamase degradation. �Clavulanic acid is the first one of this class. � Natural product from streptomyces. � Has a powerful and irreversible inhibition of βlactamase enzymes because it will covalently bind to two positions in the active site. � Normally used in combination with amoxicillin and other β-lactamase sensitive penicillins

SAR for β-lactamase inhibitors �β-lactam ring is essential. �The enol ether have a rule in binding. �The elkene moiety should have Z configuration which is much more active than the E isomer. �No substitution at C 6. �R stereochemistry at C 3. �Carboxylic acid at C 3 is essential.

Cephalosporins. �The first agent discovered was cephalosporin C, obtained from the fungus cephalosporium acremonium. � 1/1000 the antibacterial activity of penicillin G. �Has the same mechanism of action as penicillins (inhibits cell wall cross linking). �Has greater stability toward acid and β-lactamase.

SAR of cephalosporins �The bicyclic system is essential. �The carboxylic acid at C 4 is essential. �Acylamino group at C 7 is essential. �Acetyloxy group at C 3 is important and act as a leaving group when the molecule binds to transpeptidase.

Synthesis of Cephalosporins �Unlike 6 -APA, 7 ACA was difficult to isolate and purify. �Instead, 7 ACA was synthesized from cephalosporin C as follows

st 1 Generation Cephalosporins �Have lower activity than penicillin but they have broader spectrum action. �Still susceptible to β-lactamase degradation. �Steric shield helped to improve stability toward βlactamase degradation but proved to reduce antibacterial activity.

st 1 Generation Cephalosporins �Have the good leaving group, pyridinium ion. . This improved activity. �This group is not hydrolysable compared to the acetyloxy group found in cephalothin. �Poorly absorbed from the gut because it will be ionized all the time. �Only given parenterally.

nd 2 Generation Cephalosporins �They have methoxy group at C 7 which make them active against the resistant strains. �They have a carbamate group at C 3 that increase stability toward hydrolysis compared to the acetyloxy group found in 1 st generation derivatives.

nd 2 Generation Cephalosporins �Other agents are the oximinocephalosporins: �Have the iminomethoxy group at the α-carbon in the acyl side chain, this increased stability toward βlactamase.

rd 3 Generation Cephalosporins �Here the aminothiazole ring has replaced the furan ring of cefuroxime: �This enhanced the penetration through the outer membrane of gram –ve bacteria, �Increase the affinity for transpeptidase. �Not recommended as first line therapy to prevent the rapid development of resistance.

rd 3 Generation Cephalosporins �Cefdinir (Omnicef®): �It has a broad spectrum activity. �More active on gram –ve bacterial infections such as respiratory, skin and soft tissues infections. �Estimated oral bioavailability is 20 -25% (WHY? ). �Log. P = 0. 02 �p. Ka = 3. 27

th 4 Generation Cephalosporins �They have a positively charged group at C 3 which become a good leaving group during the binding with transpeptidase. �They are more polar than the old generation, better penetration for the outer membrane of gram –ve bacteria. �More stable toward β-lactamase.

New generation Cephalosporins Cephtobiprole � 5 th generation cephalosporin (2008). �Only given IV (Why? ). �Resistant to staphylococcal β-lactamase (Why? ). �activity against methicillin-resistant S. aureus, penicillinresistant S. pneumoniae, P. aeruginosa, and Enterococci.

New generation Cephalosporins Cefsulodin � 3 rd generation cephalosporin. �has very specific activity against P. aeruginosa. �limited activity against Gram-positive bacteria and anaerobic bacteria. �Is not clinically used nowadays (difficult to purify during synthesis).

New generation Cephalosporins Ceftaroline � 5 th generation cephalosporins. �It retains the activity of later generation cephalosporins having broad spectrum activity against Gram -ve and gram +ve bacteria especially on resistant strains. �Approved for clinical use in USA in 2010. �Still in clinical trials (phase III).

New generation Cephalosporins Cefmenoxime rd generation cephalosporins. � 3 �It is mainly active on gram –ve bacteria. �Only available as intramuscular injections. �Log. P = - 0. 87
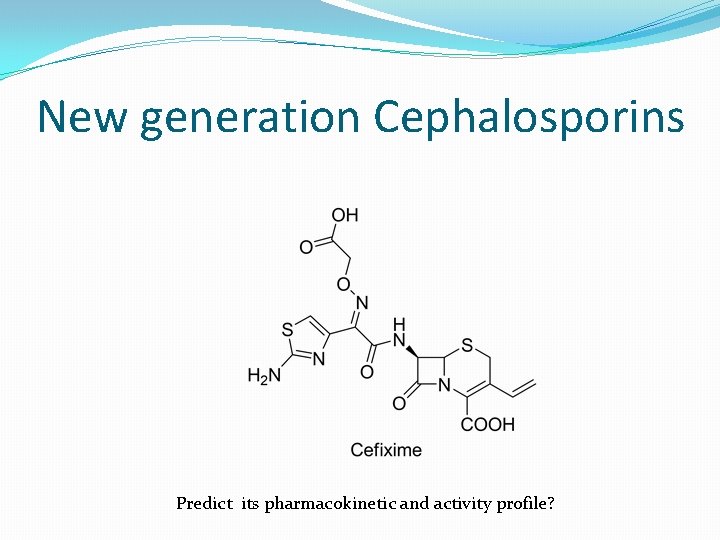
New generation Cephalosporins Predict its pharmacokinetic and activity profile?

Carbapenems �No thiazolidine or dihydrothiazine ring. �They have two strained rings which decrease the chemical stability as well as acid stability. �The inverse stereochemistry at C 6 and the presence of hydroxyl group increase stability toward β-lactamase enzymes. �They have broad spectrum activity.

Monobactams �It has a limited activity against gram +ve bacteria. �Because it does not have the fused ring system, Aztreonam is believed to have different mechanism of action. . �highly polar structure which reduce the oral bioavailability… it is recommended to be given parenterally
- Slides: 19