Laboratory Medicine Grand Rounds Health Technology Assessment for

Laboratory Medicine Grand Rounds Health Technology Assessment for developers Janet Bouttell 15 November 2017 Health Economics and Health Technology Assessment (HEHTA)

What I am going to talk about • Who HEHTA are • What health economics is • NICE style health economics • vs what we are doing • Health economics and precision medicine • Current projects Health Economics and Health Technology Assessment (HEHTA) 1

Who we are and where we come from Health Economics and Health Technology Assessment (HEHTA) 2
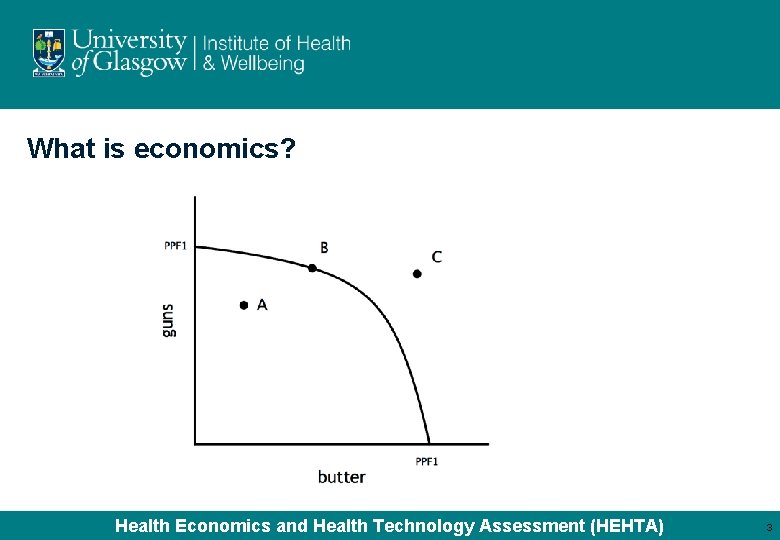
What is economics? Health Economics and Health Technology Assessment (HEHTA) 3
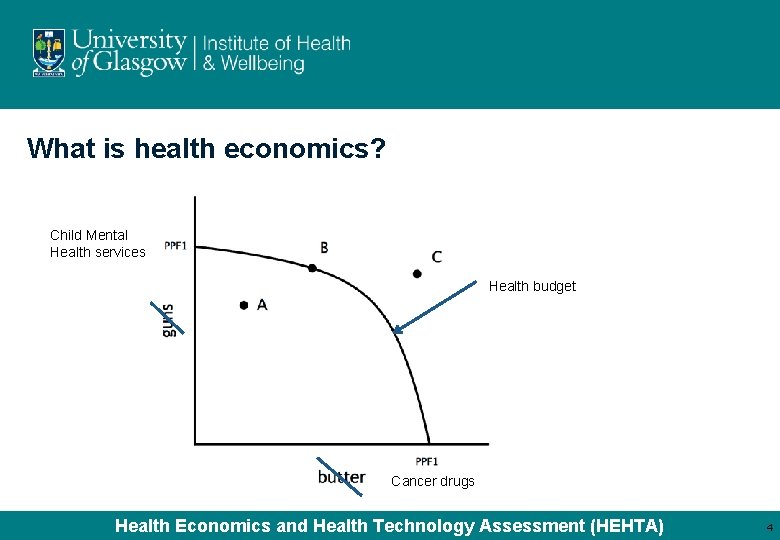
What is health economics? Child Mental Health services Health budget Cancer drugs Health Economics and Health Technology Assessment (HEHTA) 4
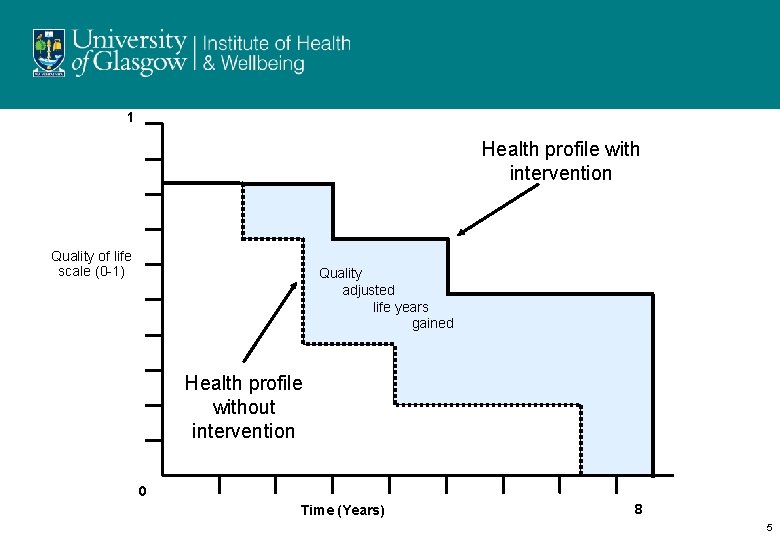
1 Health profile with intervention Quality of life scale (0 -1) Quality adjusted life years gained Health profile without intervention 0 Time (Years) 8 5

