Laboratory Calibration Week 10 Volumetric Glassware 1312022 1

Laboratory Calibration Week 10 – Volumetric Glassware 1/31/2022 1

This Lesson • Revision of last week • Calibrating volume of glassware using density of water. • Mass, volume, density • Density of water & temperature dependence • Density of water & salinity dependence • Standards – documents • Analytical glassware • Normal distribution, mean and standard dev. 1/31/2022 2

Last Week • • Calibration and calibration standards Analytical balance Accuracy and precision Summation notation Mean and standard deviation NATA Single Point and Repeatability Tests 1/31/2022 3

Check 1. Which are precise? 2. Which are accurate? 3. Which is accurate and precise? 4. What is the mean and standard deviation of: § 1, 4, 5, 6 1/31/2022 4
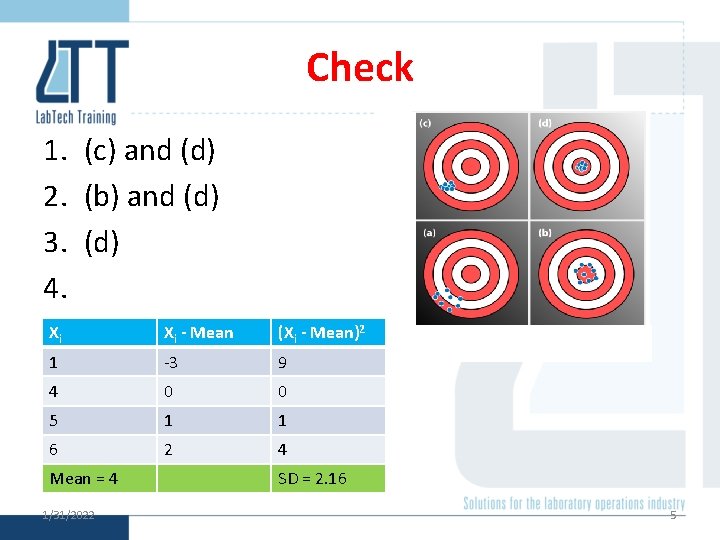
Check 1. (c) and (d) 2. (b) and (d) 3. (d) 4. Xi Xi - Mean (Xi - Mean)2 1 -3 9 4 0 0 5 1 1 6 2 4 Mean = 4 1/31/2022 SD = 2. 16 5

Mass, Volume, Density Today’s lesson revolves around three quantities: mass, volume, density. volume mass 1/31/2022 Density = mass volume 6

Mass Quantity = m, Units = g, kg, tonne 1/31/2022 7

Kilogram Standard • One primary standard for the kilogram is a platinum-iridium cylinder kept in a special vault in France. • Imagine trying to trade goods with no standard weight! 1/31/2022 8

Other Standard Weights • Other standard weights can be purchased by laboratories to calibrate and check balances. • But what about measuring liquids? 1/31/2022 9

Volume Quantity = V, Units = m 3, L, m. L 1/31/2022 10

Metric and Imperial • There are many units of volume. What do you think is the smallest and largest volume below? • • • Gill (Imperial) Gill (US) Pint (Imperial) Quart (US) Gallon (US) 1/31/2022 • • • Centilitre Gallon (Imp. ) Cup Millilitre Litre 11

Metric and Imperial • There are many units of volume. What do you think is the smallest and largest volume below? • • • Gill (Imperial) - 142 m. L Gill (US) - 118 m. L Pint (US) - 473 m. L Pint (Imperial) - 568 m. L Quart (US) - 946 m. L Gallon (US) - 3785 m. L 1/31/2022 • • • Centilitre - 100 m. L Gallon (Imp. ) - 4546 m. L Cup - 236 m. L Millilitre - 1 m. L Litre - 1000 m. L 12

Units errors • Gimli Glider • Air Canada Flight 143 • 23 July 1983 • Wrong fuel weight • Should have used litres and kilograms instead of gallons and pounds • Caused ~ 15000 Litre shortfall! Also a very heavy landing!!!! 31/01/2022 13

