Labs 6 7 Diffusion and Osmosis Diffusion dialysis

Labs 6 & 7 Diffusion and Osmosis

Diffusion • dialysis tubing filled with 0. 15 mg/ml KMn. O 4 • tubing placed into a beaker of water • one beaker kept at into room temp water, the other placed into ice cold water bath
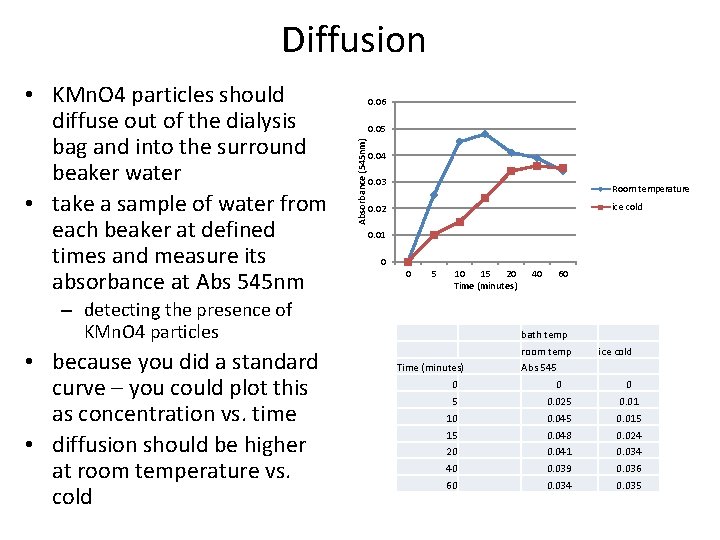
Diffusion 0. 06 0. 05 Absorbance (545 nm) • KMn. O 4 particles should diffuse out of the dialysis bag and into the surround beaker water • take a sample of water from each beaker at defined times and measure its absorbance at Abs 545 nm 0. 04 0. 03 Room temperature ice cold 0. 02 0. 01 0 0 5 10 15 20 Time (minutes) – detecting the presence of KMn. O 4 particles • because you did a standard curve – you could plot this as concentration vs. time • diffusion should be higher at room temperature vs. cold 40 60 bath temp room temp Time (minutes) ice cold Abs 545 0 0 0 5 0. 025 0. 01 10 0. 045 0. 015 15 0. 048 0. 024 20 0. 041 0. 034 40 0. 039 0. 036 60 0. 034 0. 035
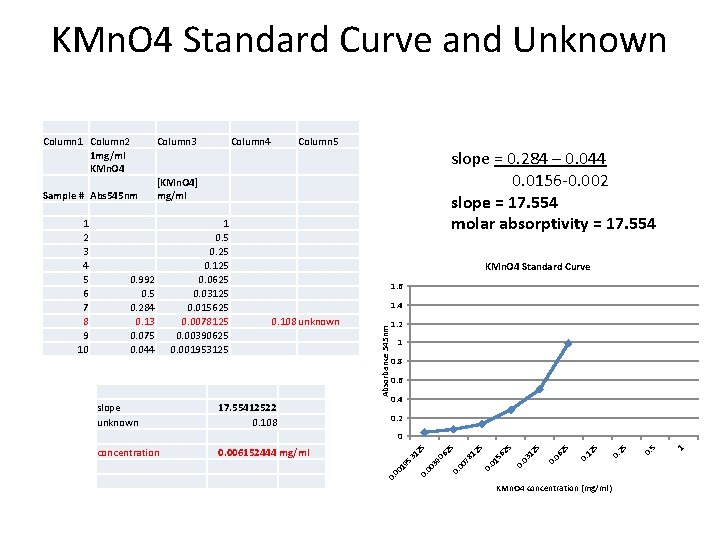
KMn. O 4 Standard Curve and Unknown Sample # Abs 545 nm Column 4 Column 5 slope = 0. 284 – 0. 044 0. 0156 -0. 002 slope = 17. 554 molar absorptivity = 17. 554 [KMn. O 4] mg/ml 0. 992 0. 5 0. 284 0. 13 0. 075 0. 044 slope unknown 1 0. 5 0. 25 0. 125 0. 0625 0. 03125 0. 015625 0. 0078125 0. 00390625 0. 001953125 KMn. O 4 Standard Curve 1. 6 1. 4 0. 108 unknown 17. 55412522 0. 108 1. 2 1 0. 8 0. 6 0. 4 0. 2 KMn. O 4 concentration (mg/ml) 1 0. 5 0. 25 5 0. 12 5 25 06 0. 25 12 0. 03 56 01 12 5 0. 06 78 25 0. 00 39 00 53 12 0. 006152444 mg/ml 19 concentration 5 0 0. 00 1 2 3 4 5 6 7 8 9 10 Column 3 Absorbance 545 nm Column 1 Column 2 1 mg/ml KMn. O 4

