Laboratory diagnosis of wounds abscesses plagues Cutaneous infections

Laboratory diagnosis of wounds, abscesses, plagues

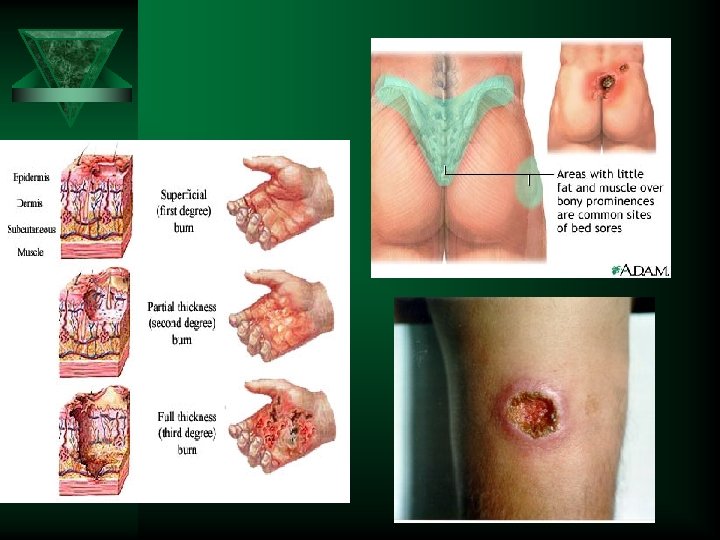
Cutaneous infections Ú Primary cutaneous infections – occur on apparently lesion free skin: – impetigo, – foliculitis, – furuncle Ú Secondary infections – occur on damaged skin: – traumatic lesion, – chronic dermatosis, – viral infection.

Purulent skin infections (PSI) Ú Class I PSI: deeper, closed Ú Class II PSI: – deep and closed, the focus communicates or has communicated with an organ containing commensal flora – deep and open, fistulas Ú Class III PSI: superficial, open with heavy contamination by commensal flora ; these sample have a high probability of containing both pathogenic and commensal flora.

Objectives Ú To identify the pathogenic bacteria in superficial primitive skin infections (anthrax, furuncle, erysipelas …) Ú To identify bacteria present in acute traumatic lesions (fractures, bites, burns, surgical wounds) Ú To identify bacteria causing secondary infections of surgical wounds Ú To identify the bacteria responsible for the signs of local chronic infections.

Physiopathology: Ú Puncture and pus aspiration from closed, undrained tissue doesn’t exclude the contamination during sampling. Ú Important is skin decontamination and drying of the skin before sampling. Any contaminant bacteria can be cause of false positive diagnosis. Ú Clinical significance of these isolates: cyto- bacterioscopic aspect of sample.

Physiopathology Ú Suppurations can be mono- or poly microbial. Ú Any suppuration is a step of a inflammatory process. Ú Inflammation evolve only in a live tissue; never in a necrotic tissue. Ú The essential problem of the pus examination is differentiation between bacteria with clinical significance and the specimen contamination.

Physiopathology: Ú The exudates and necrotic tissue from burns, bed sore, plagues are constantly contaminated with bacteria which usually are not involved in the inflammation’ s etiology. Important is to remove the stagnant tissue! Ú Rather: specimens should be aspirated from the primary infection through non-infected tissue.

Sampling methods Ú sample from healthy skin or non-exsudative lesion: 2 Ú Ú Ú swabs + transport medium skin with supperficial cutaneous lesions: two swabs + transport medium cutaneous inflammation, erysipelas, hypodermitis: skin disinfection, syringe aspiration / biopsy bites: aspirate the liquid / swab for anaerobic bacteria supperficial wounds: aspiration using a syringe ulcerations, sores, necrotic cutaneous lesions: deep tissue biopsy

Type of sampling: Ú Specimens for normal sterile cavities (ascitic fluid, peritoneal, pleural, synovial exudates); Ú Specimens from purulent collections after septic metastasis; evolve related to the anatomic site, from normal colonized regions (oral cavity, digestive system, genital areas); Ú Pus from the plagues (traumatic, surgical, bite) or abscesses contiguous with mucosal surfaces in open wounds. These specimens can be contaminated with normal flora; Ú Infected burn lesion, bedsore always contaminated. Frequently, SAMPLING from the WOUNDS SPECIMEN IS UN-REPEATABLE

Procedure Ú Clean the wound Ú Remove all exsudates Ú Debride the necrotic tissue Ú Apply cutaneous antiseptic and leave to dry Ú Rinse with sterile saline Ú Take a biopsy of the lesion or Ú Curette the active edge of the lesion and Ú Place the sample bottom of a sterile vial Ú Aspirate the inflammatory liquid – fine needle Ú Swabbing is not a adequate procedure and should be avoided

Sampling technique: Ú skin decontamination (~ blood culture); Ú remove of the stagnant exudates by washing, from the burns; Ú for biopsy: sterile cutaneous perforator - 4 mm Ú sterile swabs; Ú special transport culture media - from lab; Ú slides for smears.

