Laboratory course of biomedical engineering Introduction Vesa Vuorinen

Laboratory course of biomedical engineering Introduction Vesa Vuorinen, Noora Isoaho, Tommi Palomäki, Antti Rautiainen 2016

Expectations? 1. Choose a card that reflects your expectations for the course. 2. Show the card to other students in your group and explain it. 3. Combine your separate expectations in one mutual expectation and tell about it to the other groups/teachers.

ELEC-D 8723 Laboratory course of biomedical engineering (5 cr) Aims of the course • Course is aimed at both EST and BIO students • ”Material choice for implantable sensor” • How to approach a research problem – Recognizing the relevant research questions – Selection criteria for research methods – Research plan • Laboratory practices – Safe laboratory work

ELEC-D 8723 Laboratory course of biomedical engineering (5 cr) Main teachers and evaluation Main teachers: - Vesa Vuorinen and Emilia Peltola Evaluation: – Scale 0 -5 – Research plan (group) • 31. 3. 2016 – Report (group) • 31. 5. 2016 – Laboratory notes (personal) • 31. 5. 2016 – Essay on electrochemical sensors (personal) • 31. 5. 2016 Final grade = group ± 2

Material choice for implantable sensor • Objective is to choose the most suitable surface structure for an implantable sensor – Biocompatibility – Functionality
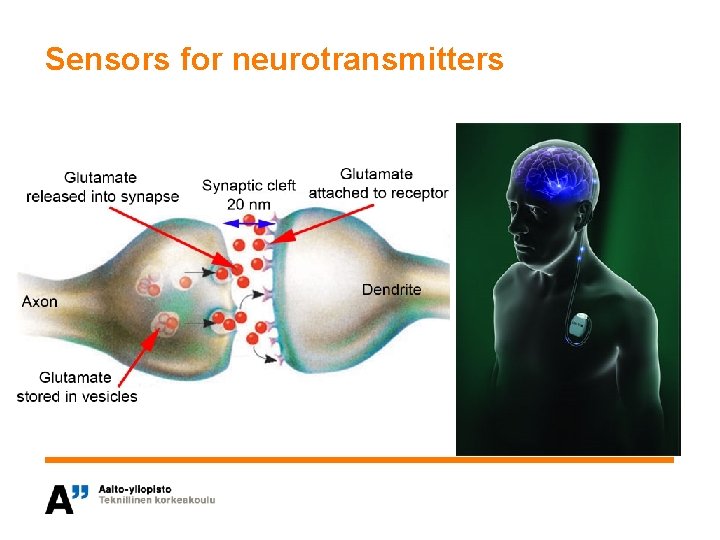
Sensors for neurotransmitters
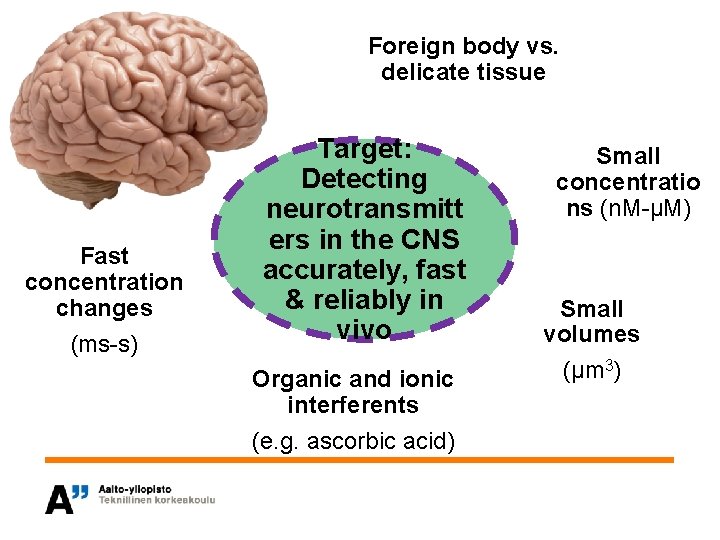
Foreign body vs. delicate tissue Fast concentration changes (ms-s) Target: Detecting neurotransmitt ers in the CNS accurately, fast & reliably in vivo Organic and ionic interferents (e. g. ascorbic acid) Small concentratio ns (n. M-µM) Small volumes (µm 3)
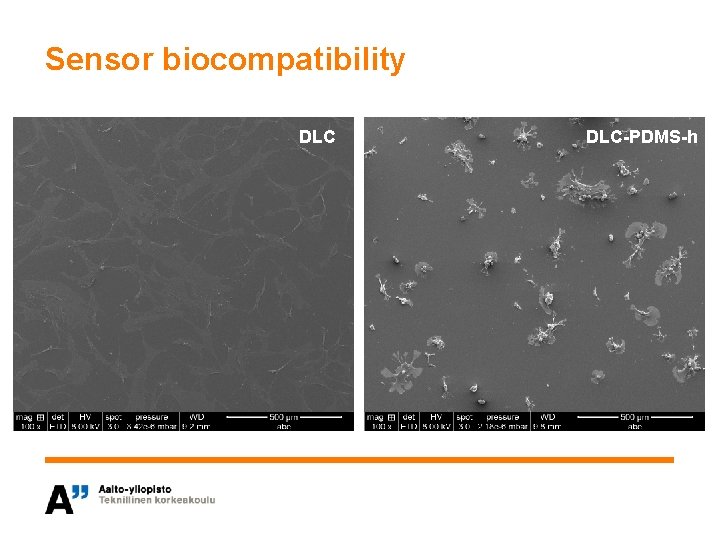
Sensor biocompatibility DLC-PDMS-h
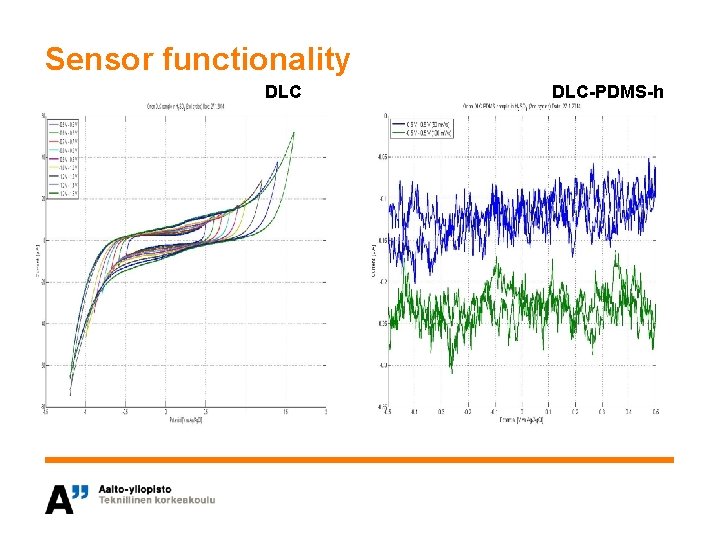
Sensor functionality DLC-PDMS-h
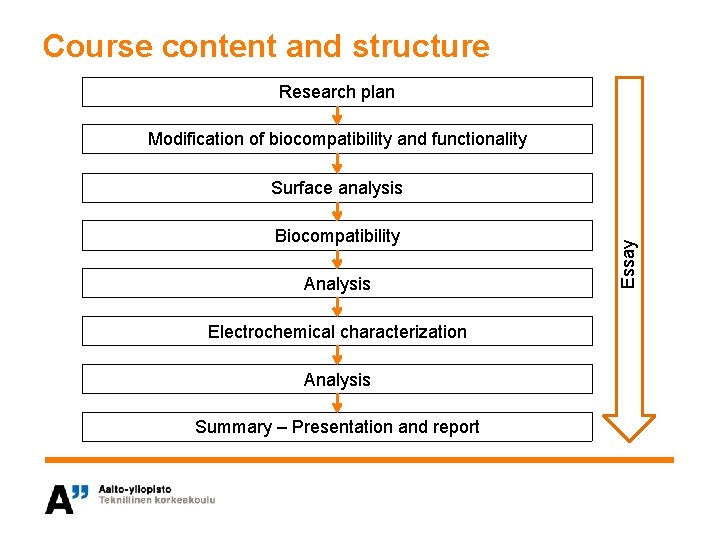
Course content and structure Research plan Modification of biocompatibility and functionality Biocompatibility Analysis Electrochemical characterization Analysis Summary – Presentation and report Essay Surface analysis
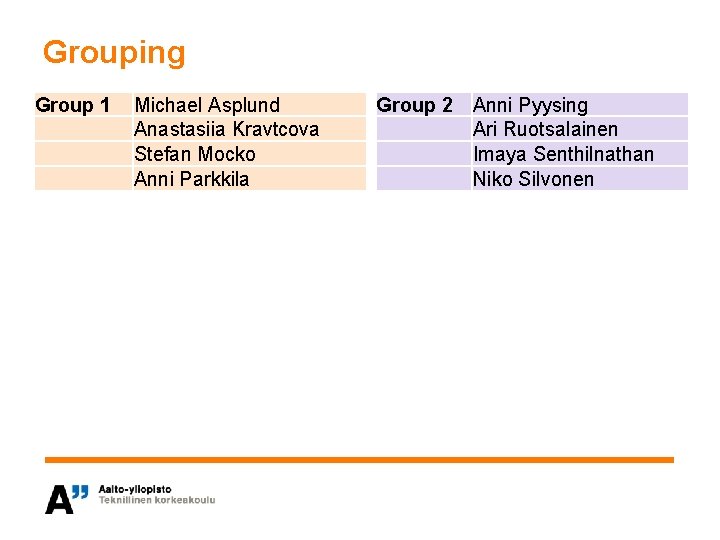
Grouping Group 1 Michael Asplund Anastasiia Kravtcova Stefan Mocko Anni Parkkila Group 2 Anni Pyysing Ari Ruotsalainen Imaya Senthilnathan Niko Silvonen