1. Define the ‘status quo’ in terms of expected cost and expected health outcomes (QALYs) 2. Specify how intervention could lead to different health outcomes (extension or improvement in Qo. L) 3. Translate clinical utility into QALY gains 4. Balance costs to give the expected value of the intervention 5. Incremental Cost Effectiveness Ratio (ICER) = cost/QALY gain 6

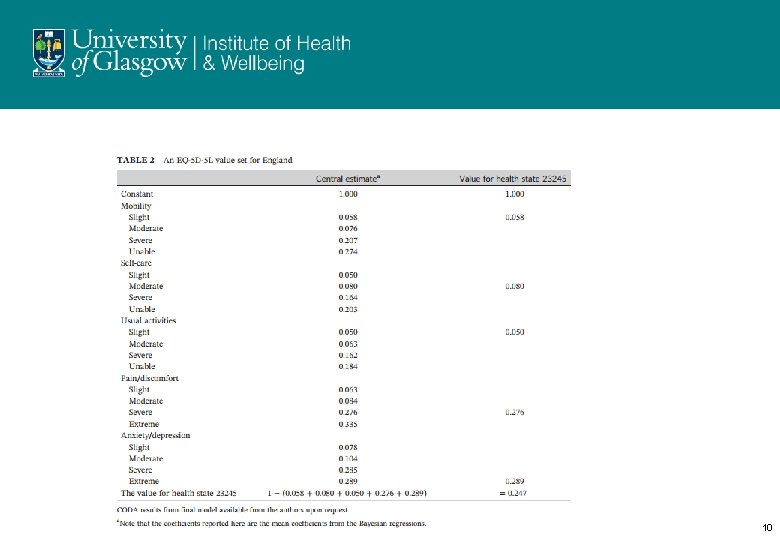
The QALY 1. Visual Analogue scale 2. EQ 5 D 3 L/5 L – Euro. Qol 5 dimensions, 3/5 levels (mobility, self-care, usual activities, pain/discomfort and anxiety/depression) - patients 3. Discrete Choice Experiments and Time Trade Off used to ‘value’ each health state – general population 7