Metric System • • • 1 Microlitre = 10− 6 Litre 1 Millilitre = 10− 3 Litre 1 Kilolitre = 103 Litre 1 Megalitre = 106 Litre Note: Prefix dictating amount 1/31/2022 14

Equivalent Volumes Submultiple Name 10− 6 L microlitre Symbols Equivalent volume µL mm 3 cubic millimetre 10− 3 L millilitre m. L cm 3 cubic centimetre 100 L litre L dm 3 cubic decimetre 103 L kilolitre k. L m 3 cubic metre Note: Varying Volumes 1/31/2022 15
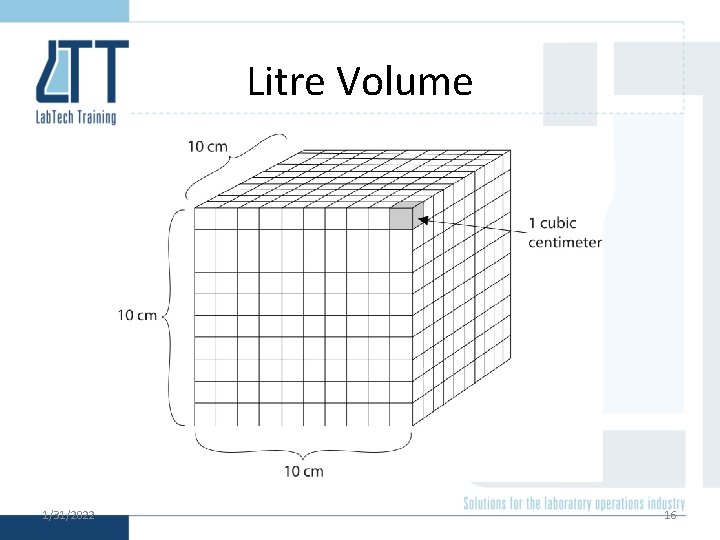
Litre Volume 1/31/2022 16

Density Quantity = r, Units = g/cm 3, g/m. L 1/31/2022 17

Density of some things Substance Cork Ice Water (pure) Water (salt) Fe Au Os 1/31/2022 Density (g/cm 3) 0. 240 0. 916 1. 000 1. 030 7. 870 19. 320 22. 570 18
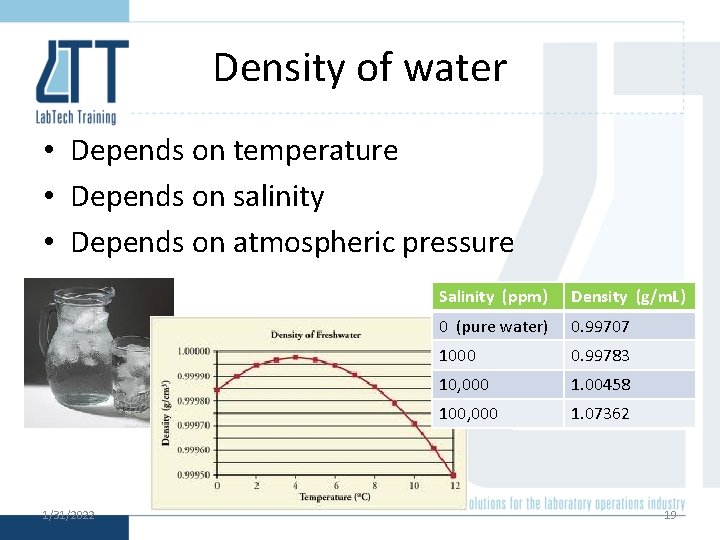
Density of water • Depends on temperature • Depends on salinity • Depends on atmospheric pressure 1/31/2022 Salinity (ppm) Density (g/m. L) 0 (pure water) 0. 99707 1000 0. 99783 10, 000 1. 00458 100, 000 1. 07362 19

