Laboratory rabbit Dep laboratory animal science Dr kourosh

Laboratory rabbit Dep. laboratory animal science Dr. kourosh pajaei D. V. M

Rabbit Taxonomy & breeds

TAXONOMY Phylum Cordata : animal with notochord & gills. sub phylum Craniata ( vertebrata ): chordata with organaized head region. class Mammalia: Warm-blooded with hair coat. sub class Theria : Viviparous Infraclass Eutheria : placental mammals Cohort Glires: Lagomorpha plus rodens Order Lagomorpha: Gnawing placentals with chisellike incisors of which there is in the upper jaw a small second pair directly behind the main pair. Family: Leporidea Subfamily: Leporinae Geneus: Oryctolagus ( true rabbit )(6 Gs )

Rabbit breeds G: Oryctolagus cuniculus European rabbit ( true rabbit)

Rabbits • Developed into forty-five recognized breeds • Divided into five weight categories 1. Dwarf or Miniature 2. Small 3. Medium 4. Large 5. Giant

q. Dwarf or Miniature Breeds Ø Britannia Petite Ø Netherland Dwarf Ø Himalayan

ØBrittania Petite • Old English breed • Smallest of the standard breeds • Rabbits are white with red eyes in the US

ØNetherland Dwarf • Developed in Holland • More recognized colors than any other breed • Rabbits are small, stocky, and compact

Ø Himalayan • One of the oldest and widest distributions • Young rabbits are white and slightly tinged with silver-gray • Matures rabbits become snow white with deep black nose, ears, feet, and tail

q. Small Breeds • Dutch • Tan • Florida White

ØDutch • Originated in Holland developed in England • Unique color markings – colored patches on each side of the head that encircle the eyes and ears – rear half is also colored the same as the head

ØTan • Originated in England • Originally black with tan underside – Tan color found around eyes, nostrils, and toes

ØFlorida White • Developed in Florida – Crossed with Dutch, Polish, and New Zealand Whites • Short and compact – well-rounded hips and hindquarters • Bred for a small meat rabbit or a laboratory rabbit

q. Medium Breeds ØEnglish Spot ØStandard Chinchilla ØEnglish Angora ØBelgian Hare ØRhinelander ØRex

ØEnglish Spot • Introduced to US from England • A unique chair of markings, the size of a pea, run from the base of the ear to the A unique chair of markings flank

ØStandard Chinchilla • Under-color is dark slate blue at the base, pearl in the middle and narrow black band with narrow light band on the top

ØEnglish Angora • Has a short, compact body and the head, ears, feet, and body are covered with wool

ØBelgian Hare • Originated in Flanders, Belgium • Has a long, fine body, long, straight and slender legs

ØRhinelander • Has a butterfly marking on the nose, cheek spots, and eye rings • Body is white – herringbone pattern or unbroken stripe running from the neck down the back to the top of the tail

ØRex • Short hair coat, with guard hairs being the same length as the under fur • The fur has a very soft, flush feel

q. Large Breeds ØCalifornian ØCinnamon ØAmerican ØEnglish Lop ØNew Zealand ØSilver Fox

ØCalifornian • Cross of a Himalayan and a Chinchilla and crossed with White New Zealand • White with black colored nose, ears, feet, and tail, and the eyes are red

ØCinnamon • Medium-length rabbit with a well-filled meaty body • The color is a rust or cinnamon ground color; the fur is ticked with smoke-gray across the back

ØAmerican • Ideal body type is described as “mandolin” because the arch of the back has this type of appearance

ØEnglish Lop • Oldest domestic rabbit breed known • Body is medium length with well arched back and low head carriage

ØNew Zealand • Leading choice for commercial meat production • White is preferred because the small hairs that remain cannot be seen

ØSilver Fox • Developed in Ohio • Resembles the real Silver Fox with silvered fur and lack of fly-back fur (fur returning to its original position)

q. Giant ØGiant Angora ØFrench Lop ØFlemish Giant

ØGiant Angora • Developed for maximum amount of wool production • Has the body structure of a commercial animal with a unique coat structure

ØFrench Lop • Ears are shorter than the English Lop and hang down in a horseshoe shape from the crown approximately 1 ½ inches or more below the muzzle

ØFlemish Giant • Originated in Belgium • This is the largest of all the domestic rabbit breeds

q Types of Rabbit Production Ø Fanciers – Pets, Breeding Stock, and Show Animals Ø Laboratory Ø Fur/Hair – Angora – Rex Ø MEAT – Largest Rabbit Market

Rabbits • Pets – With diversity in size they can fit into different home situations • Trainable to use litter box • Require little special care • Clean, gentle, and lovable pets

Rabbits • Fur/wool-divided into four types 1. Normal • regular rabbit 2. Rex • short 3. Angora • like wool 4. Satin • mutation fur • smaller in diameter • transparent outer shell

3 Most Popular Meat Breeds • New Zealand (white)

• Californian

• Florida White
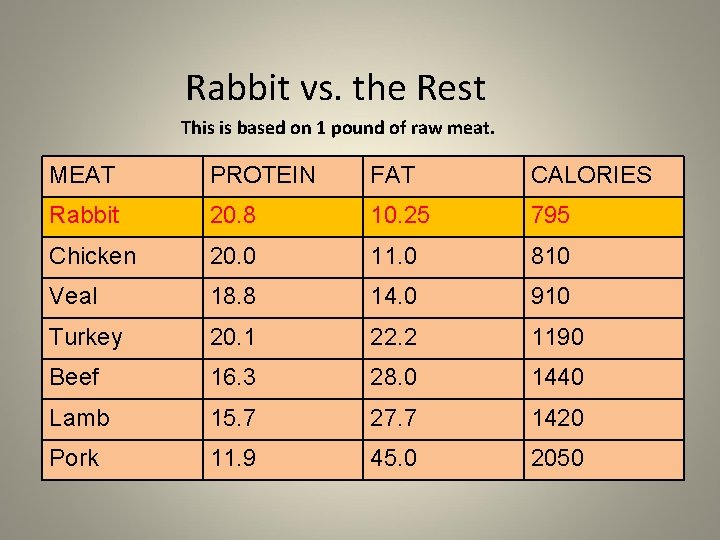
Rabbit vs. the Rest This is based on 1 pound of raw meat. MEAT PROTEIN FAT CALORIES Rabbit 20. 8 10. 25 795 Chicken 20. 0 11. 0 810 Veal 18. 8 14. 0 910 Turkey 20. 1 22. 2 1190 Beef 16. 3 28. 0 1440 Lamb 15. 7 27. 7 1420 Pork 11. 9 45. 0 2050

Examples of Small Commercial Netherland Dwarf Holland Lop Polish Mini Lop American Fuzzy Lop Mini Rex Dutch Florida White These breeds are small breeds with nice dispositions and are well suited for younger 4 -Hers.

Examples of Commercial Californian Satin Champagne D’Argent French Lop Cinnamon New Zealand Crème D’Argent Rex These breeds are suited for the older 4 -Her because these breeds have mature weights above 9 lbs.

Examples of Full Arch Breeds Belgian Hare Britannia Petite Checkered Giant English Spot Tan These breeds range in size from 2 lbs. to 14 lbs. and are suited for the more older, more experienced 4 -Her because of breed disposition and their high energy levels.

Examples of Semi Arch Breeds Flemish Giant English Lop Giant Chinchilla American Chinchilla These breeds are suited for the older 4 -Her because of size-mature weight above 9 lbs, but can be used by the older novice showman because of such easy going dispositions.

Cylindrical Breed Himilayan The Himilayan is the only breed with cylindrical body type. They are small with weights about 3 -4 pounds and are extremely docile. This breed is well suited for young and novice 4 -Her.

q. European Rabbit ØOf the six genus in the Subfamily Leporinae, only the European rabbit, Oryctolagus cuniculus is commonly used in research, testing, or teaching. Ø Three of the most commonly used breeds are the New Zealand White, American Dutch, and Californian.

v. American Dutch The American Dutch rabbit, shown here, is less than half the size of the New Zealand White, reaching a mature weight of only two kilograms. There are several color varieties of the American Dutch breed, but the most common is the black and white.

v. Californian Another breed occasionally used is the Californian rabbit. It is only slightly smaller than the New Zealand White, reaching a mature weight of 4. 5 kg.

v. New Zealand White The New Zealand White is an albino rabbit. It reaches a mature weight of about five kilograms, and it is the most readily available breed in the United States.