Visual analogue scale (VAS) 8

9
10

NICE-style health economics? • Review some/all new medicines – value for money • NICE multiple technology appraisals compare existing drugs in one category (often as part of guideline development) • Mandatory/advisory • Devices (including diagnostics) different pathway but same idea Health Economics and Health Technology Assessment (HEHTA) 11
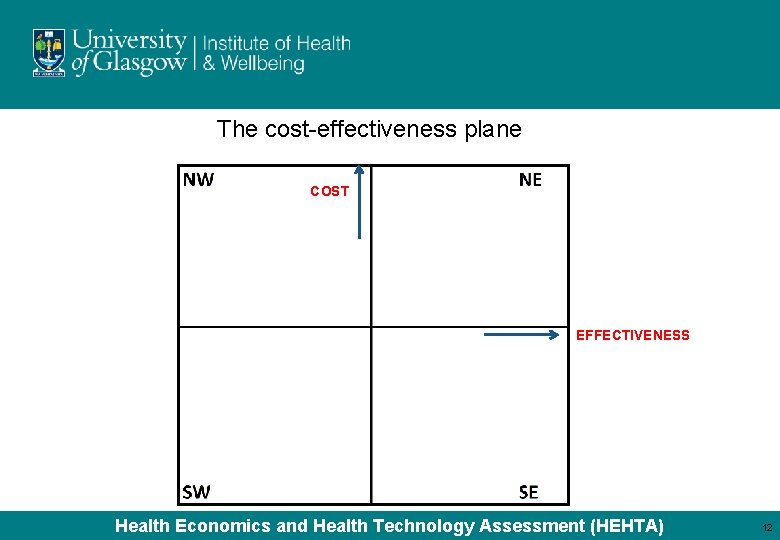
The cost-effectiveness plane COST EFFECTIVENESS Health Economics and Health Technology Assessment (HEHTA) 12
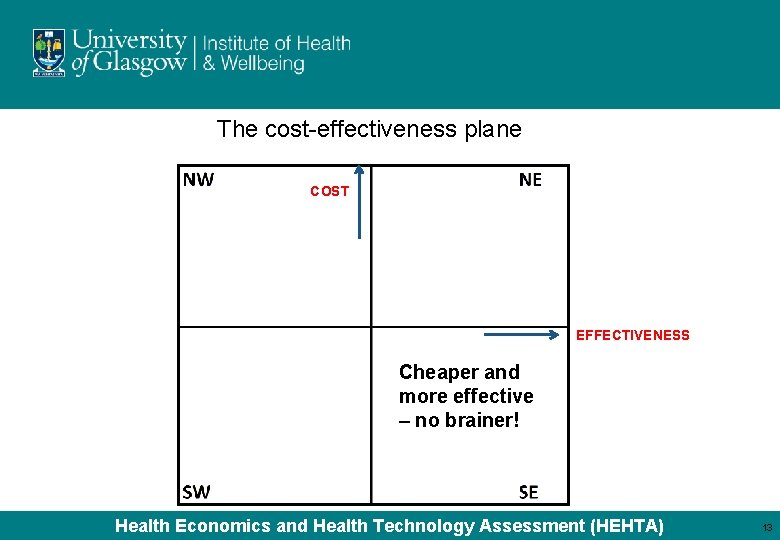
The cost-effectiveness plane COST EFFECTIVENESS Cheaper and more effective – no brainer! Health Economics and Health Technology Assessment (HEHTA) 13
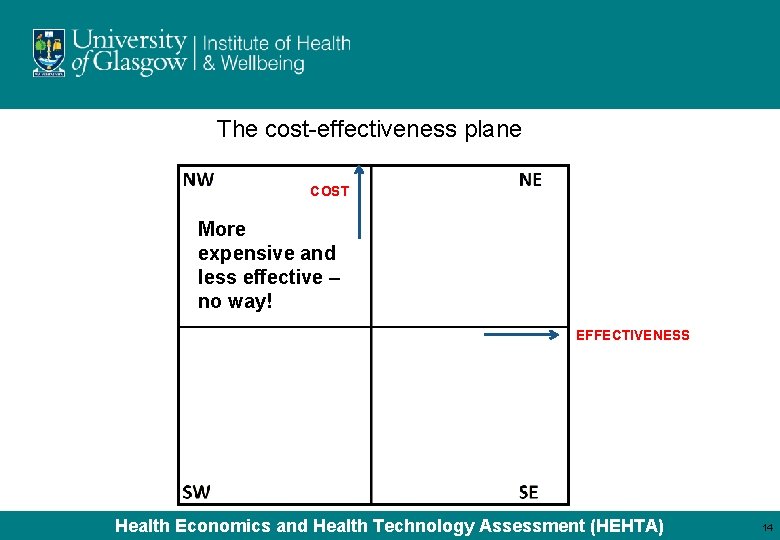
The cost-effectiveness plane COST More expensive and less effective – no way! EFFECTIVENESS Health Economics and Health Technology Assessment (HEHTA) 14
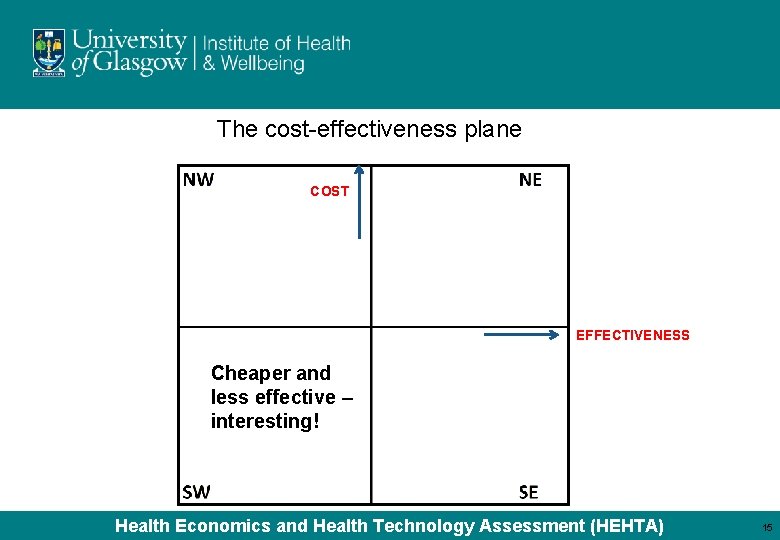
The cost-effectiveness plane COST EFFECTIVENESS Cheaper and less effective – interesting! Health Economics and Health Technology Assessment (HEHTA) 15
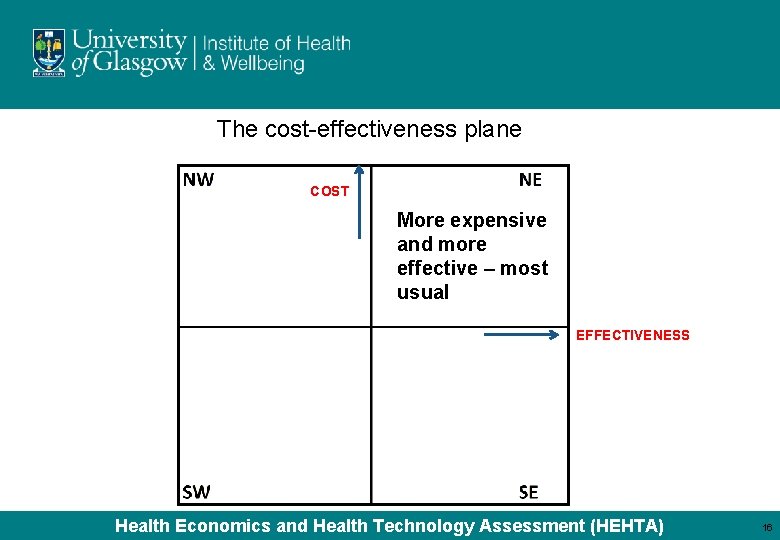
The cost-effectiveness plane COST More expensive and more effective – most usual EFFECTIVENESS Health Economics and Health Technology Assessment (HEHTA) 16
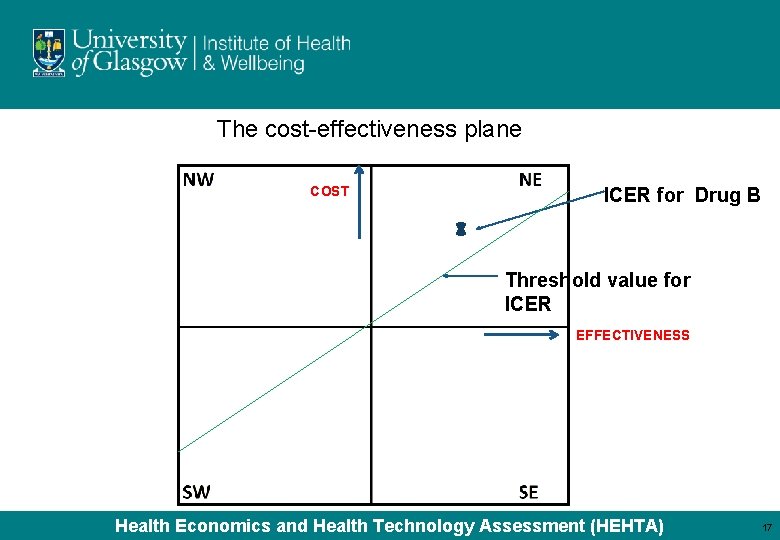
The cost-effectiveness plane COST ICER for Drug B Threshold value for ICER EFFECTIVENESS Health Economics and Health Technology Assessment (HEHTA) 17