1/31/2022 Salinity (ppt) Density (g/m. L) 0 (pure water) 9. 97075 1 9. 97831 10 1. 00458 100 1. 07362 20
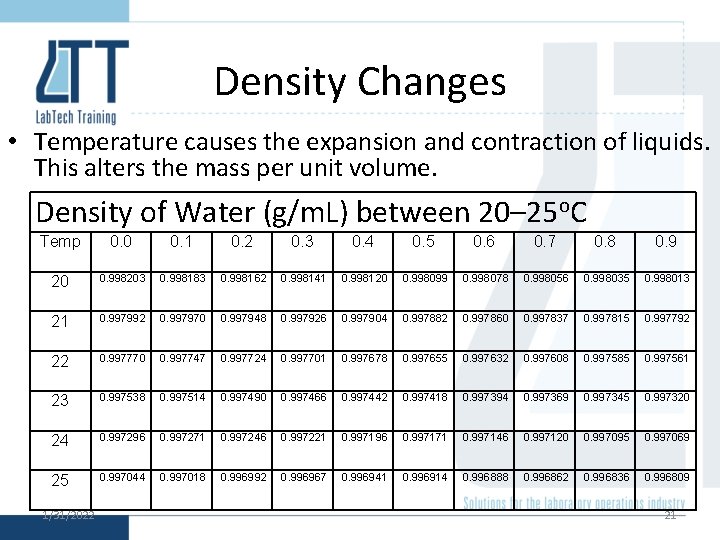
Density Changes • Temperature causes the expansion and contraction of liquids. This alters the mass per unit volume. Density of Water (g/m. L) between 20– 25 o. C Temp 0. 0 0. 1 0. 2 0. 3 0. 4 0. 5 0. 6 0. 7 0. 8 0. 9 20 0. 998203 0. 998183 0. 998162 0. 998141 0. 998120 0. 998099 0. 998078 0. 998056 0. 998035 0. 998013 21 0. 997992 0. 997970 0. 997948 0. 997926 0. 997904 0. 997882 0. 997860 0. 997837 0. 997815 0. 997792 22 0. 997770 0. 997747 0. 997724 0. 997701 0. 997678 0. 997655 0. 997632 0. 997608 0. 997585 0. 997561 23 0. 997538 0. 997514 0. 997490 0. 997466 0. 997442 0. 997418 0. 997394 0. 997369 0. 997345 0. 997320 24 0. 997296 0. 997271 0. 997246 0. 997221 0. 997196 0. 997171 0. 997146 0. 997120 0. 997095 0. 997069 25 0. 997044 0. 997018 0. 996992 0. 996967 0. 996941 0. 996914 0. 996888 0. 996862 0. 996836 0. 996809 1/31/2022 21

Check 1. What volume does 10 g of saline water with a salinity of 100, 000 ppm occupy? How does this compare to pure water? 2. How much volume does 10 g of pure water occupy at 20. 1 o. C? How much does it expand on heating to 25. 6 o. C? 1/31/2022 22 22

Check 1. 9. 314 m. L, 10. 029 m. L 2. 10. 019 m. L, 10. 031 m. L, so change is 0. 012 m. L 1/31/2022 23 23

Specific Gravity • Specific gravity is the ratio of the density of the sample to the density of a reference (say water). • i. e. 1/31/2022 24
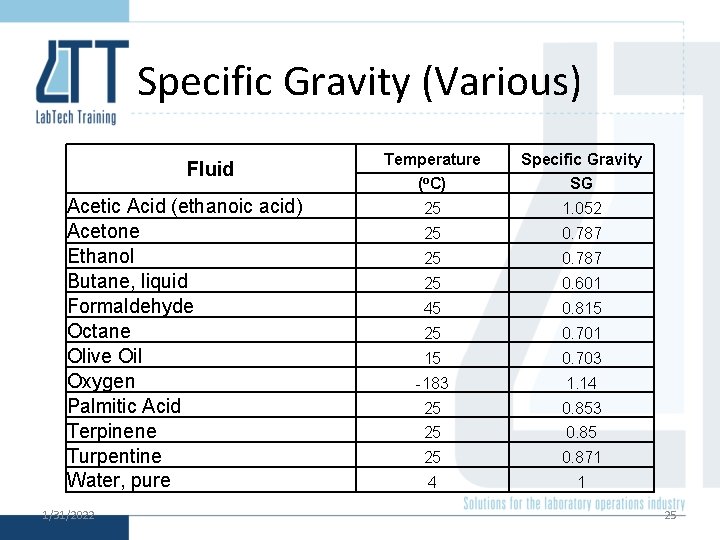
Specific Gravity (Various) Fluid Acetic Acid (ethanoic acid) Acetone Ethanol Butane, liquid Formaldehyde Octane Olive Oil Oxygen Palmitic Acid Terpinene Turpentine Water, pure 1/31/2022 Temperature Specific Gravity (o. C) SG 25 1. 052 25 0. 787 25 0. 601 45 0. 815 25 0. 701 15 0. 703 -183 1. 14 25 0. 853 25 0. 85 25 0. 871 4 1 25