Puncture of the closed collections: Ú use fine needle to avoid false negative results, when the pus is viscous; Ú skin decontamination and drying; Ú puncture and aspiring of the pus; Ú decant the pus in a special recipient which allow anaerobic bacteria to survive; Ú “anaerobic syringe”: remove the air from the syringe and put a rubber stopper in the needle. The time for analyzing is 30 minutes.
Skin lesion (blood culture if possible)

Sampling during surgery intervention: Ú for abscesses (pelvic, abdominal, chest) – sterile recipients for pus collection; Ú for purulent membranes: some fragments must be transported to laboratory in 30 minutes.
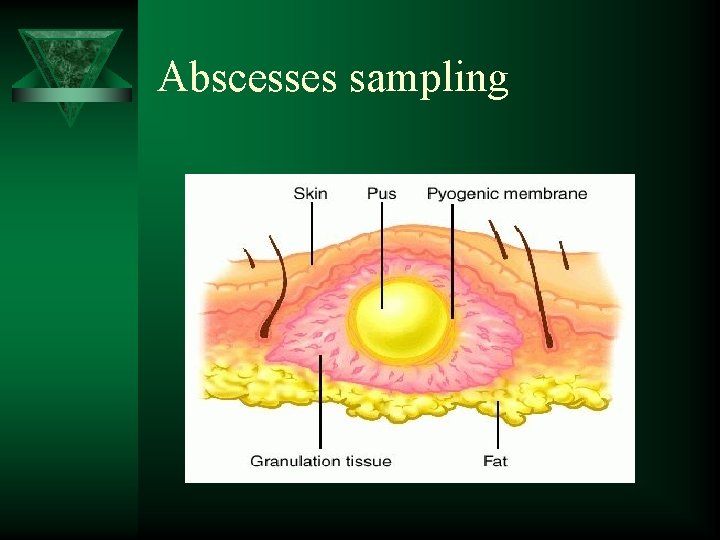
Abscesses sampling

Sampling from fistula: Ú Sampling with the swab it is not a good technique: the lack of a good identification of the microorganism with clinical significance (normal flora of the surface); Ú Preferable: washing, skin decontamination, deep curettage of the fistula and quick transport of the sample at the laboratory.

Sampling from burns, plagues and chronic skin ulcers: Ú Big areas of eroded skin: more sampling from many places of the lesion; Ú Sampling of the primary infection through uninfected tissue – Remove the stagnant exudates or topics antibiotics, by washing and using sterile swabs; – Spin the swab until a minimum bleeding. – Send the swab for examination. Ú During surgery intervention: specimens from diagnostic tissue biopsies, from the plague; Ú Oblique puncture and aspiration to normal skin: for chronic skin ulcers.

Burned wards: • Superficial skin samples: any topical agents are removed with a sterile compress soaked in normal sterile water; • the area of interest can be swabbed using two sterile swabs that are placed in transport culture medium to preserve fragile bacteria; • We can use two techniques: – Z stroke technique: a specified surface area of skin, generally 2 -4 cm 2, is swabbed using several zigzag movements; – Levin technique: the tip of the swab is rotated several times over a localised area.

Burned wards: • Skin biopsy: should be used a disposable biopsy punch (3 – 5 mm) or by cutting area measuring 1 -2 cm by 1, 5 cm • sterile scalpel and forceps, • detaching the tissue along the subcutaneous fat layer, and also • sampling healthy, unburned tissue; fragments usually weigh 20 -50 mg.