Available materials and treatments • Base materials: – Silicon – Tetrahedral amorphous carbon (ta-C) • Surface treatments: • • Plasma (Ar, O 2) APTES SAMs (functionalizing) Sputtering (Au, Pt, Cr, Ti, Ni, Al, Ti. W) Evaporation [C(graphite)] Acid/base treatments (no HF) Mechanical coarsening Anodic/cathodic treatment Graphene flakes, carbon nanotubes

Available analysis methods – – Contact angle measurement Electrochemical characterization Cell culture (direct contact/extracts, MTT) Microscopy (Optical, SEM+EDS)

Course contents and structure Research plan Modification of biocompatibility and functionality, surface treatments Surface analysis, contact angle, SEM Biocompatibility, cell culture Analysis, optical microscopy, SEM, MTT Electrochemical characterization, CV Analysis Summary – Presentation and report

Schedule Wed 2. 3. 2016 Introduction, aims, materials and methods, initializing research plan week 12 -14 Q&A session (16. 3. ) and finishing the research plan week 15 Coatings, contact angle experiments week 16 -17 Cell culture, analysis week 18 -19 Electrochemical characterization week 20/21 Summary, presentation and report

Research plan • 3 -4 reasonably chosen materials • What is going to be tested and why? • Description of methods – What is going to be measured and what is going to be analyzed? – Protocols for cell culture experiments (cell count, time points, sample preparation) – Parameters and setup of electrochemical measurements (solution, analytes, concentrations) – What is the experimental output? • What are the target conclusions? – How do the results reflect the real in vivo situation?

