Laboratory and Clinical Services Clinical Biometrics CMC DMPK

Laboratory and Clinical Services Clinical Biometrics CMC DMPK Bioanalytical 1

15+ Years of Strong Growth 2004 Company Founded by Dr. Song Li 2001 Opened Labs in Green Valley PA Opened Shanghai, China office 2006 2008 Began Phase I Clinical Operations 2014 Expanded & Relocated HQ to Exton, PA 2012 March 2015 Acquired BDM with Data Management and Biostats Services Strategic Investment Agreement with Tigermed 2015 June 2015 Opened new clinical site in Secaucus, NJ Sister CDMO company Frontida acquired in Philadelphia, PA 2016 Staff number grew from 1 to 500+ 2
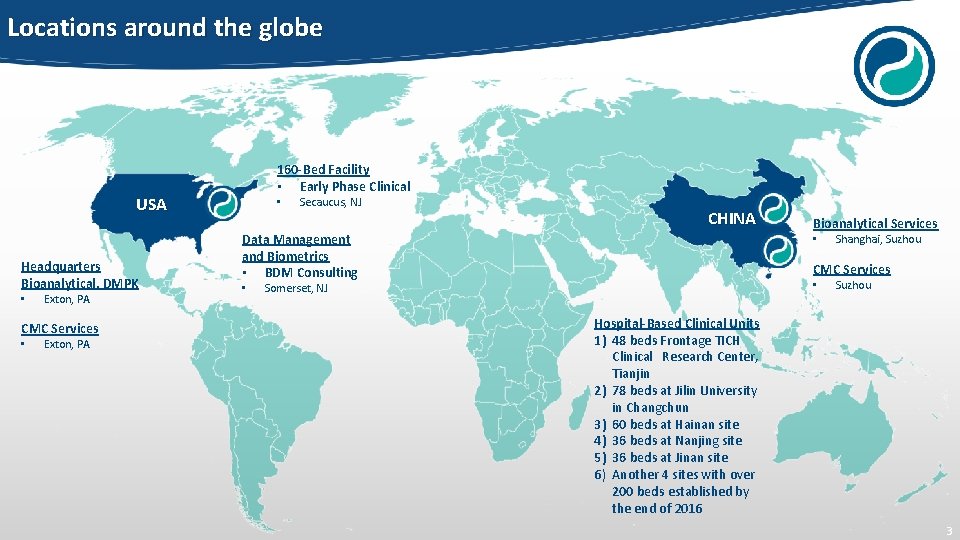
Locations around the globe 160 -Bed Facility • Early Phase Clinical USA Headquarters Bioanalytical, DMPK • Exton, PA CMC Services • Exton, PA • Secaucus, NJ CHINA Data Management and Biometrics • BDM Consulting • Bioanalytical Services • Shanghai, Suzhou CMC Services • Somerset, NJ Suzhou Hospital-Based Clinical Units 1) 48 beds Frontage TICH Clinical Research Center, Tianjin 2) 78 beds at Jilin University in Changchun 3) 60 beds at Hainan site 4) 36 beds at Nanjing site 5) 36 beds at Jinan site 6) Another 4 sites with over 200 beds established by the end of 2016 3