NICE-style health economics (AKA HTA) Health Economics and Health Technology Assessment (HEHTA) 18

NICE-style health economics (AKA HTA) Source of model diagrams – Buisman et al. Arthritis Research & Therapy (2016) 18: 135 DOI 10. 1186/s 13075 -016 -1020 -3 Health Economics and Health Technology Assessment (HEHTA) 19
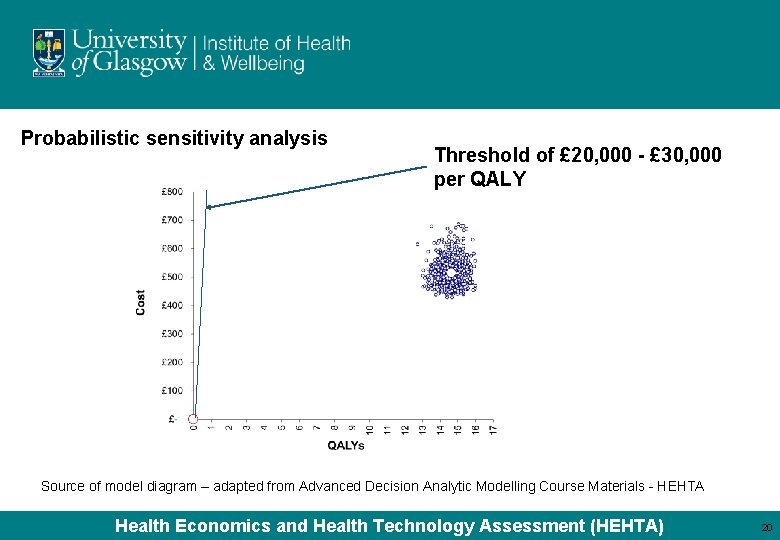
Probabilistic sensitivity analysis Threshold of £ 20, 000 - £ 30, 000 per QALY Source of model diagram – adapted from Advanced Decision Analytic Modelling Course Materials - HEHTA Health Economics and Health Technology Assessment (HEHTA) 20