Deionised or Tap Water? • If we are going to calibrate our glassware we need to have a liquid with an accurately known weight per unit volume. • Presence of ions in tap water raises the specific gravity. • Is tap water around the World (or even across Perth) of a standard composition? 1/31/2022 26

Standards 1/31/2022 27

Putting it all together • Glassware must therefore be calibrated per unit volume and temperature using a standardised liquid with a known specific gravity. Here enters the Australian Standard! 1/31/2022 28

What is a Standard? • Standards are published documents setting out specifications and procedures designed to ensure products, services and systems are safe, reliable and consistently perform the way they were intended to. They establish a common language which defines quality and safety criteria. • Standards can be guidance documents including: • • • 1/31/2022 Australian Standards®; International Standards and Joint Standards; Codes; Specifications; Handbooks; and Guidelines. 29

What is a Standard? • These documents are practical and don’t set impossible goals. They are based on sound industrial, scientific and consumer experience and are constantly reviewed to ensure they keep pace with new technologies. • They cover everything from consumer products and services, construction, engineering, business, information technology, human services to energy and water utilities, the environment and much more. 1/31/2022 30

AS 2162. 1 - 1996 1/31/2022 31

Analytical glassware 1/31/2022 32

Analytical glassware • Analytical glassware comes in many shapes and sizes. Glassware also has different many grades. 1/31/2022 33

Bulb Pipette ISO 648: 2008 specifies requirements for volumetric pipettes with one mark. Ex = 5 s, means wait 5 seconds at end of delivery ‘A’ quality twice as accurate as ‘B’. 1/31/2022 34

Other apparatus for todays lab. Volumetric Flask Weighing bottle 1/31/2022 Burette. 35