Rabbit Anatomy & physiology

anatomical and physiological characteristics of the rabbit that are important to their use in research, testing, or teaching; and recognize deviations from normal parameters.

q Teeth The dental formula of rabbit is: • I 2/1 , C 0/0 , PM 3/2 , M 2 -3/3 x 2 = 26 -28 • As in rodent incisors grow continuously • Color : white • Malocclusion
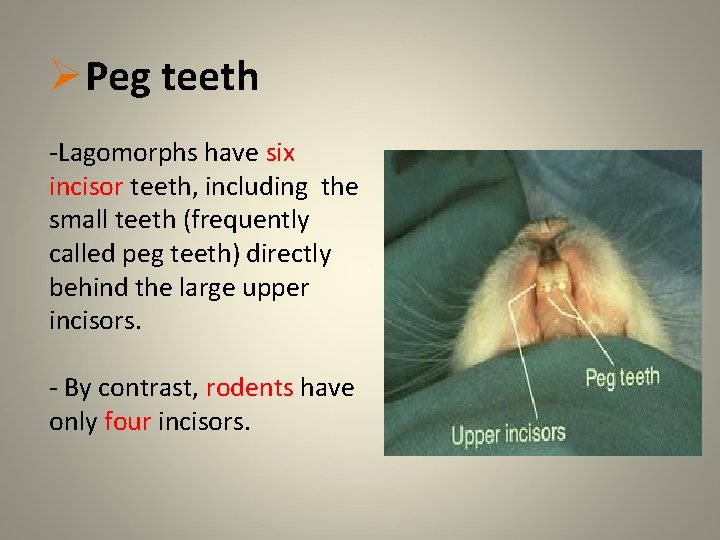
ØPeg teeth -Lagomorphs have six incisor teeth, including the small teeth (frequently called peg teeth) directly behind the large upper incisors. - By contrast, rodents have only four incisors.
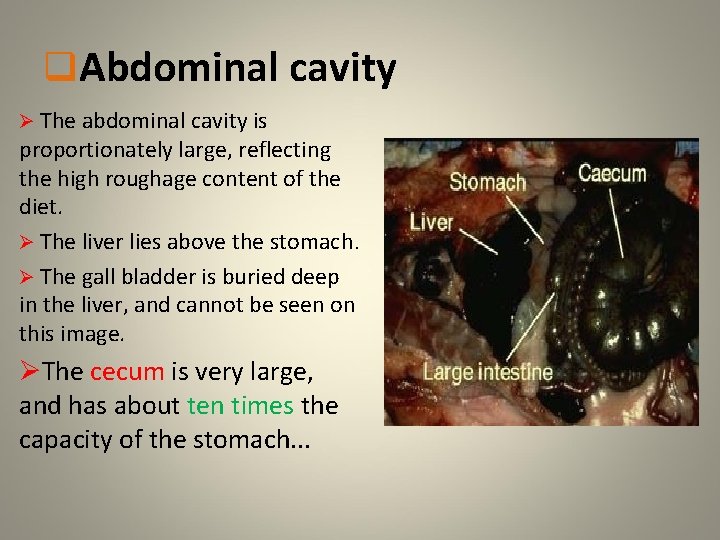
q. Abdominal cavity Ø The abdominal cavity is proportionately large, reflecting the high roughage content of the diet. Ø The liver lies above the stomach. Ø The gall bladder is buried deep in the liver, and cannot be seen on this image. ØThe cecum is very large, and has about ten times the capacity of the stomach. . .

q. Caecum • A rabbit's caecum is large, about twice the length of the abdominal cavity and 10 times the capacity of the stomach, 40% of the entire alimentary tract. It is folded into four parts that flex upon each other. It is thin-walled. Visible on the external surface is a spiral constriction that runs 20 -30 times around the caecum.
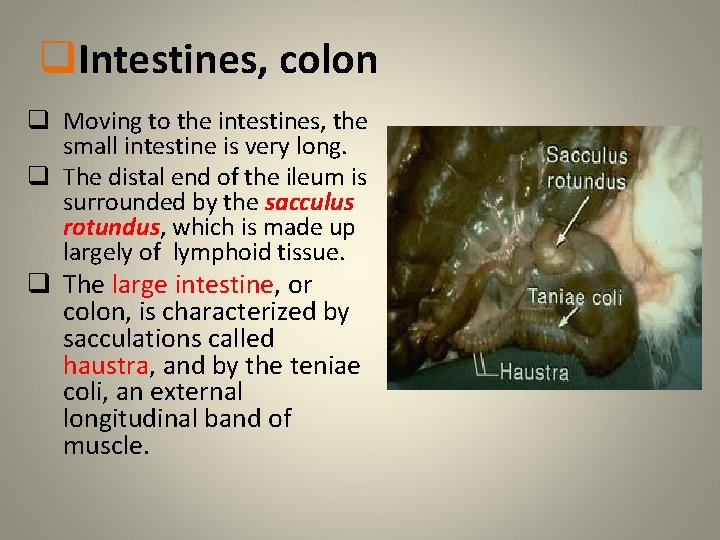
q. Intestines, colon q Moving to the intestines, the small intestine is very long. q The distal end of the ileum is surrounded by the sacculus rotundus, which is made up largely of lymphoid tissue. q The large intestine, or colon, is characterized by sacculations called haustra, and by the teniae coli, an external longitudinal band of muscle.
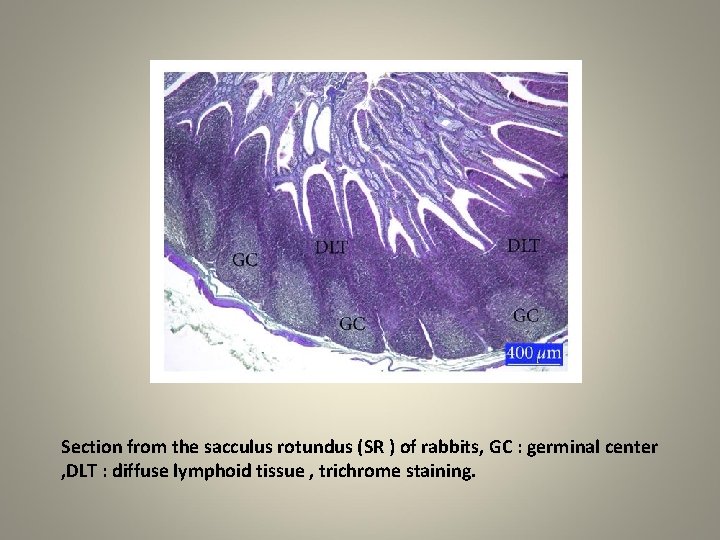
Section from the sacculus rotundus (SR ) of rabbits, GC : germinal center , DLT : diffuse lymphoid tissue , trichrome staining.

qappendix The cecum terminates in the lymphoid appendix, a narrow, thick-walled vermiform tube.

q Rabbit feces : ØHealthy rabbits produce two types of feces. Ø The droppings seen in the pan under the cage are hard fecal pellets that contain mostly waste fiber. ØThe other type is a softer feces that is produced in the cecum, and is often called cecotrophs or night feces. which is very soft & covered w/ thick mucus. ØThis material is rich in vitamins and protein, and the rabbit may consume it directly from the anus. Ø eating stool = coprophagy
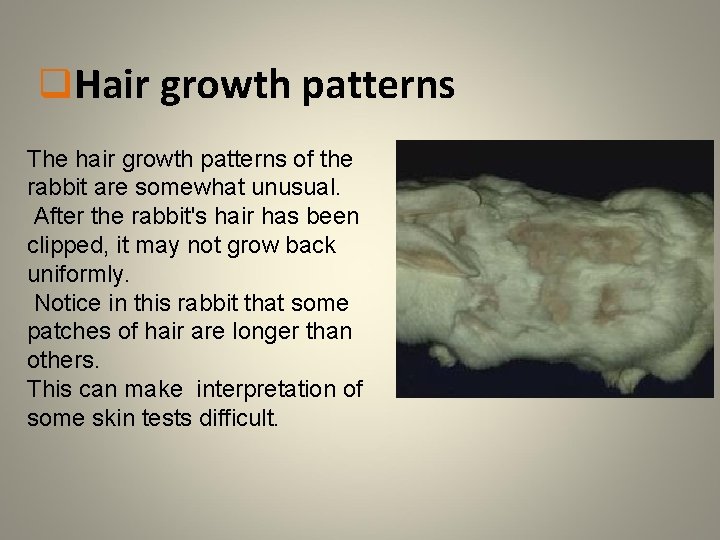
q. Hair growth patterns The hair growth patterns of the rabbit are somewhat unusual. After the rabbit's hair has been clipped, it may not grow back uniformly. Notice in this rabbit that some patches of hair are longer than others. This can make interpretation of some skin tests difficult.
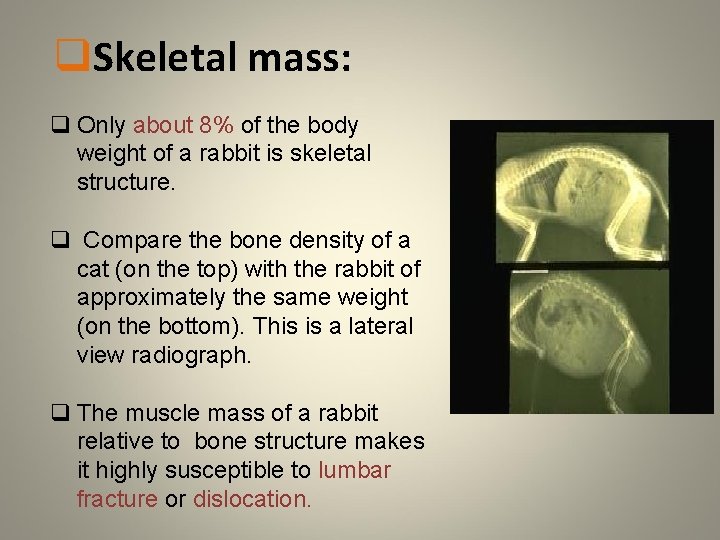
q. Skeletal mass: q Only about 8% of the body weight of a rabbit is skeletal structure. q Compare the bone density of a cat (on the top) with the rabbit of approximately the same weight (on the bottom). This is a lateral view radiograph. q The muscle mass of a rabbit relative to bone structure makes it highly susceptible to lumbar fracture or dislocation.