Some major controversies in NICE style health technology assessment • The amount of the threshold • End of life/orphan disease provisions • The QALY – is it sensitive enough? • The QALY – does it capture all aspects of value? • The QALY - should it distinguish between ages/working status? 21

But NICE style health economics (or Health Technology Assessment) not really appropriate for us here because…. . Health Economics and Health Technology Assessment (HEHTA) 22
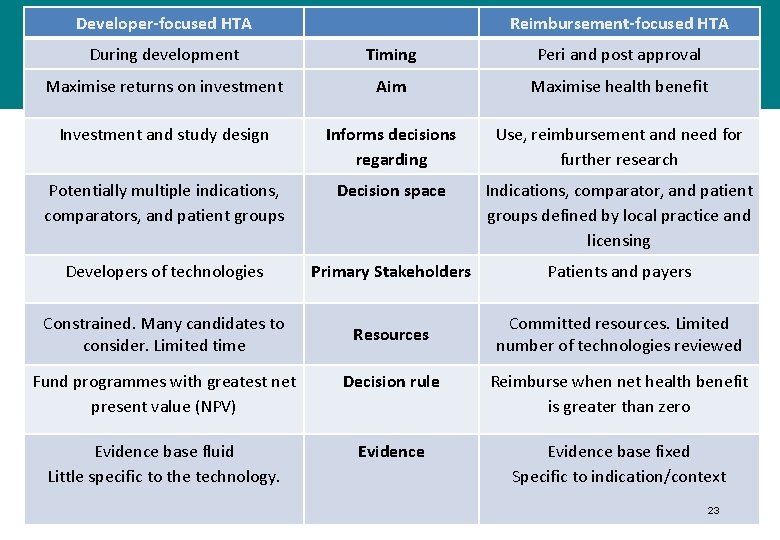
Developer-focused HTA Reimbursement-focused HTA During development Timing Peri and post approval Maximise returns on investment Aim Maximise health benefit Investment and study design Informs decisions regarding Use, reimbursement and need for further research Potentially multiple indications, comparators, and patient groups Decision space Indications, comparator, and patient groups defined by local practice and licensing Developers of technologies Primary Stakeholders Patients and payers Constrained. Many candidates to consider. Limited time Resources Committed resources. Limited number of technologies reviewed Fund programmes with greatest net present value (NPV) Decision rule Reimburse when net health benefit is greater than zero Evidence base fluid Little specific to the technology. Evidence base fixed Specific to indication/context 23

Steps for economic assessment in early stage of development process Health Economics and Health Technology Assessment (HEHTA) 24
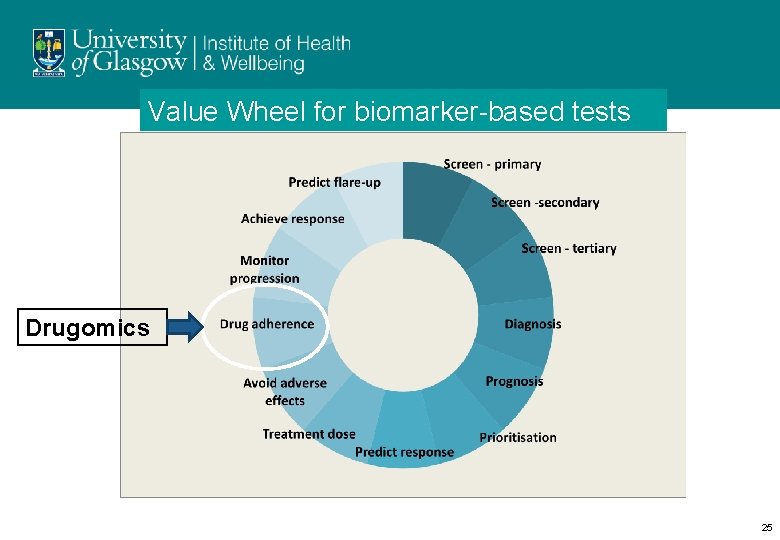
Value Wheel for biomarker-based tests Drugomics 25