Service Offerings Laboratory Services Clinical Services DMPK BIOANALYTICAL CMC CLINICAL BIOMETRICS Insight. Expertise. Quality. Agility. Intelligence. • Rapid PK; In Vitro ADME • Mass Balance • Early Discovery to Late Development • Metabolite Identification • Biomarker Services • Preclinical and Clinical Support • Global Bioanalytical Services (50+ LC-MS/MS) • Biologics Assay (Antibody Drug, ADC, Oligonucleotide) • Analytical • API Synthesis • CTM Manufacturing • Formulation Development • Multiple Clinical Centers in US and China • Phase I, SAD, MAD • First to File Packages • BA/BE, Food Effect, DDI • CDISC-Compliant Datasets • SAS Programming • Preclinical-Phase IV Development Support • EDC & Data Management It’s how your candidates become real contenders. It’s how you get from the molecule to the market. It’s how you get from formulation to the pharmacy. It’s what gets you from the clinic to the cabinet. It’s how you turn data into decisions. 4
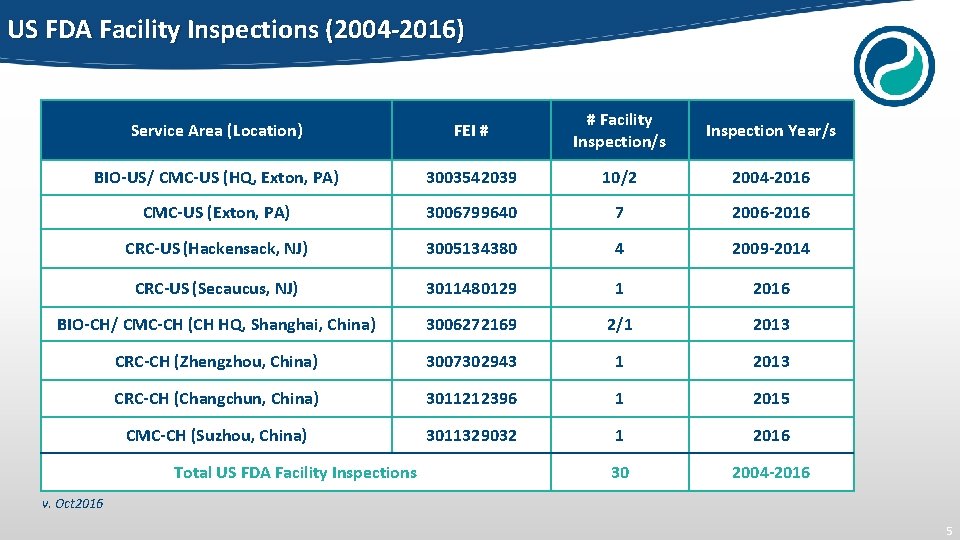
US FDA Facility Inspections (2004 -2016) Service Area (Location) FEI # # Facility Inspection/s Inspection Year/s BIO-US/ CMC-US (HQ, Exton, PA) 3003542039 10/2 2004 -2016 CMC-US (Exton, PA) 3006799640 7 2006 -2016 CRC-US (Hackensack, NJ) 3005134380 4 2009 -2014 CRC-US (Secaucus, NJ) 3011480129 1 2016 BIO-CH/ CMC-CH (CH HQ, Shanghai, China) 3006272169 2/1 2013 CRC-CH (Zhengzhou, China) 3007302943 1 2013 CRC-CH (Changchun, China) 3011212396 1 2015 CMC-CH (Suzhou, China) 3011329032 1 2016 30 2004 -2016 Total US FDA Facility Inspections v. Oct 2016 5
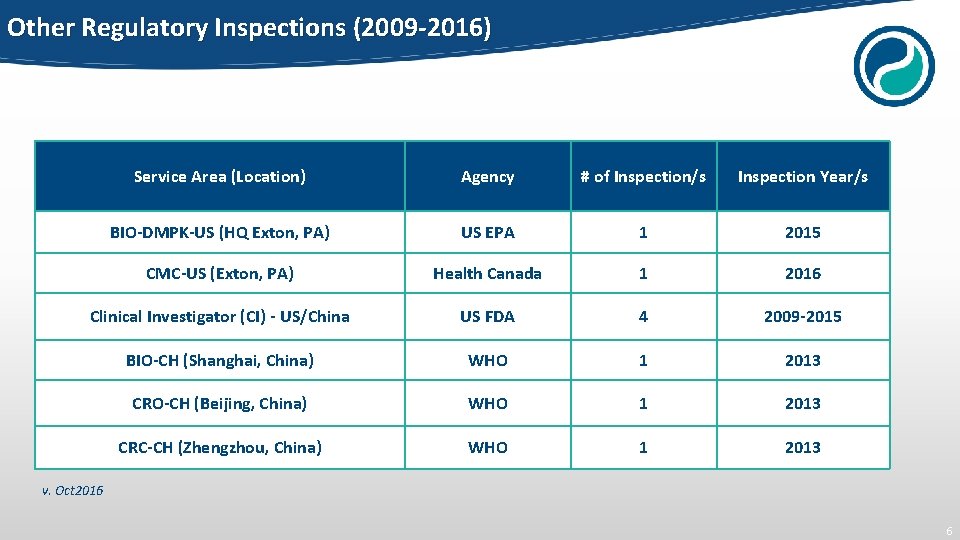
Other Regulatory Inspections (2009 -2016) Service Area (Location) Agency # of Inspection/s Inspection Year/s BIO-DMPK-US (HQ Exton, PA) US EPA 1 2015 CMC-US (Exton, PA) Health Canada 1 2016 Clinical Investigator (CI) - US/China US FDA 4 2009 -2015 BIO-CH (Shanghai, China) WHO 1 2013 CRO-CH (Beijing, China) WHO 1 2013 CRC-CH (Zhengzhou, China) WHO 1 2013 v. Oct 2016 6

Why Frontage? Technical Expertise With 70% of our scientists holding advanced degrees and our depth & breadth of experience, we can solve complex problems Highest Standards We have an unparalleled compliance and quality track record with 29 FDA Inspections across GXP Flexibility We value agile project management, & a commitment to completing projects on time and on budget Innovation Our $40 M facility investment and 450 FTE in USA & China give us the latest equipment and unparalleled access to China 7