q urine Ø A healthy rabbit will produce about 130 milliliters of urine per kilogram of body weight each day. Ø It will normally be dark yellow to brown and will appear turbid. Ø This turbidity is due to mineral precipitates. Ø Rabbit urine is normally alkaline with a p. H of over 8. 0.

q. Calcium Metabolism • Rabbits have unique calcium metabolism. Calcium is absorbed across the intestinal wall in the absence of vitamin D, which other mammals require for calcium absorption. Less -regulated calcium absorption results in calcium being excreted in the urine. Parathyroid hormone and calcitonin regulate blood levels. Kidney stones and calcification of soft tissue may occur if a high calcium diet is fed to rabbits for a prolonged period of time.

q Atropinesterase Ø A notable feature of many rabbits is that their serum contains an enzyme, atropinesterase, that deactivates atropine and related drugs. Ø The amount of this enzyme a rabbit has is a heritable characteristic, and there are in vivo and in vitro techniques to screen for it. The presence of atropinesterase will make a dramatic difference in how individual rabbits respond to premedication with atropine prior to surgery.

q Rabbit Hematology Ø Ø Ø Ø The normal range for the red blood cell count is 4. 5 - 7. 0 million/μl. The total white blood cell count and differential fall within the range expected of most other species of laboratory animals, as shown here: Total WBC 4. 0 - 13 million/μl Lymphocytes 30 - 50% Neutrophils 30 - 50% Monocytes 2 - 16% Eosinophils 0. 5 - 5% Basophils 2 - 8% Anisocytosis, polychromasia, small numbers of reticulocyte(up to 4% ), and hawell – jolly bodies can be normal findings in rabbit blood films. the erythrocytes show marked anisocytosis , with up to 25% of erythrocytes being Microcytic in some animals.

q. Neutrophil, eosinophil The rabbit neutrophil, shown on the left, contains granules that stain red with eosin. Because of this staining characteristic, rabbit neutrophils are sometimes called pseudoeosinophils, amphophils, or heterophils. The eosinophil, shown on the right, is larger than the neutrophil. It has much larger granules that fill the cytoplasm. Normally, eosinophils do not exceed 5% of the total WBC count.

q. Physiological Data Ø Ø Ø Ø Body temperature: 38 -40°c Heart rate: 200 -330 beat/min Respiratory rate: 30 -60 beat/min Weight: adult, 2 -6 kg newborn: 30 -80 gm Water consumption: 200 -500 ml / day, or 50 - 100 ml / 1 kg body weight daily Food consumption: 100 -200 gm / day, or 50 gm / 1 kg body weight daily Life span: 5– 7 years

q. Physiological Data Mammary glands: 8 -10 ØGestation : 28 -36 days ( 31 -32 days ) ØLitter size: 4 -10 ØGrowth rate : 15 -20 gr/day to 8 weeks 100 -150 gr/day to 26 w ØWeaning age : 30 -35 days ØYoung-eyes open: 7 days ØYoung-star eating solid food: 3 weeks ØBlood volume: 45 -75 ml/kg(60 ) Ø

q. Physiological Data Ø poly estrous ØNon seasonal Ø induced ovulation ØOvulation after 10 -12 h

Breeding

q. Breeding Ø Buck polygamous – mates with many F Ø Buck may be bred up to 5 X a week. Ø Take doe to buck’s cage for breeding. ØDoe is territorial - fights buck in her cage. ØIn doe’s cage, buck may show > interest in territory marking than in copulation.

q. Breeding Ø Female does not have a true estrous cycle. v certain rhythmic periods during which they are receptive to bucks. v Doe does not ovulate until after mating. Ø = induced ovulation, a characteristic shared w/cat&ferret

q. Birth and Weaning Ø Need nest box to give birth in. Øwood shavings for nesting material ØDoe will pull fur from herself to line nest. Ø Babies (kits) born blind, naked & helpless. Ø Parturition (giving birth) = kindling

Nest Boxes

q. Birth and Weaning Ø Sexual maturity: 4 -6 months Ø Estrous cycle: No regular cycle, receptive to breeding at 4 -6 day intervals; induced ovulator Ø Gestation: 29 -35 days Ø litter size: 4 -10 Ø Weaning: 4 -6 wks Ø eat dry food at ~ 3 wks of age

q. Behavior Ø Active, curious, explore objects in environment. Ø Adept at escaping from unsecured cages.

q. Behavior Ø When stressed or frightened: Ø Ø Ø bite or jump at handler stomp hind feet emit a high-pitched scream Ø Sensitive to noise & may react violently. Ø Toenails grow rapidly.

q. Behavior Ø More active at twilight than during day. Ø Feed & drink water mostly in late afternoon & early evening, sleep during day.

q. Husbandry Ø Optimum temperature for most rabbits 18 -22°C. Ø Lower temps reduce shedding. Ø Shed large quantities of hair. Ø Frequent cleaning of filters necessary.

q. Husbandry Ø Spaces in cage floor must be small enough to prevent catching feet but large enough to allow fecal pellets to fall through. Ø Some cage racks are equipped with automatic flushing pans.

q. Husbandry Ø Wash cages at least every 2 wks. Ø Empty & clean cage pans 2 or 3 x / wk. Ø Pans are usually treated with an acid solution prior to washing to remove urine scale. Ø Urine contains large amounts of minerals which remain on the pan surface when urine dries.

q. Husbandry Ø Commonly develop overgrown incisor teeth.

q. Diet Ø Feed pelleted diets, from hopper-type feeders. Ø Tend to overeat if fed ad libitum, often fed only measured amounts of feed. prevents obesity, promotes health & reduces waste Ø 1 st sign of illness = loss of appetite.

q. Diet Ø Fasten water bottles & feeders securely. Ø If a rabbit stops eating, check the water supply. Ø Train to use automatic watering valves.

Use in research

ATTRIBUTES OF RABBITS AS RESEARCH ANIMALS q Justification: Considering our current socio-legal environment, it has become very important for an investigator to be able to justify use of a particular animal species; e. g. , the species selected must correspond with the research or teaching objectives and the research must produce meaningful outcomes that cannot be obtained by using another method or a lower species. The following are attributes of rabbits that may justify their selection.

q. Size Rabbits may be preferred over other common laboratory animals because of their size. They have sufficient blood volume to allow for large blood samples to be taken either singly or serially. Because of their size, they may be preferred over rodents for studies that require temperature monitoring or surgical procedures.

q Inbred strains When we think of inbred strains, rodents may come to mind first. But the rabbit is highly desirable for studies that require larger animal and also an inbred strain. It is probably the largest laboratory species in which inbred strains are commonly available, and is often chosen for genetic studies. Multiple litters : The rabbit is one of the largest laboratory species to produce multiple litters per year. A rabbit can produce 6 or more litters a year, with a typical litter size of 7 or 8. Availability: Rabbits are readily available and relatively inexpensive when compared with species of similar size. Also, specific-pathogen free (SPF) rabbits are available. This means that the breeder will supply animals that are free of Pasteurella and other infectious agents.
q handling ØRabbits are generally docile animals that are easy to handle. Ø They are, however, capable of inflicting serious injuries by scratching or kicking, and of seriously injuring themselves if they are frightened. Ø A rabbit should be handled only by persons trained in proper methods of handling and restraint.

q. Caging / care ØAnother attribute is that they are relatively easy to maintain and have no special requirements. ØAn adult rabbit requires a smaller cage area than cats or dogs, they produce less waste, and they have no special requirements such as exercise or psychological enrichment, making them easier to maintain than other larger species.

q Analogous systems Four other characteristics of rabbits make them desirable for specific research tasks. First, by analogous systems, we mean that some of the rabbit's organ systems are very similar to systems in man. The rabbit is often the model of choice to study immune responses for this reason.

q Embryonic susceptibility ØThe placental barrier of the doe is hemochorial, similar to the human placental barrier; and rabbit embryos are extremely susceptible to both embryotoxic and teratogenic agents. Ø Therefore, the rabbit is the model of choice for testing any product that might be used by pregnant women.
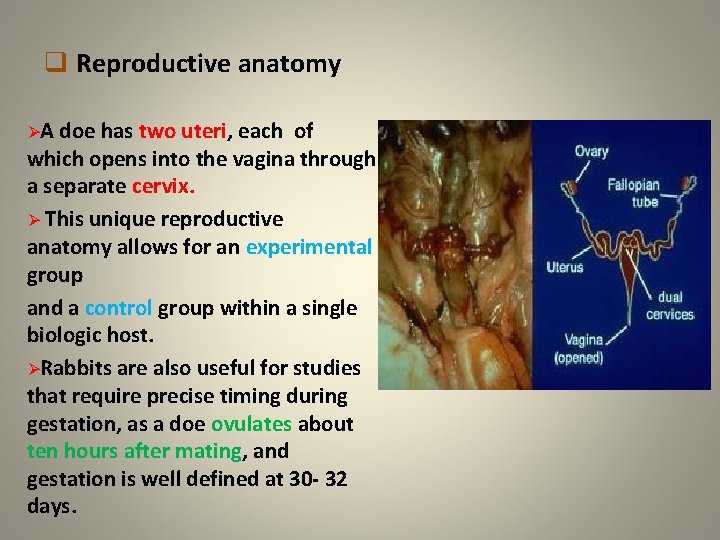
q Reproductive anatomy ØA doe has two uteri, each of which opens into the vagina through a separate cervix. Ø This unique reproductive anatomy allows for an experimental group and a control group within a single biologic host. ØRabbits are also useful for studies that require precise timing during gestation, as a doe ovulates about ten hours after mating, and gestation is well defined at 30 - 32 days.