Drugs don’t work in patients who don’t take them. — C. Everett Koop, M. D. [1] 26

Drugomics - Stage 1 of the early HTA framework: Understand disease and treatment pathway: • Chronic treatment for asymptomatic disease • Associated with high levels of non-adherence • Worse health outcomes and increased resource use 27

Drugomics - Stage 2 of the early HTA framework: Define the decision problem: • What is the value of a routine test of drug adherence in hypertension? • How would we demonstrate the value of the test? • Should we invest in development of the test? 28
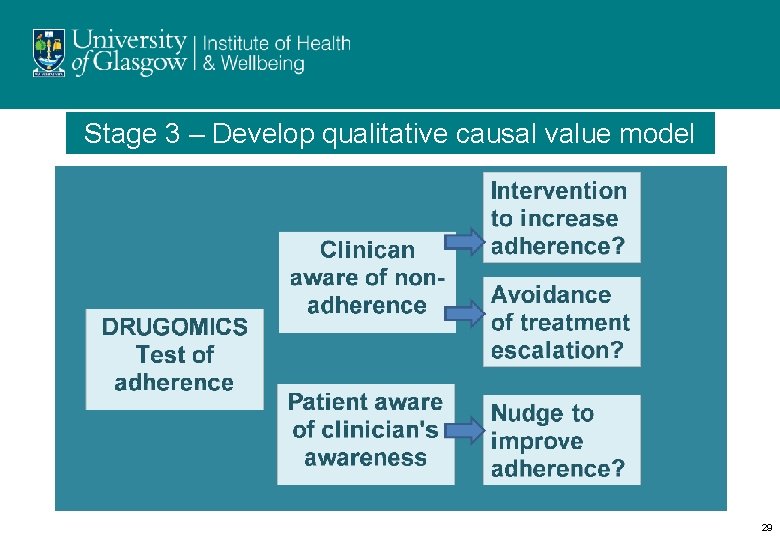
Stage 3 – Develop qualitative causal value model 29