Introduction of available methods
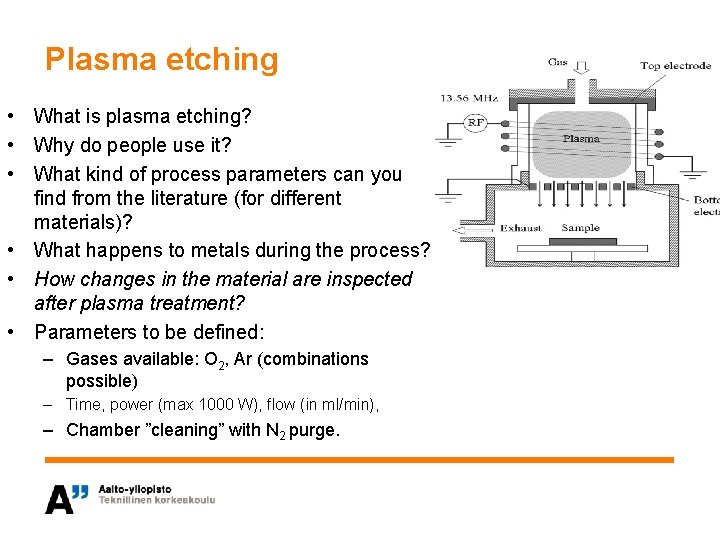
Plasma etching • What is plasma etching? • Why do people use it? • What kind of process parameters can you find from the literature (for different materials)? • What happens to metals during the process? • How changes in the material are inspected after plasma treatment? • Parameters to be defined: – Gases available: O 2, Ar (combinations possible) – Time, power (max 1000 W), flow (in ml/min), – Chamber ”cleaning” with N 2 purge.

Sputtering Atoms / molecules are removed from the target surface by ion bombardment → particles are transferred through the gas phase and condensate on the substrate surface as a film. + Uniform and thick coating + Applicable to complex compounds - Difficult to control the chemical composition of the surface - Line of sight technique

SAM coating SAMs are formed spontaneously from the components of the system. + Uniform thin film that is (relatively) easy to make. + Coating of curved surfaces possible. - Limited amount of available molecules and substrates. - Hard to make thicker films than monolayers. - Mechanically and chemically frail.

Contact angle measurement Principle: A drop of liquid (polar/nonpolar) is dropped on the surface. The contact angle is measured and defined with a computer. + + + - Fairly simple experiment. Surface energy (min. 2 different liquids, preferably more) Information on protein adsorption (some) Several sources of error (surface roughness, debris, experimenter…) Different models for defining surface energy Hydrophilic Hydrophobic [Yuan and Lee, 2013]

SEM+EDS • Field emission scanning electron microscope (FESEM) – JEOL JSM-6335 F – Magnification 10 - 500000 – The optimal resolution is 1. 5 nm. – Imaging modes SEI, BEI • LINK x-ray microanalyzer (EDS)