q Susceptibility to disease The last of these characteristics that make the rabbit valuable for specialized studies is its susceptibility to spontaneous and induced diseases; such as : syphilis, herpetic conjunctivitis, tumors associated with papilloma viruses, glaucoma, hyperlipidemia, and nutritional muscular dystrophy.

q Historical uses Ø The cause and treatment of many human diseases have been studied using the rabbit as a model. Ø Because of its response to different diets, the rabbit was the first animal model used to study atherosclerosis. Ø Other noninfectious diseases studied in the rabbit include osteoarthritis, pregnancy toxemia, endometrial adenocarcinoma, drug teratogenesis, hydrocephalus, muscular dystrophy, glomerulonephritis, and gallstones. Ø Ophthalmology&vision ( Glaucoma, Cataract , … )

q Atherosclerosis Ø Atherosclerosis is induced typically in rabbits by feeding a commercial rabbit diet to which 0. 2 -2% cholesterol and sometimes up to 4 - 8% fat is added. Ø Two strains of rabbits with endogenous hypercholestrolemia and associated atherosclerosis , the watanabe heritable hyperlipidemic (WHHL) rabbit and the st. thomas’s hospital rabbit strain, have been described.

q Infectious diseases Ø Infectious diseases studied in the rabbit include: staphylococcal infection, bacterial endocarditis, and HIV. Ø Hereditary studies include familial hypercholesterolemia, dwarfism, spina bifida, and glaucoma.

q Syphilis - darkfield ØThere are some diseases of man for which only the rabbit can serve as a model. One of these is syphilis. ØThe rabbit is the only other mammal in which syphilis occurs naturally. ØThe causative agent of human syphilis, Treponema pallidium, cannot be grown in vitro.

§ Syphilis ØBut we can put the human organism into rabbits and study its growth there. Ø If and when a vaccine is developed for human syphilis, trials will undoubtedly be conducted using the rabbit first.

qpyrogeny test Ø Similar progress has been made in pyrogen testing. Ø Traditionally, rabbits have been to monitor parenteral drugs and solutions for the presence of contaminating endotoxins. Ø In 1976, Levine and Bang made a serendipitous observation that endotoxins caused horseshoe crab blood (amebocytes) to coagulate – an observation that led to the development and commercialization of the limulus amebocyte lysate assay (LAL).

q Pyrogen test ØThe LAL is now in wide use for detection of endotoxins in a variety of settings, although the FDA still views the rabbit pyrogen assay as the definitive assay for many parenteral compounds. ØThis may change, however, with refinement and supplementation of the LAL.

q antibody production v Serum and total blood v Polyclonal antibody: • Young , 2. 5 -3. 0 kg, 2 -3 animals per antigen. • Female: gentle, ( of many species ) are sensitive to lower doses of Ag and may have significantly higher and more prolonged responses to immunization than males. • 10 - 16 W • ID, SC, IM • At this age maternal Ig. G have declined to undetectable levels while the individual’s immune system is approaching near adult levels.

Rabbit Biomethodology

q. Rabbit Cage Technique • This is a typical rabbit caging rack used in research facilities today – all stainless steel construction is another popular system • The large cage area required • for the animal’s well-being also allows the handler easy access to the animal Litter pans are located beneath the animal and may be changed without disturbing the animal

q. Rabbit Cage Technique • Approach the animal with confidence, avoiding sudden movement and loud noises • Sudden movement or loud noises will startle the animal – they may thump their hind feet on the cage floor in protest • Open the cage door fully to allow full access to the animal • Slowly reach into the cage and place your hand on the rabbit’s neck just behind the ears

q. Rabbit Cage Technique Ø Grasp the animal firmly but gently by the nape of neck – DO NOT grasp by the ears Ø Place your other hand under the animal’s hind quarters to support its weight • Pull the rabbit from the • cage and rotate your hands slightly so that the animal’s hind quarters are beneath it next to your abdomen Your hand holding the nape of the neck should be directly over the hindquarters and against your chest

q. Rabbit Cage Technique Ø From this position, you can transfer the animal to another cage or restrain the animal for transport Ø To transfer to another cage simply place the animal in the cage and remove your hand from under its hind quarters while still holding the nape of the neck • Once the animals rear feet are on the cage floor, release the nape of the neck
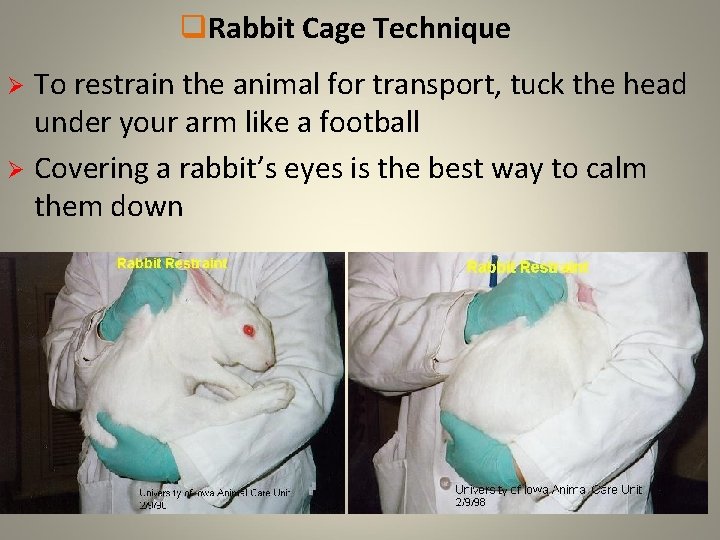
q. Rabbit Cage Technique To restrain the animal for transport, tuck the head under your arm like a football Ø Covering a rabbit’s eyes is the best way to calm them down Ø
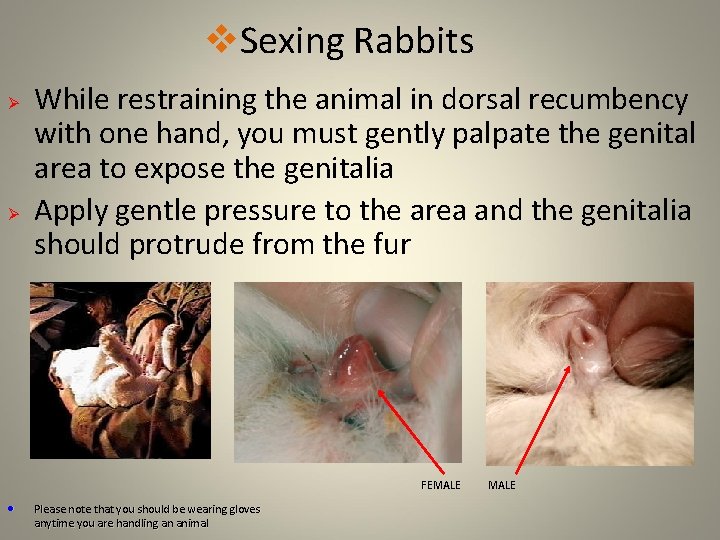
v. Sexing Rabbits Ø Ø While restraining the animal in dorsal recumbency with one hand, you must gently palpate the genital area to expose the genitalia Apply gentle pressure to the area and the genitalia should protrude from the fur FEMALE • Please note that you should be wearing gloves anytime you are handling an animal
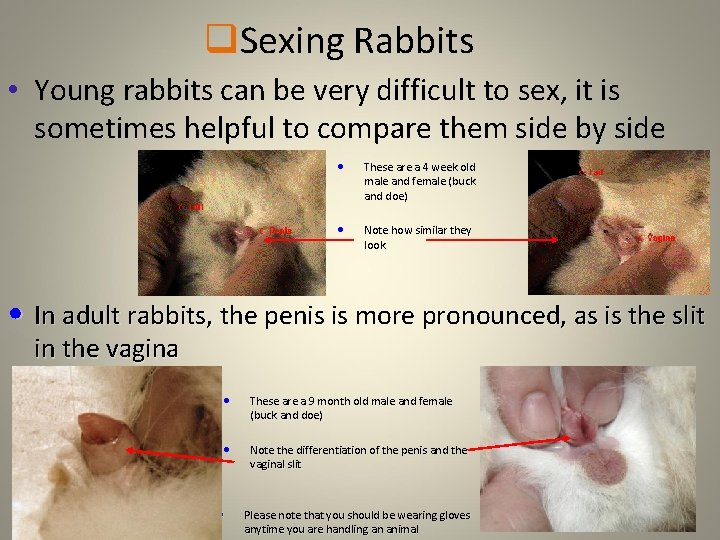
q. Sexing Rabbits • Young rabbits can be very difficult to sex, it is sometimes helpful to compare them side by side • These are a 4 week old male and female (buck and doe) • Note how similar they look • In adult rabbits, the penis is more pronounced, as is the slit in the vagina • These are a 9 month old male and female (buck and doe) • Note the differentiation of the penis and the vaginal slit • Please note that you should be wearing gloves anytime you are handling an animal

