Laboratory Diagnosis for avian flu H 5 N

Laboratory Diagnosis for avian flu (H 5 N 1) in human Wattana Auwanit MT, Ph. D Biohazard Laboratory and Immunology Section National Institute of Health Department of Medical Sciences Ministry of Public Health Thailand

Influenza Virus § Influenza A - variety of different host species (avian, human, swine, etc. ) - HA undergoes minor and occasional major changes - Cause severe disease and associated with epidemic and pandemics § Influenza B - Infects only in human but not severe as type A virus - Relative slow change in HA § Influenza C - Uncommon strain, infects in man only

Influenza type A Human influenza : H 1 N 1, H 3 N 2, H 1 N 2 Avian influenza : H 5 N 1

H 5 N 1 infection laboratory diagnosis

Questions When to collect specimen? What specimens to collect? How to store / transport specimens? How to test? How to interpret results?

specimens Quality of specimen is a most importance. Best test cannot give a accurate result on poor specimen.

Type of specimens Respiratory specimens in VTM – viral RNA detection – – Nasopharyngeal aspirate Nasopharyngeal swab Throat swab Nose swab (not recommend) Feces – low virus level Blood for serology – viral antibody detection Autopsy tissues – viral RNA detection

Timing 2 -3 days after illness onset End the peak at 7 -8 days Nasopharyngeal wash/aspirate > Nasopharyngeal swab ~ throat swab

Storage of specimens At hospital and during transportation – wet ice, refrigerator (4 o. C) At the laboratory – Store at – 70 o. C not at -20 o. C – Avoid freeze-thaw

Laboratory diagnosis § Detection of viral antigen • Immunofluorescence assay (IFA) • Rapid test (Bed-side rapid assay) § Detection of viral genome • Reverese transcription and polymerase chain reaction assay (RTPCR) • Real-time RT-PCR § Virus culture in cell lines § Serology test • Micro-neutralization test for seroconversion (blood)
Rapid test
Rapid tests § Simple (1 hr), differentiate only A/B not H 3, H 1, H 5 § Commercially available - Directigen Flu A - Now Flu A+B - etc. § Less sensitivity (17/35 : 48%)

Sensitivity of rapid antigen tests for H 5 N 1 disease Pos (%) Hong Kong 1997 9/11 (82%) Yuen et al Lancet 1998 Test Directigen. Flu. A Thailand 2004 17/35 (48%) Directigen, Binax NOW Thai MOPH/US CDC- unpublished Vietnam 2004 3/15 (20%) Directigen and Quick. Vue de Jong M & Farrar J - Unpublished Data from : JSM Peiris, The University of Hong Kong, and Queen Mary Hospital
Immunofluorescence assay (IFA) § Sensitive method to identify influenza A and B, parainfluenza, adenovirus, etc. § Using type specific monoclonal antibody to viral antigen (A/H 5, A/H 1, A/H 3, etc. ) § 2 -3 hours of testing § Need enough cells
Molecular assay Reverse Transcription-PCR (RT-PCR) § For detection of viral gene and sub-typing (influenza A, B, A/H 1, A/H 3, A/H 5) using specific primers and probe § Target on viral haemagglutinin gene § 24 hours testing § High specificity

Molecular assay Real-time RT-PCR § Increase sensitivity § Increase speed (4 -5 hr) § Quantitative § Multiple genetic target (H and N gene)