Wrong techniques: Ú sampling on swab for anaerobic bacteria; Ú sampling on swab, instead of syringe, biopsy or curettage; Ú sampling of necrotic tissue, expose to contamination

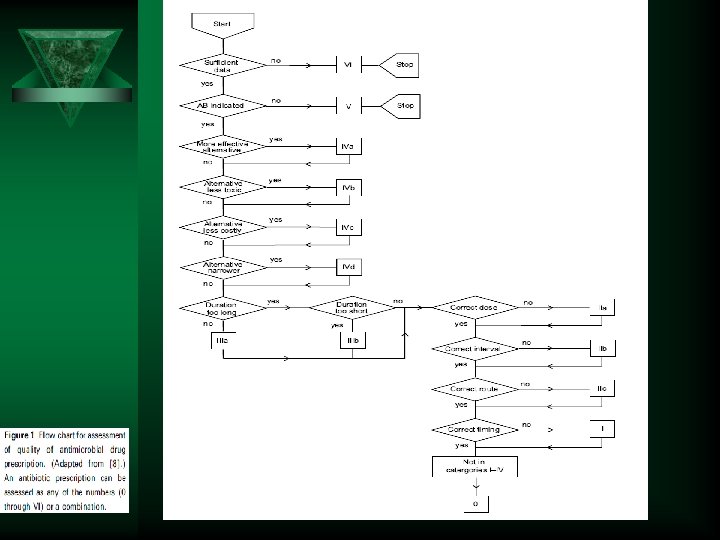
Results and interpretations Ú Clinical information is essential to the interpretation of the microbiological examination: – nature and site of the lesion – sampling method – global context: age, immune status, diabetes, ongoing infections, corticotherapy, – recent antibiotic treatment. Ú Only comparison of the microscopic examination and results of culture with the symptoms can lead to reliable interpretation Ú Mixed culture (>3 bacterial species): contamination

Results’ s interpretation: Ú Puncture / aspiration of the closed collections: – has clinical significance the isolated with the same microscopic appearance like bacteria from directly smear; – when the patient has systemic infection, isolating of the same bacteria from the blood culture can be a major criteria of clinical significance of the bacteria from the pus; Ú Specimen during surgery intervention: – has clinical significance the bacteria observed through direct examination;

Results’ s interpretation: Ú Specimens obtained by curettage: – has clinical significance bacteria associated with PMNs, in direct microscopy. Ú Specimens from plagues and burns: – has clinical significance the conditioned pathogens ≥ 105 CFU/g tissue; – β-hemolitic streptococci always has clinical significance; – the presence of epithelial cell means inefficient removing of the stagnant exudates; – for the correct sampling: quantitative criteria: ≥ 106 CFU/ swab.
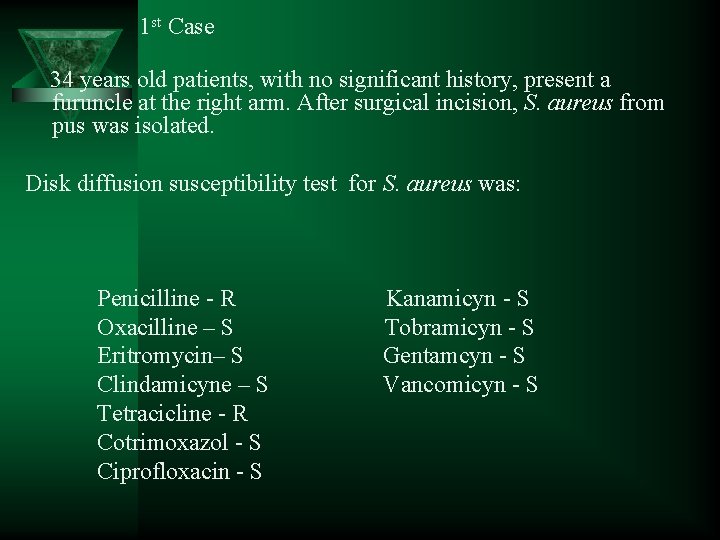
1 st Case 34 years old patients, with no significant history, present a furuncle at the right arm. After surgical incision, S. aureus from pus was isolated. Disk diffusion susceptibility test for S. aureus was: Penicilline - R Oxacilline – S Eritromycin– S Clindamicyne – S Tetracicline - R Cotrimoxazol - S Ciprofloxacin - S Kanamicyn - S Tobramicyn - S Gentamcyn - S Vancomicyn - S

1 st Case: comments • Strain from community, resistance to penicilline (penicillase production); • No Vancomycin test diffusion method (the method is no appropriate for this test); • Susceptibility test - it is NOT necessary (treatment = surgery + local antibiotics or antiseptics).