q. Restraint Ø When attempting to restrain rabbits, sudden, jerky moves should be avoided to decrease the likelihood of injury Ø Approaching with gentle confidence is best Ø Select the appropriate method of restraint for the procedure you wish to perform Ø If the rabbit begins to struggle violently, it should immediately be placed on a solid surface and calmed Ø Struggling frequently leads to fracture of lumbar vertebrae and injury to the spinal cord that may necessitate euthanasia

q. Manual Restraint Ø There is predominately ONE method of restraint in rabbits aside from mechanical or chemical Ø The rules are: 1) Support the animal’s weight 2) Cover the animal’s eyes 3) Restrain the hind legs • • Restraining the hind legs can be accomplished by holding the animal tight against your body or placing the animal on a hard surface and applying firm but gentle downward pressure to the hind end If the hind legs are not secured, the animal can kick and injure itself or the handler

q. Mechanical Restrainers Ø Several types of devices are commercially available to restrain rabbits Ø Quality devices prevent the animal from turning around or twisting yet allow easy access to the head and ears Ø Care should be taken when placing a rabbit in a restraint device since struggling may result in damage to the spine Ø Struggling is reduced if the device snugly secures both the head, back and hindlegs

Mechanical Restrainers ØBecause of the potential for injury to the animal and the skill required to correctly place the rabbit in a commercial restrainer, this procedure must be demonstrated live by a trained individual

q. Identification Methods Ø It is important to select the appropriate identification method for your research purposes Ø This should be based upon the age of the animal, the number of characters you wish to include, and the duration of the experiment Ø You should record the identification information on the cage card in the event that clarification of the numbers or characters becomes necessary for any reason

q. Identification Methods • Indelible markers can be used for short-term identification • Ear tattoos are the most common form of permanent identification of rabbits • There are 2 types of rabbit tattoo devices : 1) The Pen Type (electric/battery operated) 2) The Manual Type (pliers) • Microchip transponders can also be used 1 2
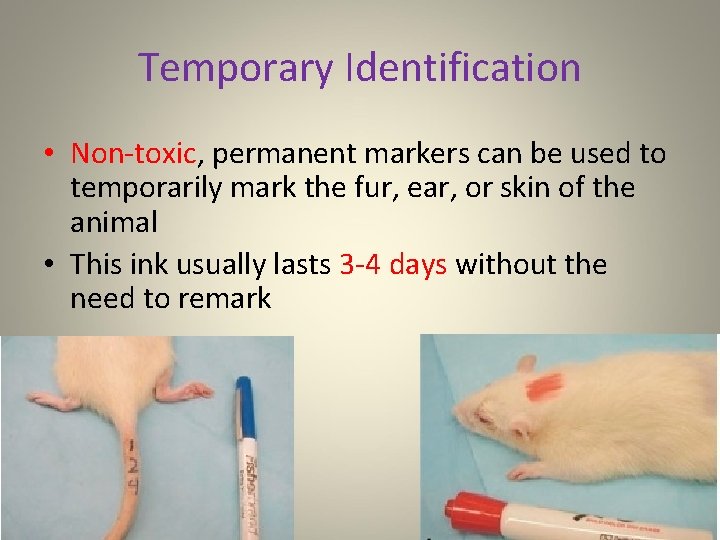
Temporary Identification • Non-toxic, permanent markers can be used to temporarily mark the fur, ear, or skin of the animal • This ink usually lasts 3 -4 days without the need to remark

Microchip Transponders • Often called “Pit Tagging”, they are implanted subcutaneously between the scapulae for permanent identification of individual animals • Each microchip is encrypted with a unique, non-replicable number and are read with a portable, hand-held scanner

Microchip Transponder Ø To implant these chips, the rabbit should be restrained in a mechanical restrainer Ø The hair is removed from the insertion site by shaving Ø The area is prepped with an iodophor, followed by alcohol Ø The implantation needle, with the syringe attached, is purchased in a sterile package Ø Make a tent from the loose skin at the implant site

Microchip Transponders • Insert the needle subcutaneously, with the bevel up, and depress the plunger Note that the bevel is facing up • Once the needle is removed the injection site should be observed for bleeding • If bleeding is noted, apply pressure with a sterile gauze pad, or a drop of super glue to the entry site

Tattooing • Tattooing should be used on animals over 6 wks of age as a permanent method of identification • Anesthesia is not required, but can be used to immobilize the animal • A tattoo device can be used to write numbers or other characters in the ears of rabbits • Tattooing can be done with either a pen type or manual type tattoo device

Tattooing • The use of tattoo equipment requires further hands -on training • All tattoo equipment should be disinfected prior to use and sterilized between animal rooms

q. Injections q Various routes exist for injecting rabbits. q Discuss the appropriate route, volume, site and needle selection with the veterinarian. q All injections must be described in your approved protocol. q All injections must be performed using sterile needles and syringes. q A new needle and syringe should be used for each rabbit.

Intramuscular Injections - IM • Regardless of the method used for IM injections, it must be noted that the sciatic nerve runs along the length of the femur • It is very important to avoid injuring this nerve • This is best accomplished by only injecting into the quadriceps muscle • It is imperative that the rabbit is properly restrained – this procedure requires hands-on training by ARF personnel • Swab the area with 70% ethanol before placing the needle and aspirate to look for blood before injecting
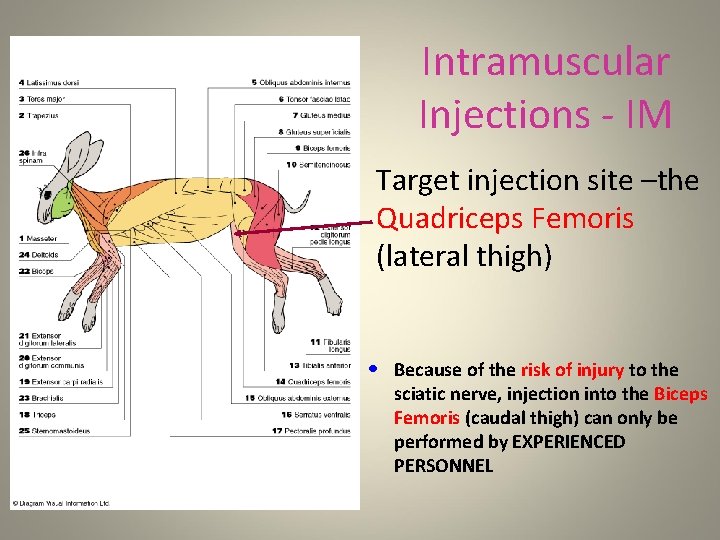
Intramuscular Injections - IM • Target injection site –the Quadriceps Femoris (lateral thigh) • Because of the risk of injury to the sciatic nerve, injection into the Biceps Femoris (caudal thigh) can only be performed by EXPERIENCED PERSONNEL
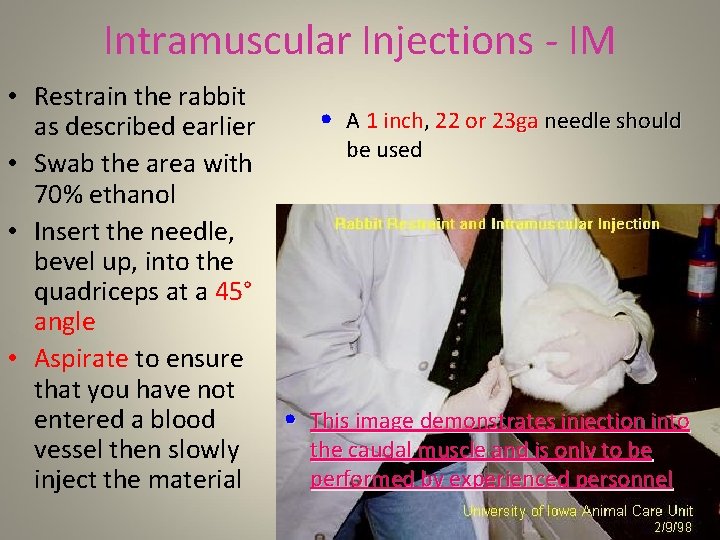
Intramuscular Injections - IM • Restrain the rabbit • A 1 inch, 22 or 23 ga needle should as described earlier be used • Swab the area with 70% ethanol • Insert the needle, bevel up, into the quadriceps at a 45° angle • Aspirate to ensure that you have not entered a blood • This image demonstrates injection into the caudal muscle and is only to be vessel then slowly performed by experienced personnel inject the material
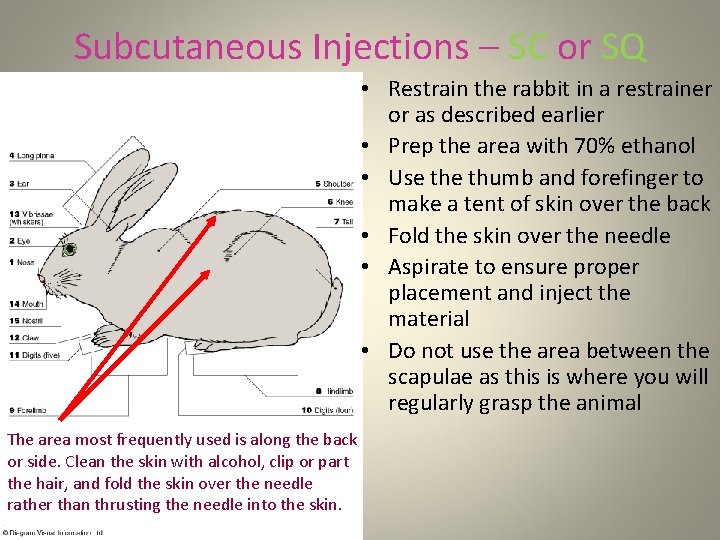
Subcutaneous Injections – SC or SQ • Restrain the rabbit in a restrainer or as described earlier • Prep the area with 70% ethanol • Use thumb and forefinger to make a tent of skin over the back • Fold the skin over the needle • Aspirate to ensure proper placement and inject the material • Do not use the area between the scapulae as this is where you will regularly grasp the animal The area most frequently used is along the back or side. Clean the skin with alcohol, clip or part the hair, and fold the skin over the needle rather than thrusting the needle into the skin.