Surface characterization • Microscopy – Optical microscopy – SEM • Surface structure and chemical analysis Not – XRD (X-Ray Diffraction) available – XPS (X-ray Photoelectron on this course Spectroscopy) – EDS (Energy-dispersive X-ray spectroscopy)
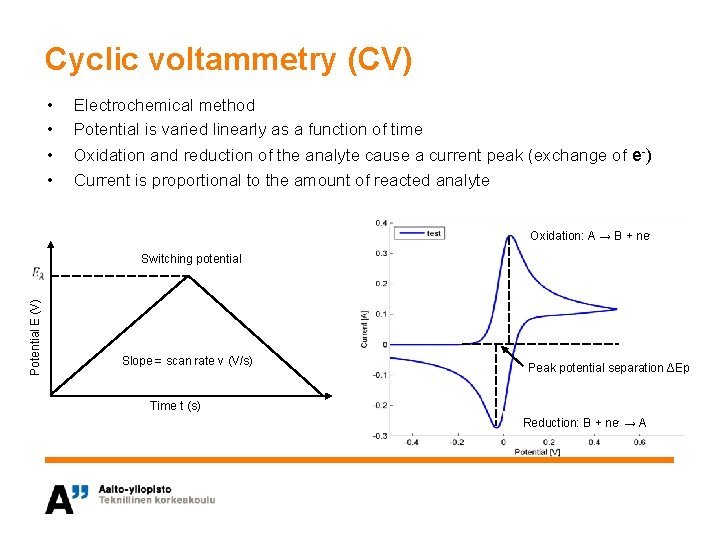
Cyclic voltammetry (CV) • • Electrochemical method Potential is varied linearly as a function of time Oxidation and reduction of the analyte cause a current peak (exchange of e-) Current is proportional to the amount of reacted analyte Oxidation: A → B + ne- Potential E (V) Switching potential Slope = scan rate v (V/s) Peak potential separation ΔEp Time t (s) Reduction: B + ne- → A
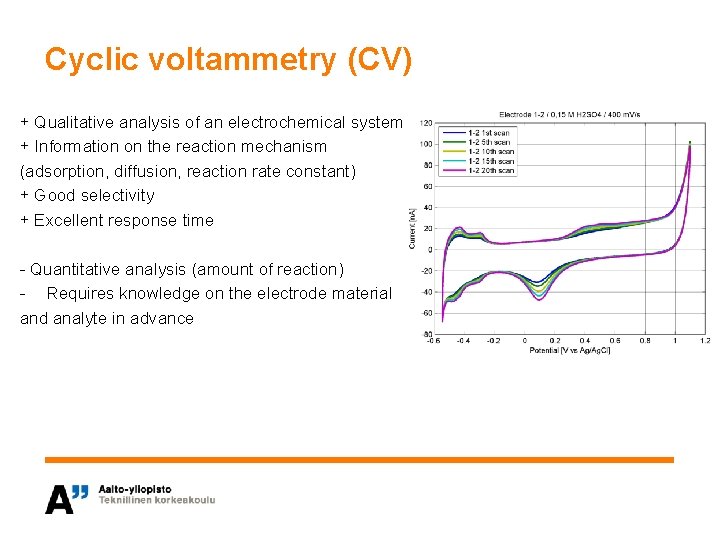
Cyclic voltammetry (CV) + Qualitative analysis of an electrochemical system + Information on the reaction mechanism (adsorption, diffusion, reaction rate constant) + Good selectivity + Excellent response time - Quantitative analysis (amount of reaction) - Requires knowledge on the electrode material and analyte in advance

Cyclic voltammetry (CV) Experimental setup: • Three-electrode cell • Working electrode is the electrode under investigation • Potential is measured against a reference electrode • Current passes through the working and counter electrodes • The electrodes are connected to a potentiostat that sets V and measures I Ar / N 2 Reference electrode (Ag/Ag. Cl) Working electrode Counter electrode (graphite rod) Analyte-containing solution Ar / N 2

For the research plan Possible measurements: • Width of the potential window • Selectivity: measurement with an analyte and an interferent • Sensitivity: lowest concentration that can be detected • Electron transfer rate for determining how fast the electrode is Choose 2 -3 analytes and appropriate solutions (H 2 SO 4, KCl, PBS…): • Dopamine • Ascorbic acid Also choose: • Uric acid Potentiostat parameters: • Ferrocenemethanol – Potential limits • Ruthenium Ru(NH 3)63+ – Scan rate Solutions and analytes: • Ferrocyanide Fe(CN)64– Concentration – Purging (Ar or N 2)

Biocompatibility - Standards define several experiments to be conducted when testing for biocompatibility. - In general, when designing biomedical devices it is best to try to avoid toxic or unknown materials. • Testing is both time consuming and expensive!
Choosing the cell line Growth properties Other properties Recognition Type Stability Availability Primary / secondary cell line? Cells available in this course are mouse 3 T 3 fibroblasts.