Diffusion KMn. O 4 concentration (mg/ml) • you have already calculated the molar absorptivity for KMn. O 4 (see spec lab) • but you did a standard curve and you could recalculate it • for each absorbance value – divide by the molar absorptivity to get the concentration • the units are the same as the standard – which was mg/ml • plotting concentration vs. time looks just like absorbance vs. time 0. 003 0. 0025 0. 002 0. 0015 room temperature ice cold 0. 001 0. 0005 0 0 5 Time (minutes) 0 5 10 15 20 40 60 10 15 20 Time (minutes) 40 60 room temp ice cold mg/ml 0 0 0. 00142418 0. 00057 0. 00256352 0. 000855 0. 00273442 0. 001367 0. 00233565 0. 001937 0. 00222172 0. 002051 0. 00193688 0. 001994

Diffusion • place a crystal of KMn. O 4 in a petri dish of water and measure its spread through the water over time – diffusion results in the increasing diameter of the KMn. O 4 “cloud” in the water • place a drop of Na. OH in an agar dish and measure its diffusion through the agar – agar is a colloid and will slow the diffusion of Na. OH vs. water • same concept at the dialysis tubing study – high concentration to low concentration
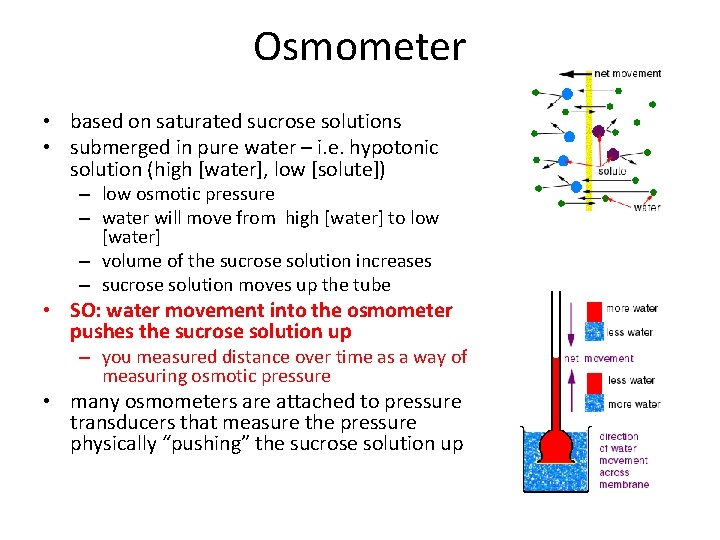
Osmometer • based on saturated sucrose solutions • submerged in pure water – i. e. hypotonic solution (high [water], low [solute]) – low osmotic pressure – water will move from high [water] to low [water] – volume of the sucrose solution increases – sucrose solution moves up the tube • SO: water movement into the osmometer pushes the sucrose solution up – you measured distance over time as a way of measuring osmotic pressure • many osmometers are attached to pressure transducers that measure the pressure physically “pushing” the sucrose solution up
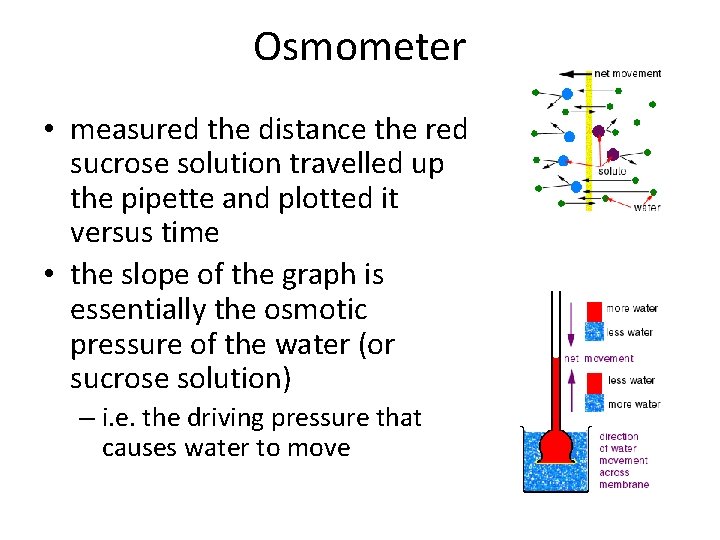
Osmometer • measured the distance the red sucrose solution travelled up the pipette and plotted it versus time • the slope of the graph is essentially the osmotic pressure of the water (or sucrose solution) – i. e. the driving pressure that causes water to move