Intraperitoneal Injections – IP Ø A state of "hypnosis" or tonic immobility can be induced in rabbits and is used for the IP injection Ø Proper hands-on training is required to learn IP injection in the rabbit
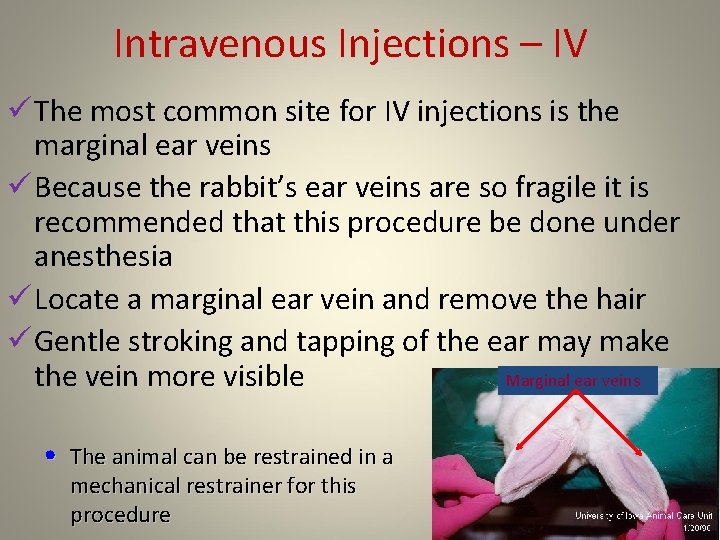
Intravenous Injections – IV ü The most common site for IV injections is the marginal ear veins ü Because the rabbit’s ear veins are so fragile it is recommended that this procedure be done under anesthesia ü Locate a marginal ear vein and remove the hair ü Gentle stroking and tapping of the ear may make the vein more visible Marginal ear veins • The animal can be restrained in a mechanical restrainer for this procedure
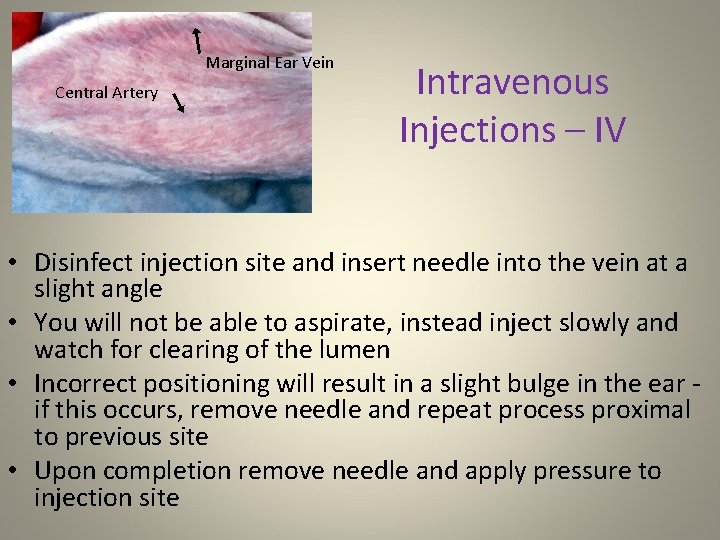
Marginal Ear Vein Central Artery Intravenous Injections – IV • Disinfect injection site and insert needle into the vein at a slight angle • You will not be able to aspirate, instead inject slowly and watch for clearing of the lumen • Incorrect positioning will result in a slight bulge in the ear - if this occurs, remove needle and repeat process proximal to previous site • Upon completion remove needle and apply pressure to injection site
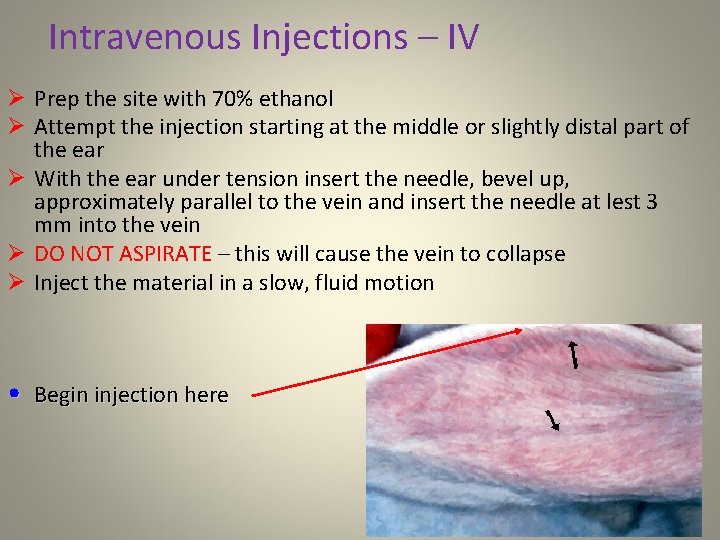
Intravenous Injections – IV Ø Prep the site with 70% ethanol Ø Attempt the injection starting at the middle or slightly distal part of the ear Ø With the ear under tension insert the needle, bevel up, approximately parallel to the vein and insert the needle at lest 3 mm into the vein Ø DO NOT ASPIRATE – this will cause the vein to collapse Ø Inject the material in a slow, fluid motion • Begin injection here

Intradermal Injections – ID Ø In order to perform ID injections the animal should be anesthetized Ø Shave an injection site on the back of the animal to remove the hair Ø Swab the site with 70% ethanol Ø Insert the needle into the skin, bevel up, holding the needle nearly parallel to the plane of the skin Ø Do not aspirate Ø Inject the material Ø A properly performed ID injection should result in a small, round skin welt
Injection Sites and Volumes Ø Ø Ø SQ – in the Back or sides – Maximum 30 -50 ml – 2023 ga needle IM – Lateral Thigh (quadriceps) – 0. 5 ml – 22 -23 ga needle IP – Lower Ventral Quadrants – 50 -100 ml – 19 -20 ga needle ID – Lateral Abdomen/Thorax – 0. 05 ml – 25 ga needle IV – Marginal Ear Vein – 5 ml – 20 -25 ga needle

Oral Gavage Ø Gavaging is used to dose an animal with a specified volume of material directly into its stomach Ø Only a specialized, commercially available gastric feeding tube should be used for this procedure Ø A mouth speculum used in concert with a rubber tube (4 mm diameter) can be used to administer compounds Ø Flexible or plastic tubes may be bitten or chewed and some care must be taken to prevent this - a tongue depressor with a centrally placed hole, when placed on end behind the diastema will serve as a satisfactory speculum Ø Proper hands-on training is required to learn gavage dosing in the rabbit Ø Please contact the ARF to receive this training
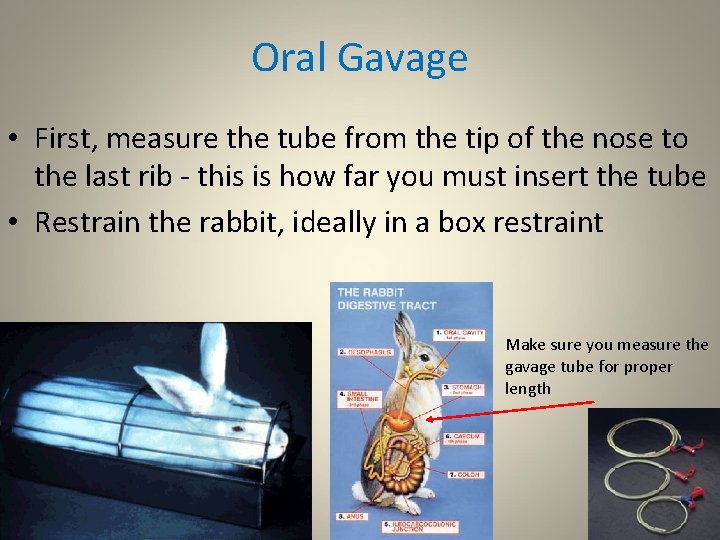
Oral Gavage • First, measure the tube from the tip of the nose to the last rib - this is how far you must insert the tube • Restrain the rabbit, ideally in a box restraint Make sure you measure the gavage tube for proper length