Drugomics Early HTA value added – from qualitative discussion • Focus at on what the value proposition is and be able to communicate it in a simple way • Identification of potential barriers/evidence requirements at an early stage • Can identify disease context factors which will add value – e. g. treatment escalation 30
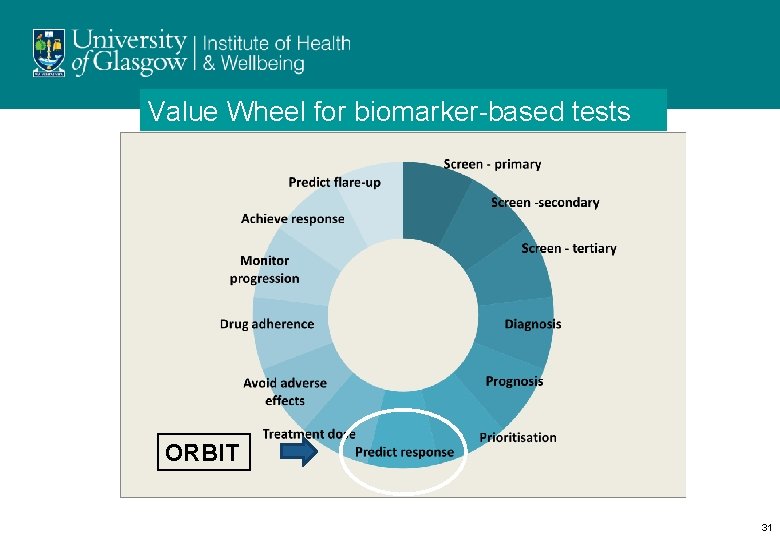
Value Wheel for biomarker-based tests Drugomics ORBIT 31
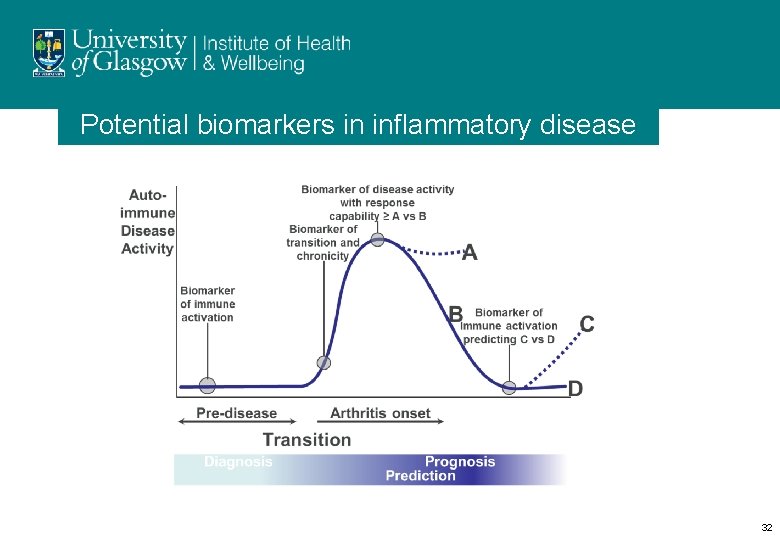
Potential biomarkers in inflammatory disease 32
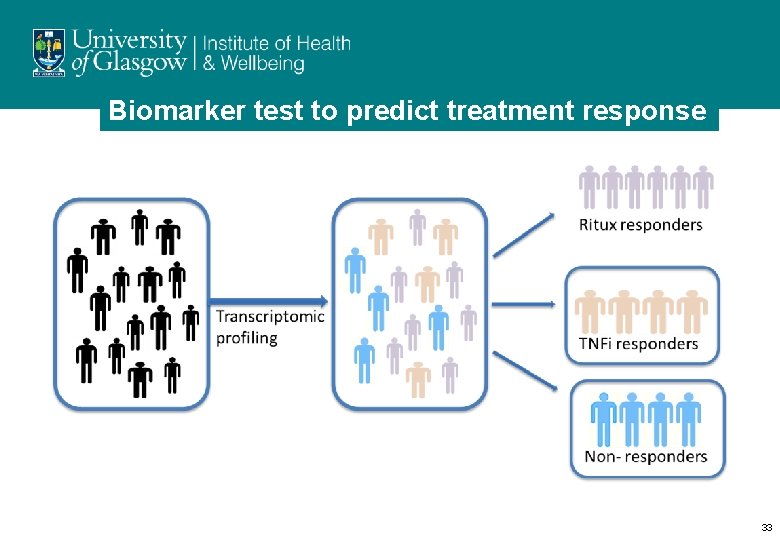
Biomarker test to predict treatment response 33
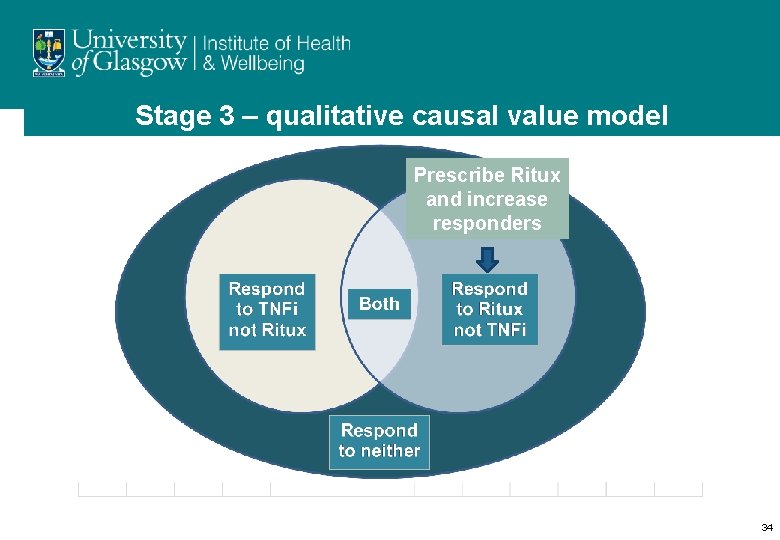
Stage 3 – qualitative causal value model Prescribe Ritux and increase responders 34
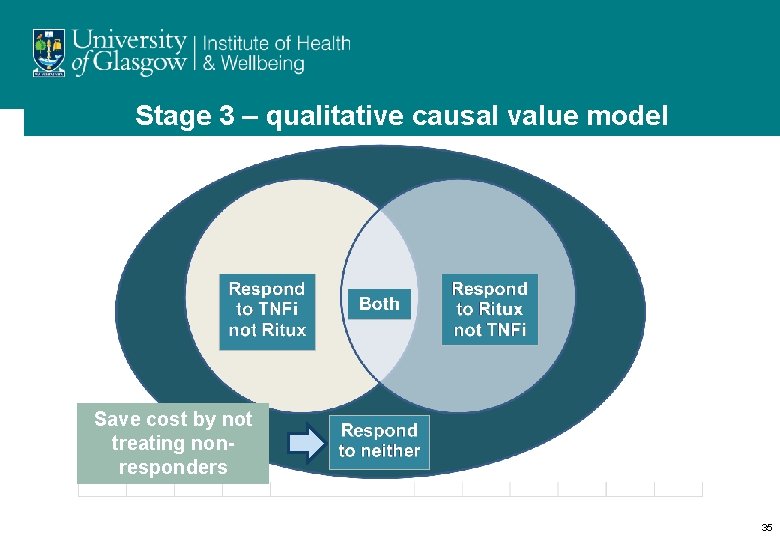
Stage 3 – qualitative causal value model Save cost by not treating nonresponders 35
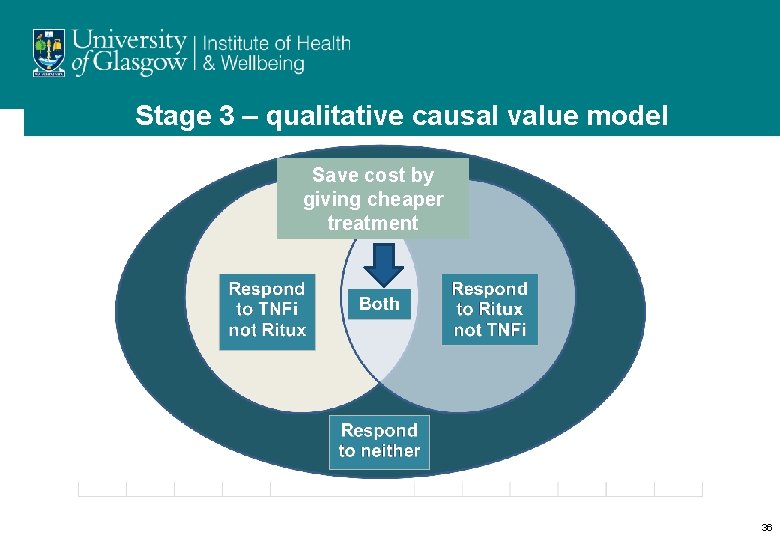
Stage 3 – qualitative causal value model Save cost by giving cheaper treatment 36
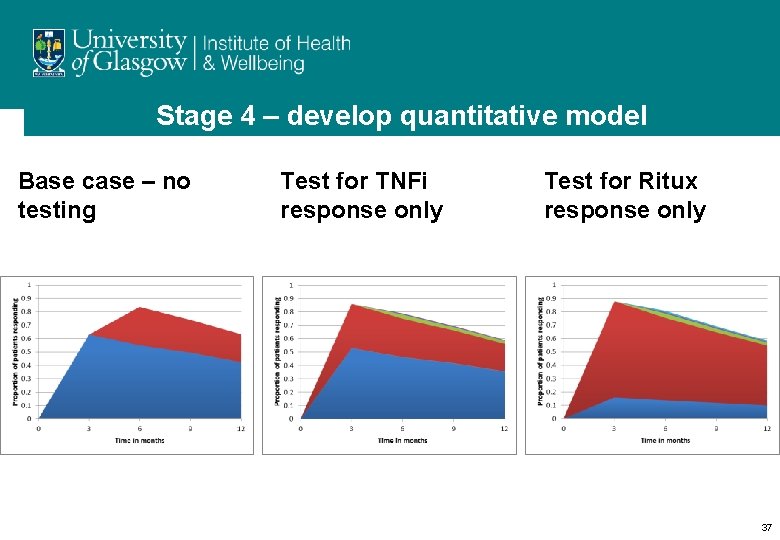
Stage 4 – develop quantitative model Base case – no testing Test for TNFi response only Test for Ritux response only 37

• • • Other discussions/projects Extension of KRAS/NRAS/BRAF testing in colorectal cancer Potential for hormonal treatment of ovarian cancer (‘what if’ analysis) Potential value of panel in Chronic Myeloid Leukemia Potential health economics input to trial of pancreatic cancer panel Cardiochip – point of care test for the simultaneous measurement of cardiac biomakers 38

References 1) Health Economics 2017: 1 -16, 23 -06 -2017 Valuing health‐related quality of life: An EQ‐ 5 D‐ 5 L value set for England Devlin N, Shah K, Feng Y, Mulhern B, van Hout B. 2) 1] Adherence to Medication Lars Osterberg, M. D. , and Terrence Blaschke, M. D. n engl j med 353; 5 www. nejm. org august 4, 2005 39

Project Summary • Project Summary Form to be completed and returned to Node Project Manager • Proposal forwarded to ISAB for review and comment electronically. Circulation ISAB review Feedback Recommendation • IP landscape • adoption impact • globalisation potential and impact • global regulatory and reimbursement environments. • Forms to be returned to Node Project Manager for collation within 1 week. • Recommendation to the Board – to include anonymised comments from the ISAB. 40
- Slides: 41