2 nd Case 8 years old child with osteomyelitis (traumatism in recent past). S. aureus was isolated. Results of disk diffusion susceptibility test for S. aureus: Penicilline – R Kanamycin - S Oxacilline – R Tobramycin - R Cefoxitin – R Gentamycin - S Ceftriaxone – S Vancomycin - S Eritromycine– R Linezolid - S Clindamicine - S (inducible resistance) Tetracycline - R Cotrimoxazol - S Ciprofloxacine - S

2 nd Case: comments • Resistance to Oxacilline – confirmation by Resistance to Cefoxitine; • False sensitivity to Ceftriaxone (treatment failure when we use beta – lactamase antibiotics for MRSA); • Low level resistance strains to Vancomycin can’t be identified by disk diffusion test; • Clindamycin – the best for ostheomyelitis’ s treatment – high level in the inflammatory focus; • Inducible resistance can induce treatment failure.

Acute diarrhea

Causative agents: Ú Choleric syndrome: Ú Ú – Vibrio cholerae – NAG Vibrio – ETEC, EPEC, ECAD, ECEAg Dysenteric syndrome: – Shigella spp. – EIEC – Campylobacter – Non - typhoid Salmonella – Yersinia enterocolitica Hemorrhagic diarrhea: – EHEC Enteric fever diarrhea: – Salmonella enterica serovar Typhi, Paratyphi A, B, C Diarrhea post antibiotics treatment: – Clostridium difficile – Staphylococcus aureus Ú Viruses: – – Ú rotavirus, enteroviruses, adenoviruses, coronaviruses, etc. Parasites: – Giardia duodenalis, – Cryptosporidium parvum, – Entamoeba.

Stool sampling: Ú Necessary: – sterile recipient – Cary Blaire transport culture media – 1 g of feces (blood, pus, mucus).

Sampling by swab: Ú Useful for patient with dysenteric syndrome; Ú After sampling the swab is introduced in transport culture media (Cary Blaire).

Transport Ú Maximum 1 hour. Ú If the transport need to be delayed, the sample must be preserved at 4°C for maximum 24 hours. Ú Requesting paper must specify the clinical syndrome.
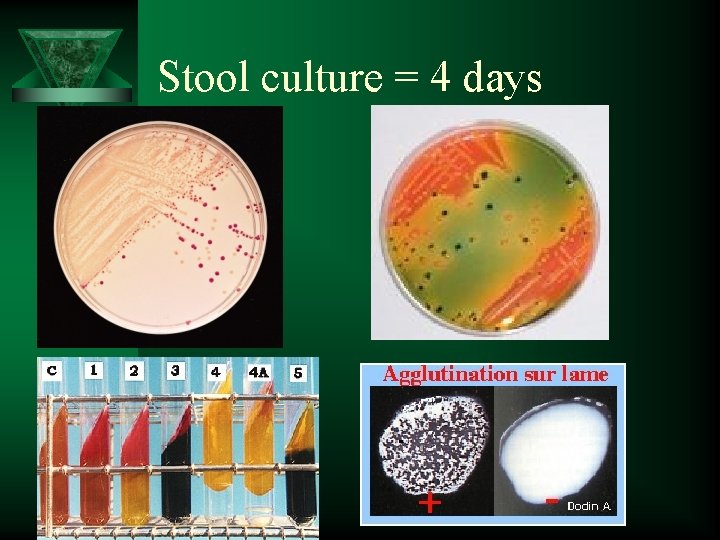
Stool culture = 4 days

3 rd Case 7 years old child, went a party in a fast food restaurant. After 12 hours, the child present numerous stools (8 / 24 hours), yellow, with mucus. The same symptoms were noted fort other 8 children who were at the same party. Pathologic product: feces. Suspected food? Hamburger Coprocitogram: ~ 10 PMN s / field (x 400) Stool culture: present Salmonella group B Antibiogram: Ampicilin - R Cotrimoxazol - R Ciprofloxacine - S Ceftazidime – S Nalidixic acid– R Treatment?

Causative agents of food borne diseases Superior GI syndrome Ú – – Bacillus cereus Staphylococcus aureus Inferior GI syndrome Ú – – – – Clostridium perfringens Escherichia coli ( ECET, ECEI, ECEH) Non typhoid Salmonella Shigella Vibrio cholerae V. parahaemolyticus Yersinia enterocolitica Campylobacter Ú Neurologic syndrome – Clostridium botulinum Ú Pharynx syndrome – Streptococi group A, C, G – S. zooepidemicus

4 th Case 23 years old, in last 24 hours presented 6 stools semi-consistent. Pathologic product: feces. Coprocitogram: ~ 20 PMN s/ field (x 400) Stool culture: present in high quantity: - Proteus vulgaris, - Yersinia enterocolitica. Ethiology?