Passage

Detaching the cells Method Equipment Shaking Careful shaking or Loosely adherent intense pipetting cells, mitotic cells Scraping Cell scraper Protease sensitive cells Enzymatic Trypsine Adherent cells Dispase Detaching epidermal cells as ”sheets” Temperature responsive culture dish Type of cells
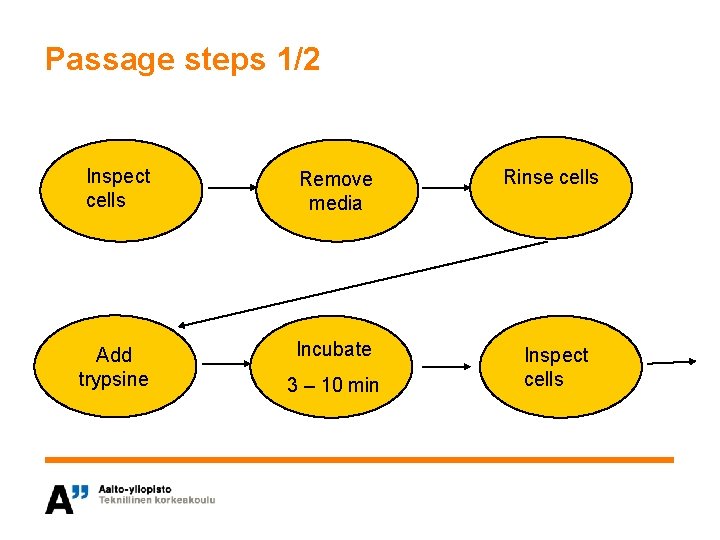
Passage steps 1/2 Inspect cells Remove media Rinse cells Add trypsine Incubate Inspect cells 3 – 10 min
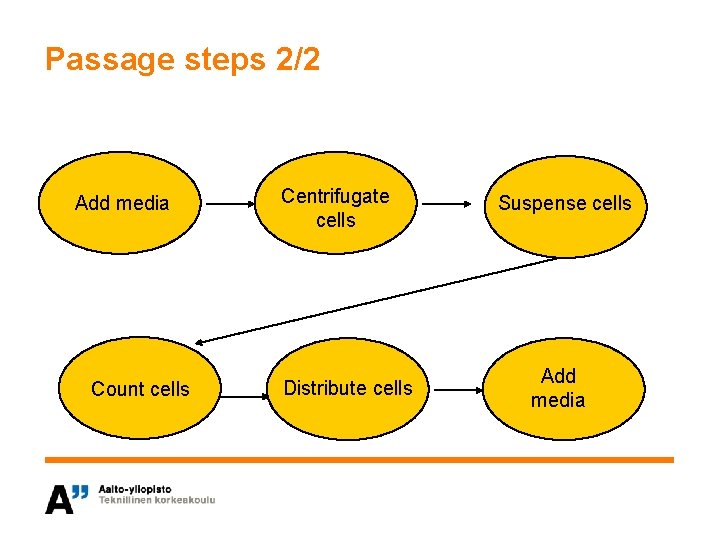
Passage steps 2/2 Add media Count cells Centrifugate cells Distribute cells Suspense cells Add media