Normal distribution 1/31/2022 36
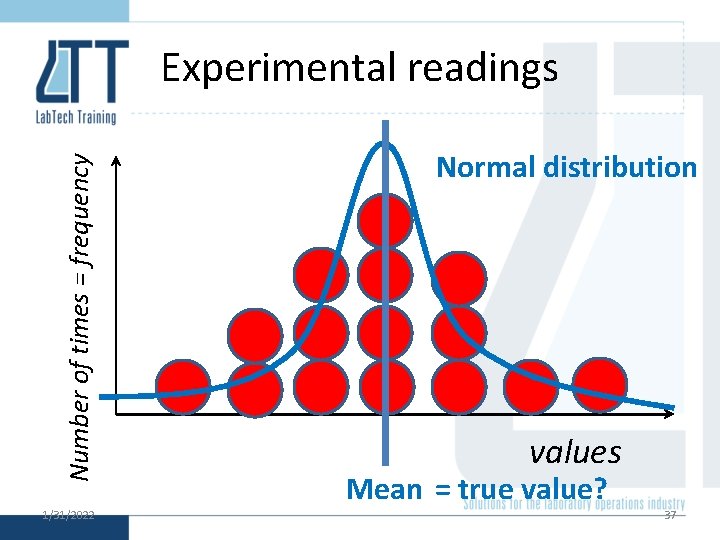
Number of times = frequency Experimental readings 1/31/2022 Normal distribution values Mean = true value? 37
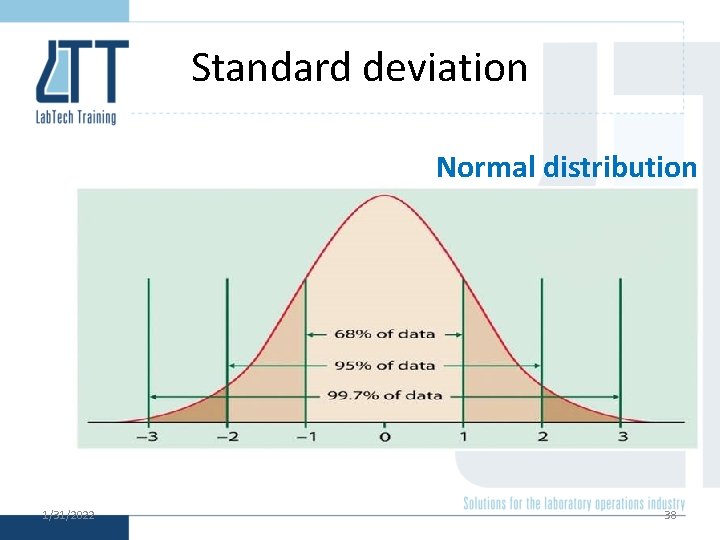
Standard deviation Normal distribution 1/31/2022 38
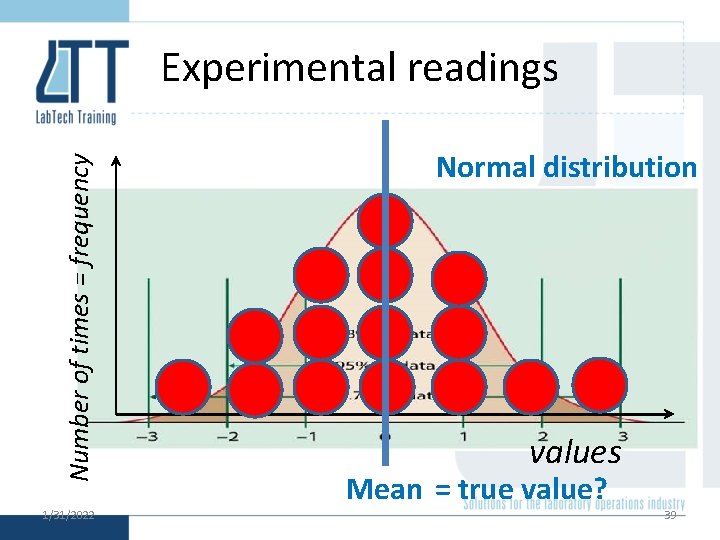
Number of times = frequency Experimental readings 1/31/2022 Normal distribution values Mean = true value? 39

Accuracy and Precision 1/31/2022 40
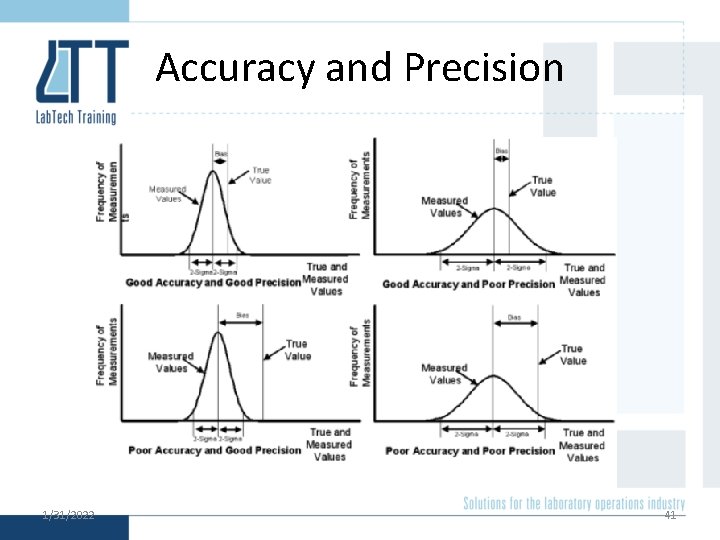
Accuracy and Precision 1/31/2022 41

Practical • Today we are going to investigate the accuracy and precision for a bulb pipette, burette and a volumetric flask. 1/31/2022 42

Summary • • • Mass, volume, density Density of water & temperature dependence Density of water & salinity dependence Standards – documents Analytical glassware Normal distribution, mean and standard dev. 1/31/2022 43 43
- Slides: 43