5 th Case January, 2007 2 years old patient, 12 watery stool / 24 hours. Pathologic product: stool. Coprocitogram: inflammatory reaction – absent (x 400). Stool culture: Proteus mirabilis in high quantity. Latex agglutination – positive for rotaviruses.

The final conclusions !!! Ú Why did you made Clinical Microbiology ?

You have refreshed your memory regarding: Ú The main rules for sampling pathologic products; Ú The diagnostic methods for the most important infectious; Ú The interpretation of the results; Ú The types of antibiograms and Ú Interpretative antibiogram.

Why perform in vitro susceptibility testing? Ú DUAL PURPOSE OF SUSCEPTIBILITY TESTING: – Individual (to guide the selection and modification of antimicrobial therapy) – Epidemiological.

When should a susceptibility test be performed? Ú Susceptibility testing should not be routinely performed on organisms that are part of the normal bacterial flora and usually not considered pathogenic. Ú Establishing the need for susceptibility testing requires a close working relationship between microbiologists and clinicians.

Can susceptibility and/or resistance of bacteria to an antibiotic be predicted? Ú NATURAL RESISTANCE: – permanent characteristic of the species, which is known and predictable. Ú ACQUIRED RESISTANCE: – characteristic of some bacterial strains, which is evolutionary, unpredictable and justifies the need for susceptibility testing.

What criteria are used to select the antibiotics to be tested? Ú The choice of antibiotics to be tested is made in relation to their therapeutic value and their usefulness to detect resistance mechanisms.

Some current resistance issues Ú healthcare-associated MRSA (HA-MRSA) Ú community-associated MRSA (CA-MRSA) Ú extended spectrum -lactamase (ESBL)

Conclusion – Antibiotic susceptibility testing, at the interface between the diagnosis and therapeutic decision, is a key element essential for guiding both microbiologically-documented and empiric antibiotic therapy. – Not only is the susceptibility test result of immediate interest for the clinician to guide selection of antimicrobial therapy, but it also plays a role as an epidemiological surveillance tool for local bacterial.

Conclusion Ú The evolution of resistance, as well as the development of new antibiotics and laboratory techniques make a close working relationship between the microbiologist and the clinician more necessary now than ever before.


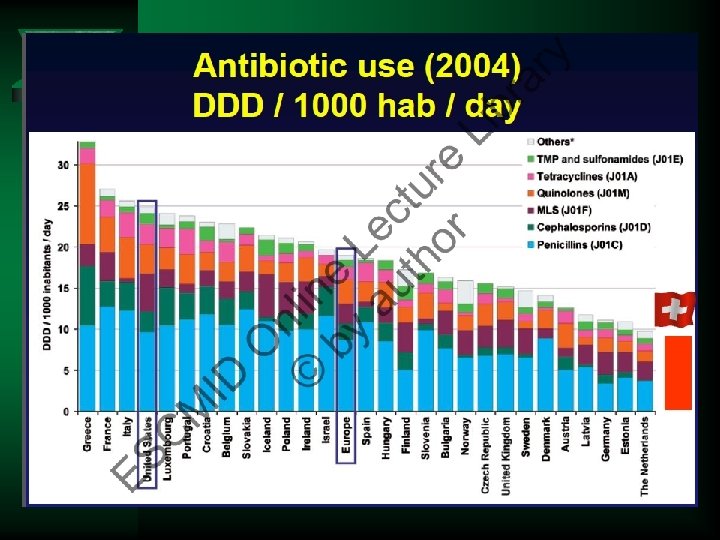
Antimicrobial Stewardship: Measuring, Auditing and Improving Ú How to measure antimicrobial consumption Ú How to audit antimicrobial prescriptions in a hospital Ú Linking antimicrobial resistance to antimicrobial exposure with patient or aggregate data Ú Educational interventions to improve hospital prescribing

Antimicrobial Stewardship: Measuring, Auditing and Improving Ú Audit of antibiotic surgical prophylaxis Ú What are the competencies required to be a good antibiotic prescriber Ú Increase of antimicrobial consumption in a medical intensive care unit Ú Social and behavioural approach to antibiotic prescribing in hospitals












Thank you !
- Slides: 69