SEM Electron microscopy – Preparations for biological sample • Dehydration • Coating with conducting material
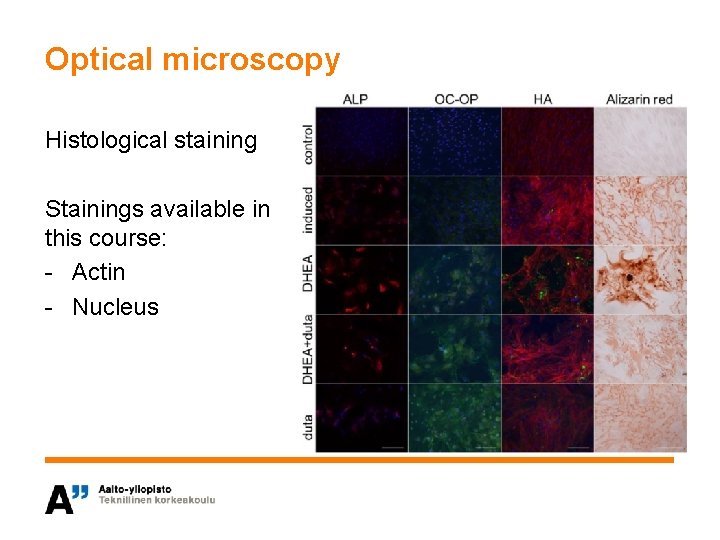
Optical microscopy Histological staining Stainings available in this course: - Actin - Nucleus

MTT assay Color change of yellow MTT to purple is related to (live) cell metabolic activity. Adsorbance can be measured with a spectrometer. MTT e. g. (3 -(4, 5 -Dimethylthiazol-2 -yl)-2, 5 -diphenyltetrazolium bromide

People as contamination sources People generate particles continously (≥ 0. 3µm) • Standing still 100 000 particles / min • Small hand gesture 500 000 particles / min. • Normal hand/body movement 1 000 particles / min. • Walking 7 500 000 particles / min • Walking fast 10 000 particles / min

Aceptic technique • • • Washing hands Labcoat with long sleeves Gloves Spraying surfaces, gloves and equipment with Et. OH Reagents and equipment used only for cell culture Working in laminar flow cabinet
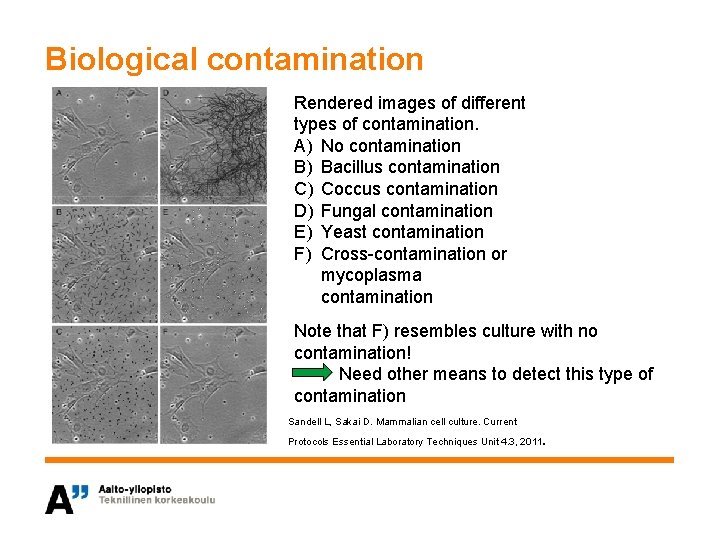
Biological contamination Rendered images of different types of contamination. A) No contamination B) Bacillus contamination C) Coccus contamination D) Fungal contamination E) Yeast contamination F) Cross-contamination or mycoplasma contamination Note that F) resembles culture with no contamination! Need other means to detect this type of contamination Sandell L, Sakai D. Mammalian cell culture. Current . Protocols Essential Laboratory Techniques Unit 4. 3, 2011

Cross contamination 246 cell lines were inspected in 1976. • 14% were contaminated with cells from different species • 25% of human cells were He. La cells • → almost 30% were labeled wrongly

Most common mistakes in aceptic technique I

Most common mistakes in aceptic technique II

Most common mistakes in aceptic technique III

Hazardous situations Watch video on aceptic technique from My. Courses!

Final report • What did your group do? • Did you deviate from your plan? Why? • What kind of results did you get? • How would you continue the study?

Materials etc. - My. Courses - Use the forum for questions - Other groups might have been wondering the same!
- Slides: 46