Potatoes and Turgidity • immersed pieces of potato in increasing molarities of sucrose – 0. 0 M sucrose = pure water – up to 1. 0 M sucrose • each sucrose solution has a defined number of sucrose particles and free water molecules – the higher the concentration – the higher the osmotic pressure of the solution • the osmotic pressure of the potato is defined also – it has to be compared to the OP of the surrounding sucrose solution in order to figure out which way water will flow – hypotonic solutions – water flows out of solution into cells – hypertonic solutions – water flows into solutions from the cells • increased water movement into the potato slices will increase its weight
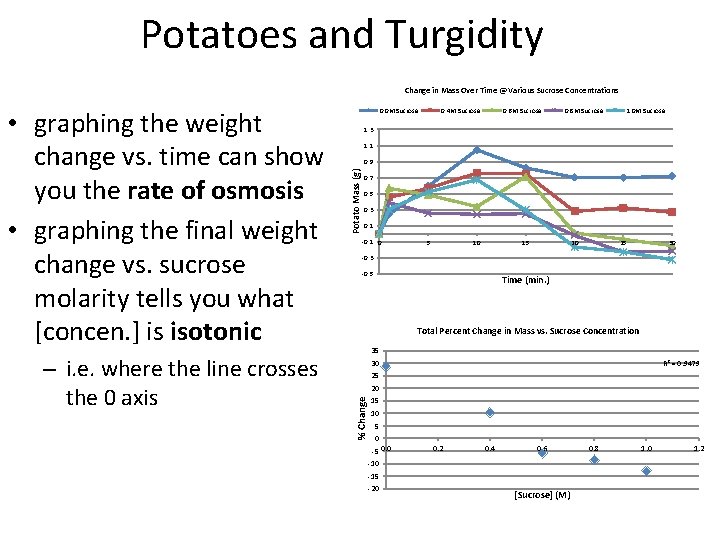
Potatoes and Turgidity Change in Mass Over Time @ Various Sucrose Concentrations – i. e. where the line crosses the 0 axis 0. 4 M Sucrose 0. 6 M Sucrose 0. 8 M Sucrose 1. 0 M Sucrose 1. 3 1. 1 Potato Mass (g) 0. 9 0. 7 0. 5 0. 3 0. 1 -0. 1 0 5 10 15 20 25 30 -0. 3 -0. 5 Time (min. ) Total Percent Change in Mass vs. Sucrose Concentration 35 R 2 = 0. 9479 30 25 20 % Change • graphing the weight change vs. time can show you the rate of osmosis • graphing the final weight change vs. sucrose molarity tells you what [concen. ] is isotonic 0. 0 M Sucrose 15 10 5 0 -5 0. 0 0. 2 0. 4 0. 6 -10 -15 -20 [Sucrose] (M) 0. 8 1. 0 1. 2

• go to this website and try the self quizzes at the ends of the exercises: http: //www. phschool. com/science/biology_ place/labbench/lab 1/intro. html
- Slides: 11