Virus culture § Virus culture in MDCK cell, LLC-MK 2, HEp -2 c and RD cells § Results time 7 -14 days § The virus identification by IFA or haemagglutination inhibition assays using reference antisera is required § Need Biosafety laboratory level 2/3
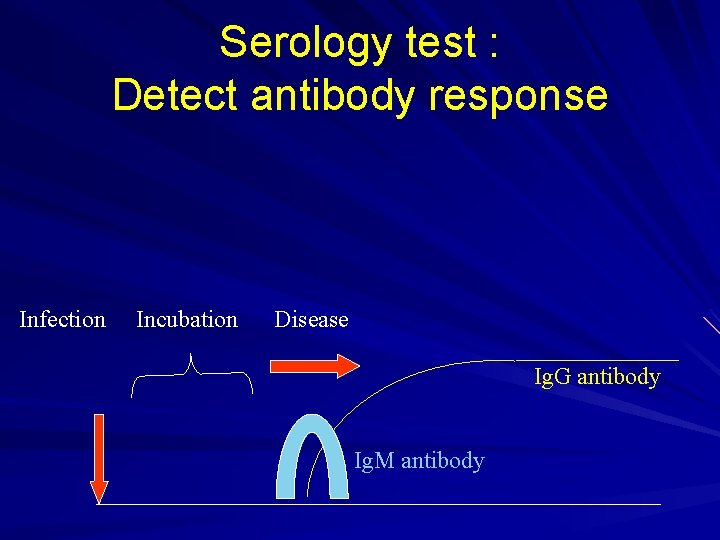
Serology test : Detect antibody response Infection Incubation Disease Ig. G antibody Ig. M antibody

Serology § For viral antibody detection § Micro-neutralization test (BSL-3) § Detection of 4 -fold rising of specific antibody between acute (3 days) and convalescence (14 days) sera. § For confirmation of suspected case, not for routine diagnosis. § 14 days or more.

Gold standard for diagnosis of H 5 N 1 infection Culture RT-PCR Sero-conversion by micro-neutralization tests (BSL-3(




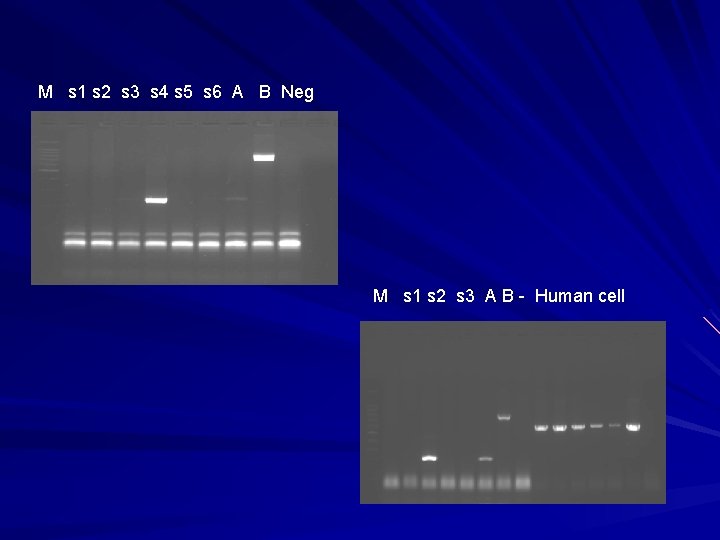
M s 1 s 2 s 3 s 4 s 5 s 6 A B Neg M s 1 s 2 s 3 A B - Human cell

Interpretation of H 5 N 1 infection Positive by RT-PCR or real-time PCR with at least 2 sets of specific primers to H 5 If only 1 set of primer is positive report inconclusive then follow-up samples will be requested Seroconversion of H 5 N 1 antibody (1 day, >14 days sera) by microneutralization test is a confirmation of H 5 N 1 infection DNA sequencing will be used only for risk assessment and molecular epidemiology not for confirmation of H 5 N 1 infection
DNA Sequencing • Genetic analysis of influenza viruses • Risk assessment • Molecular epidemiology

Influenza A Viruses § Influenza virus contains 8 segments of viral genome which increase the potential to form the recombinants by interchange of gene segments from two different viruses, both among human strains and avian and human strains. This may cause virulent human strains to evolve. § A major change in HA and sometime a new NA as well (antigenic shift) result in pandemic strain appears in man (10 -15 years interval) § A minor change in HA and NA (antigenic drift) result in epidemic (2 -3 years interval)

Thank you
- Slides: 29