Oral Gavage • Lubricate the tube with sterile lubricant • In most cases, introduction of the tube toward the rear of the mouth will induce swallowing and the tube will readily enter the esophagus • At this time straighten out the tube and continue to pass • In the event resistance is met, DO NOT FORCE as this can cause serious injury • The most common place to meet restriction is in the back of the throat; the tube should pass freely when the rabbit swallows • A violent reaction (coughing, gasping) usually follows accidental introduction of the tube into the larynx or trachea

Oral Gavage Ø Check for proper insertion by injecting a few milliliters of water and aspirate for the presence of greenish-brown stomach contents Ø If correct placement is achieved, inject solution Ø Upon completion remove the tube by pinching the end of the tube and gently removing the tube Ø Pinching the end of the tube prevents contents in the tube from draining into the oral-pharyngeal region Ø When the procedure is complete, observe the rabbit for signs of distress such as gasping or frothing at the mouth

Blood Collection • It is important to select the proper method of blood collection that corresponds to the volume required for your research purposes • Some methods are intended for survival and others are not • Consult the veterinarian for more information Typical Blood Collection Sites Includes the Following: • Marginal Ear Vein – Survival • Central Ear Artery – Survival • Cardiocentesis (cardiac puncture) – Non-Survival
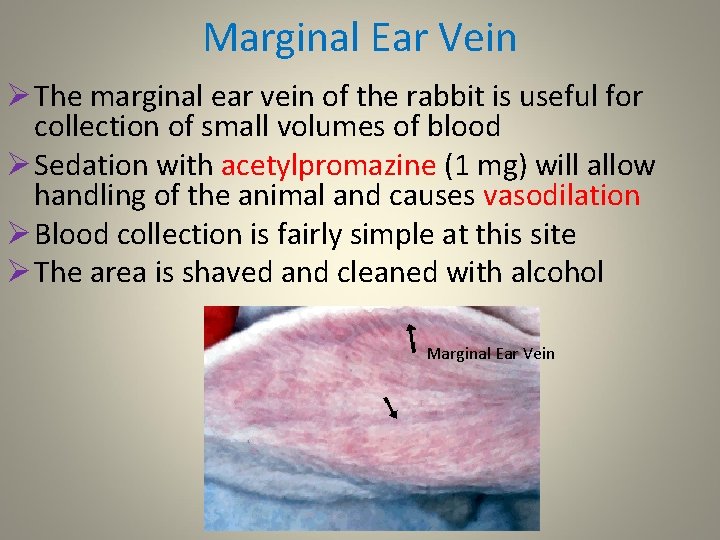
Marginal Ear Vein Ø The marginal ear vein of the rabbit is useful for collection of small volumes of blood Ø Sedation with acetylpromazine (1 mg) will allow handling of the animal and causes vasodilation Ø Blood collection is fairly simple at this site Ø The area is shaved and cleaned with alcohol Marginal Ear Vein
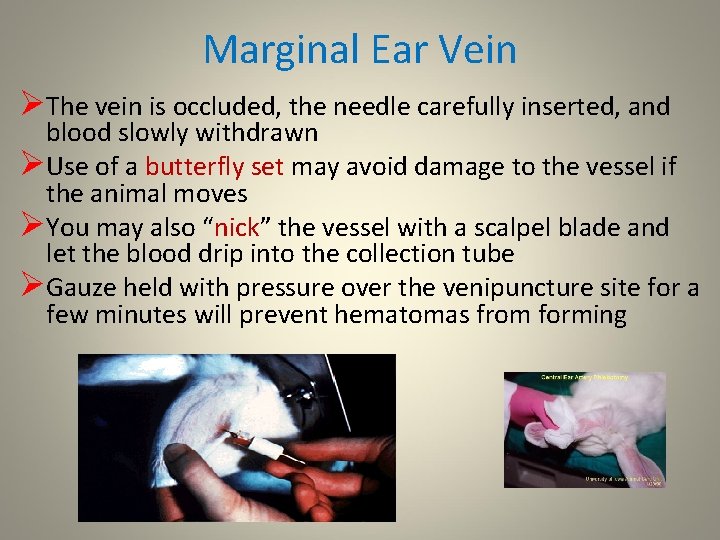
Marginal Ear Vein ØThe vein is occluded, the needle carefully inserted, and blood slowly withdrawn ØUse of a butterfly set may avoid damage to the vessel if the animal moves ØYou may also “nick” the vessel with a scalpel blade and let the blood drip into the collection tube ØGauze held with pressure over the venipuncture site for a few minutes will prevent hematomas from forming
Central Ear Artery q q The central ear artery can be used for collecting larger volumes of blood from the rabbit Phlebotomy from the central ear artery requires the use of a plain 20 Ga. one inch needle attached to a silicone-coated tube Alternatively, cutting the hub off a 20 gauge needle and collecting the flowing blood into a tube will decrease the time till coagulation of the needle Ten ml of blood/kg body weight can be collected in this manner but the rabbit must be carefully restrained and hematomas must be prevented by direct pressure
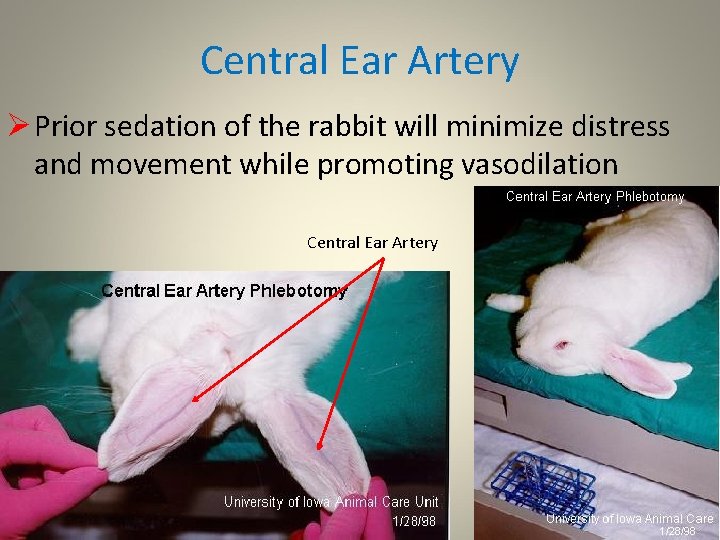
Central Ear Artery Ø Prior sedation of the rabbit will minimize distress and movement while promoting vasodilation Central Ear Artery
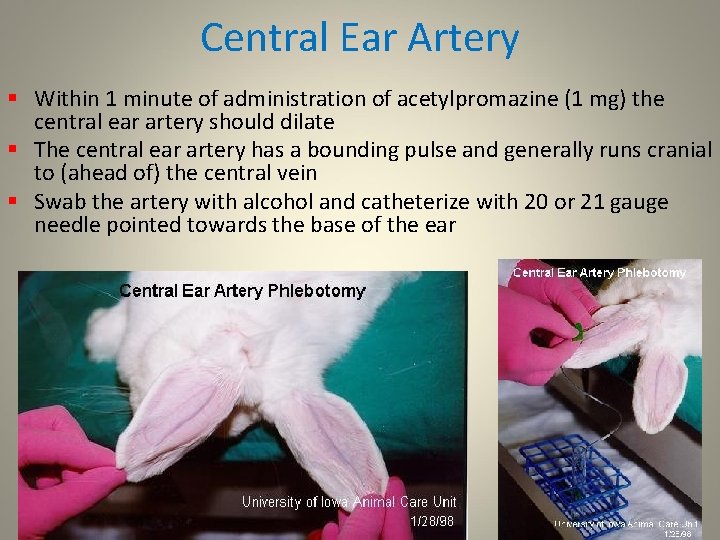
Central Ear Artery § Within 1 minute of administration of acetylpromazine (1 mg) the central ear artery should dilate § The central ear artery has a bounding pulse and generally runs cranial to (ahead of) the central vein § Swab the artery with alcohol and catheterize with 20 or 21 gauge needle pointed towards the base of the ear
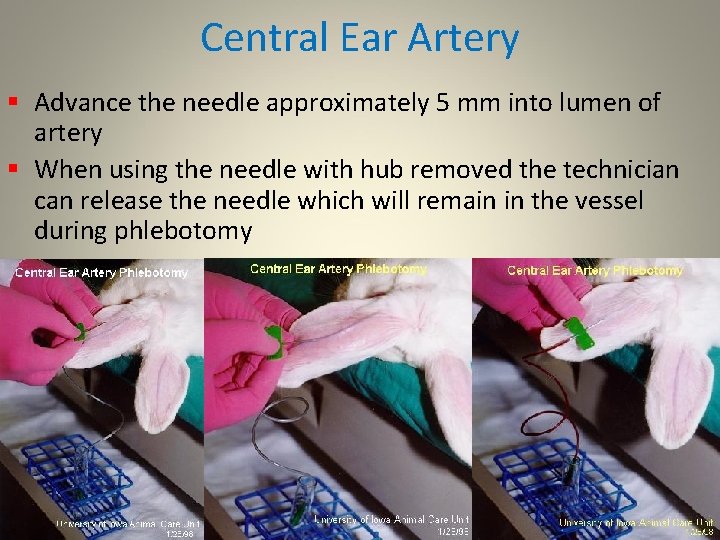
Central Ear Artery § Advance the needle approximately 5 mm into lumen of artery § When using the needle with hub removed the technician can release the needle which will remain in the vessel during phlebotomy
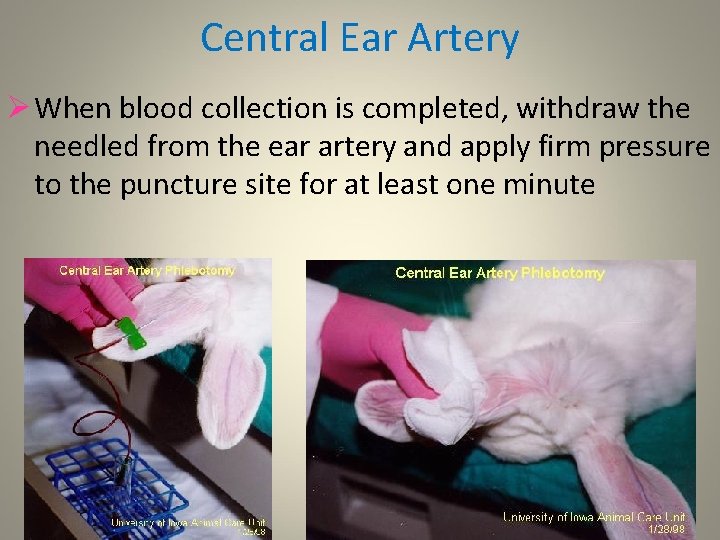
Central Ear Artery Ø When blood collection is completed, withdraw the needled from the ear artery and apply firm pressure to the puncture site for at least one minute

