Laboratory Operations and Techniques lare the proper ways

Laboratory Operations and Techniques lare the proper ways of manipulating/utilizing the apparatus in the laboratory.

l l l l l l A. Handling Liquids 1. transferring into a narrow-mouthed container (such as test tube, graduated cylinder, flasks) 2. transferring into a wide-mouthed container (such as beaker) B. Handling Solids/Powder 1. transferring into a narrow-mouthed container 2. transferring into a wide-mouthed container (beaker) C. Heating 1. using a test tube 2. using other containers (beaker, flask, etc. ) D. Determining the odor of a chemical E. Tasting chemicals F. Diluting an Acid G. Determining the acidity and alkalinity of a chemical H. Filtration I. Measuring 1. Volume of Liquid 2. Volume of Solid a. regularly-shaped ( with dimensions--- rectangle, square, cylinder) b. irregularly-shaped (without dimensions – marble, stones, etc) 3. Mass 4. Force 5. Temperature

A. Handling liquid 1. transferring into a narrowmouthed container (such as test tube, graduated cylinder, flasks) 2. transferring into a wide-mouthed container (such as beaker)

B. Handling Solids/Powder 1. transferring into a widemouthed container 2. transferring into a narrowmouthed container

C. Heating l Using a test tube l Using other containers (beaker, flask, etc. )

D. Measuring a liquid Measuring the Volume of Liquid – measure at an eye level

Measuring Solid 1. Measuring the volume a. Irregularly-shaped solid – use Water Displacement Method
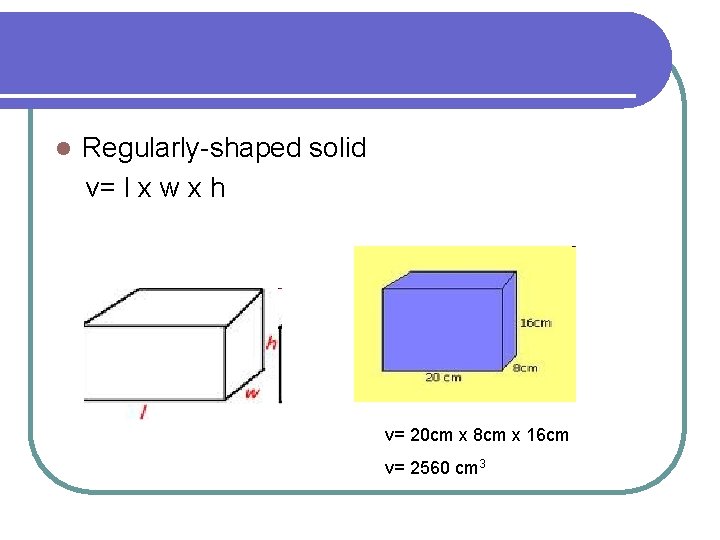
l Regularly-shaped solid v= l x w x h v= 20 cm x 8 cm x 16 cm v= 2560 cm 3
l Measuring the mass

l Measuring the Temperature

E. Filtration

F. Diluting Acid l Always add the acid to the water. Otherwise the acid can splash you and get on your skin and also if it is a concentrated acid, as you dilute it, a lot of heat is generated, and you want the larger volume of water to be able to absorb that heat.

G. Determining the acidity and alkalinity of a chemical Blue litmus paper turns red - acidic l Red litmus paper turns blue - basic l
- Slides: 13