Laboratory Information Management Systems LIMS Lindy A Brigham

Laboratory Information Management Systems (LIMS) Lindy A. Brigham Div of Plant Pathology and Microbiology Department of Plant Sciences PLS 595 D Regulatory Issues in Laboratory Management February 2, 2006

LIMS • Definition: – A method a laboratory uses to manage its data and disperse the results to designated areas

Information Technology • Why? – Management of the Information Explosion – Quality Assurance • GLP, GMP, EPA, etc. – Reduction of data entry errors – Need for faster sample/results turnaround time

General Considerations • Different labs have different requirements • Procedures and outcomes differ • But all can use the systems approach to identifying their needs and implementing solutions

Types of Laboratories • Research • Analytical Services • Manufacturing

Research Lab • Many non-routine tests • Low sample volume • Flexibility between data and analyses performed • Specification checking (FDA reqs) • Traceability (calibration, distribution, data) • Change control procedures (documentation of changes eg SOPs, QA compliance)

Testing Services Laboratory • • • Tests performed at customer’s request High sample volume Routine tests Variable workload Prioritized samples

Manufacturing Laboratory • Perform testing for various materials including: – Final product testing – Stability testing – Work-in-progress testing – Environmental testing – Raw materials acceptance testing • Many tests per sample • Dynamic environment

LIMS Acquisition • How do you determine what you need and how to get it? • Systems Approach to defining needs and specifications for a LIMS

Acquisition Decisions • Buy or Build? All or Pieces? • Options – Design and build system ‘from scratch’ – Piece together existing and off the shelf components – Buy whole system from vendor • How do you decide?

By doing an Analysis • The build or buy what decision is made based on an analysis of the needs and practices of the lab • The analysis itself can be of benefit to any lab to: – Clarify existing practices – Help eliminate non-essential practices – Organize and streamline existing procedures

Systems Analysis and Design • Project Definition – What is to be achieved, by whom and why • Functional Requirements – all user entry requirements and system output requirements described in detail • Functional Design – detailed documentation to describe the system and detail how the functional requirements are to be achieved – independent of hardware and software requirements • Implementation Design – selection of hardware and software • System Integration – Gather all required components, interface components, install software, go live • System Evaluation – Final phase – Project definition and functional requirements are revisited and compared to the final installed system to determine how well the requirements were met

Analysis of UA Research Lab
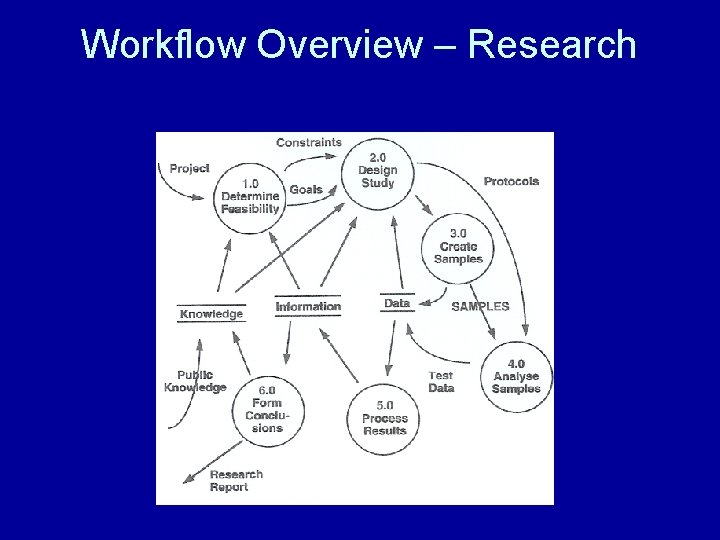
Workflow Overview – Research
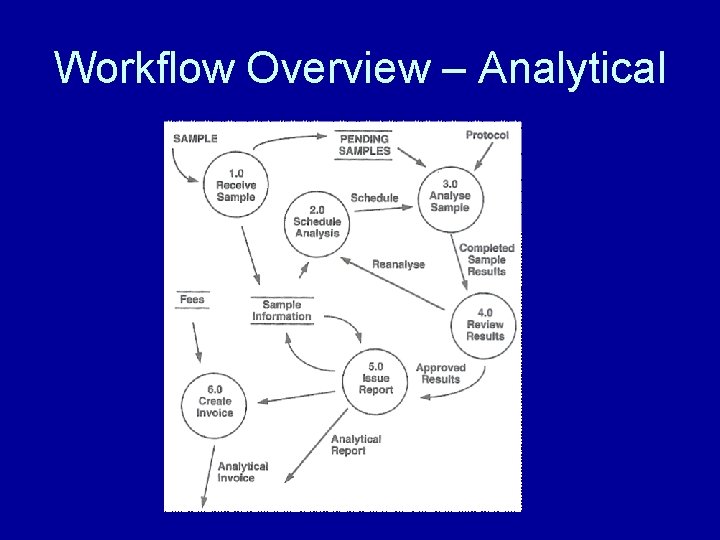
Workflow Overview – Analytical

Information Management System • Consists of: – Computer(s) – People – Procedures/Processes

LIMS Resources • Stand alone software – All purpose • Word Processing • Spreadsheets • Databases – Inventory – Document Management (Wolk) – Statistical Packages • Total systems – Lablynx – Starlims http: //www. lablynx. com/ http: //www. starlims. com/

Regulatory Requirements • ISO 9000 – Management must define, implement, communicate and maintain quality objectives and assign personnel at all levels of the organization to be responsible for verifying the company’s quality system – Primarily effect manufacturing laboratories • ISO 25 – Establishes labs technical competence • GALP (Good Automated Laboratory Practices) – Union of federal regulations, policies, and guidance documents establishing a uniform set of procedures to ensure the reliability and credibility of laboratory data (EPA) • 21 CFR 11 (1997) – Electronic signatures, Electronic records
- Slides: 18