Cardiocentesis - Cardiac Puncture Ø This procedure must be performed under deep anesthesia and is a NON-SURVIVAL procedure Ø There are several methods for performing this procedure and all require hands-on training Ø Method 1: Syringe and needle – rabbit in dorsal recumbency Ø Method 2: Blood donor set and vacuum tubes Ø Method 3: Bleeding needle and vacuum tube – rabbit in lateral recumbency

Cardiac Puncture Syringe and needle with rabbit in dorsal recumbency is a very common method Ø Once the animal is anesthetized, prep the chest with 70% ethanol Ø Insert the needle at the base of the sternum, bevel up, into the thoracic cavity at a 15 -20° angle directed just to the left of midline • Aspirate slowly • If blood begins to flow into the syringe, Ø • continue to aspirate with steady, even pressure If no blood is seen reposition the needle and try again

Cardiac Puncture Once the required blood volume is collected, the rabbit is euthanized while still deeply anesthetized Ø Up to 120 milliliters or more of blood may be collected from an adult rabbit using this method Ø The problem with this method is removing the syringe to empty it into your collection tube – the needle may become dislodged from the heart Ø
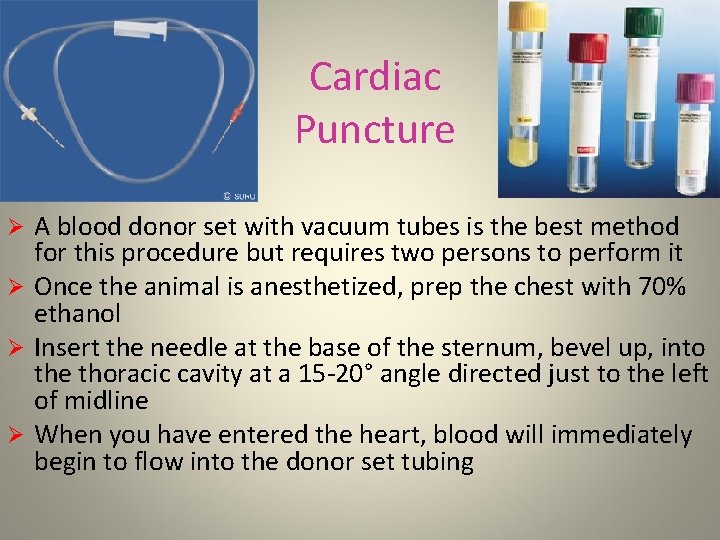
Cardiac Puncture A blood donor set with vacuum tubes is the best method for this procedure but requires two persons to perform it Ø Once the animal is anesthetized, prep the chest with 70% ethanol Ø Insert the needle at the base of the sternum, bevel up, into the thoracic cavity at a 15 -20° angle directed just to the left of midline Ø When you have entered the heart, blood will immediately begin to flow into the donor set tubing Ø
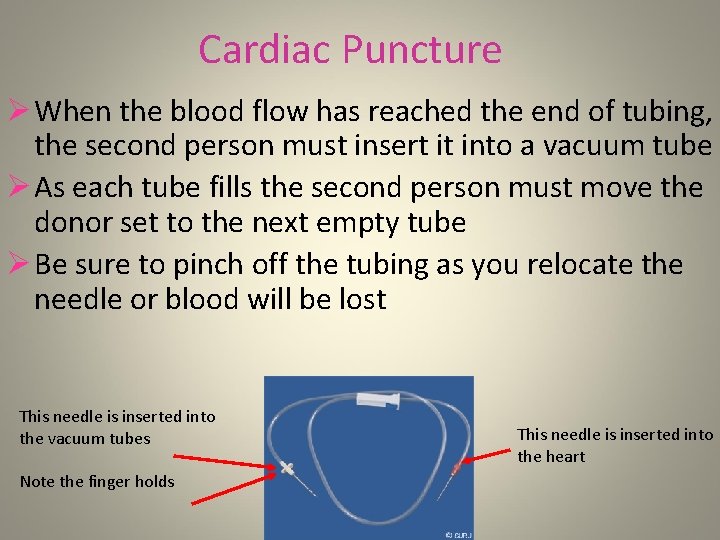
Cardiac Puncture Ø When the blood flow has reached the end of tubing, the second person must insert it into a vacuum tube Ø As each tube fills the second person must move the donor set to the next empty tube Ø Be sure to pinch off the tubing as you relocate the needle or blood will be lost This needle is inserted into the vacuum tubes Note the finger holds This needle is inserted into the heart
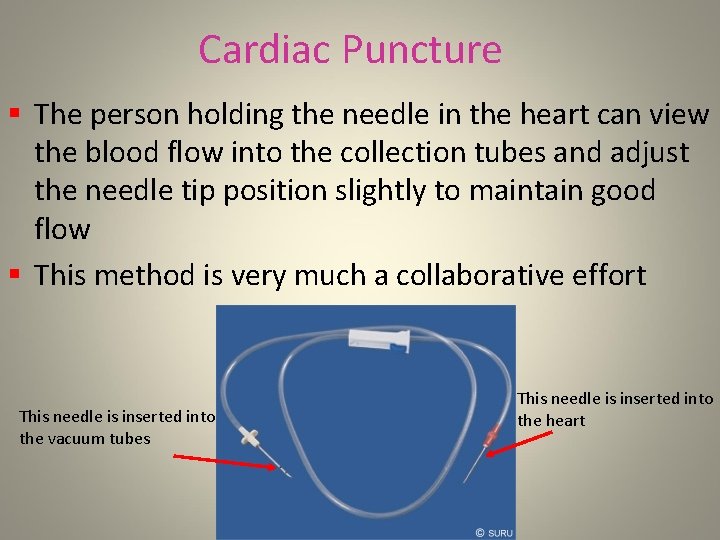
Cardiac Puncture § The person holding the needle in the heart can view the blood flow into the collection tubes and adjust the needle tip position slightly to maintain good flow § This method is very much a collaborative effort This needle is inserted into the vacuum tubes This needle is inserted into the heart

Cardiac Puncture ØOnce the required blood volume is collected, the rabbit is euthanized while still deeply anesthetized ØUp to 150 milliliters or more of blood may be collected from an adult rabbit using this method ØThe problem with this method is that it requires two persons

Cardiac Puncture Ø Bleeding needle and vacuum tube with rabbit in lateral recumbency is a less common, but equally effective method Ø Once the animal is anesthetized, the point of maximum intensity of heartbeat should be determine while palpating the laterally recumbent rabbit Ø Prep the side of the chest with 70% ethanol Ø An 18 gauge 1. 5 inch bleeding needle attached to a vacuum tube should be directed into the heart at the point of maximum intensity Ø Blood will flow into the needle when the heart chamber is entered

Cardiac Puncture § Once the required blood volume is collected, the rabbit is euthanized while still deeply anesthetized § Up to 100 milliliters or more of blood may be collected from an adult rabbit using this method § The problem with this method is changing collection tubes

Euthanasia Method Route/ technique Comments Pentobarbital overdose 150 mg/kg IV or IP CO 2 overdose 60 -70 % in an appropriate chamber Compressed co 2, in a Cylander Cervical dislocation Neck is hyperextended, separating first cervical vertebra Do NOT perform on unanesthetized rabbit , nor those weighing > 1 kg

THE END Good luck
- Slides: 154