Laboratory Services DMPK 8
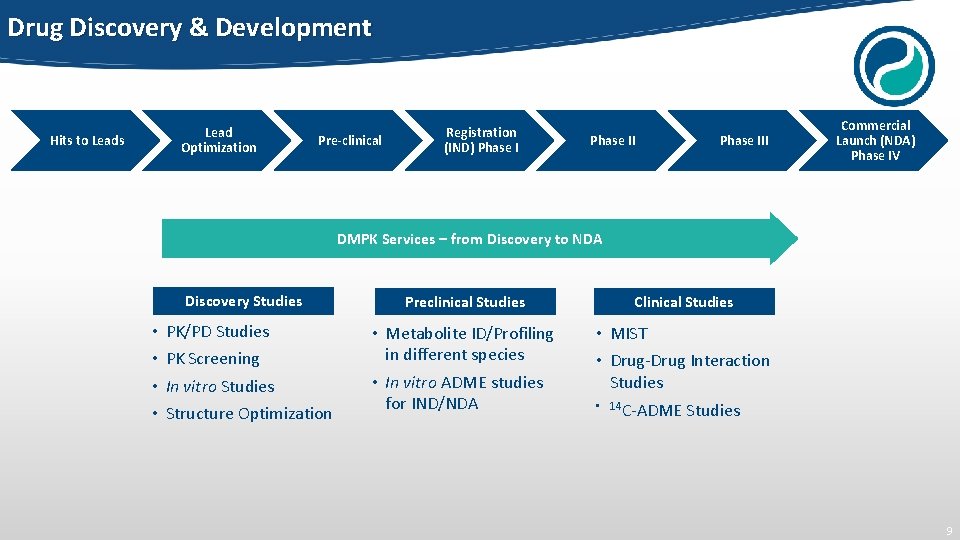
Drug Discovery & Development Lead Optimization Hits to Leads Pre-clinical Registration (IND) Phase III Commercial Launch (NDA) Phase IV DMPK Services – from Discovery to NDA Discovery Studies • • PK/PD Studies PK Screening In vitro Studies Structure Optimization Preclinical Studies Clinical Studies • Metabolite ID/Profiling in different species • In vitro ADME studies for IND/NDA • MIST • Drug-Drug Interaction Studies • 14 C-ADME Studies 9
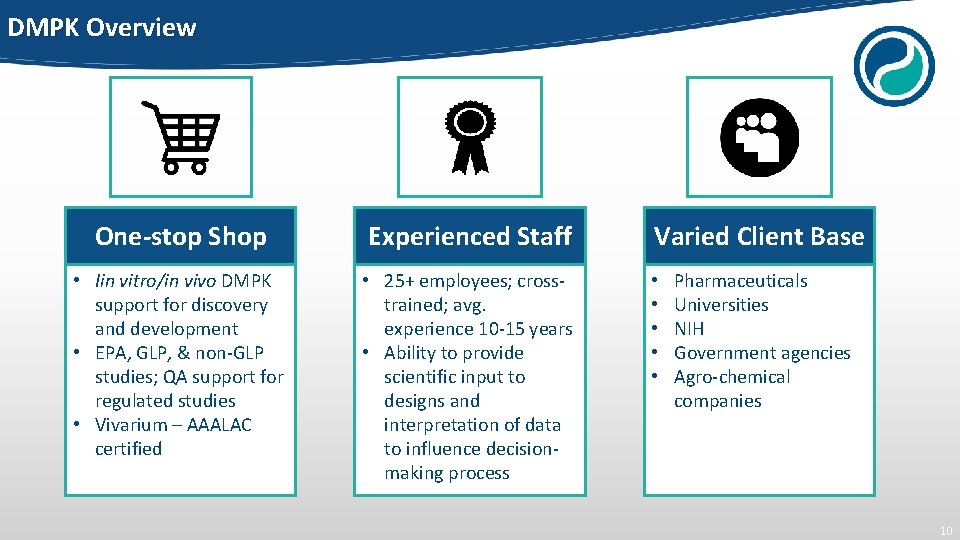
DMPK Overview One-stop Shop Experienced Staff • Iin vitro/in vivo DMPK support for discovery and development • EPA, GLP, & non-GLP studies; QA support for regulated studies • Vivarium – AAALAC certified • 25+ employees; crosstrained; avg. experience 10 -15 years • Ability to provide scientific input to designs and interpretation of data to influence decisionmaking process Varied Client Base • • • Pharmaceuticals Universities NIH Government agencies Agro-chemical companies 10

Laboratory Services Bioanalytical 11

Global Bioanalytical Operations • ~ 130 Staff in USA • 1, 400+ Mostly • 80, 000+ Sq. Ft. • 500, 000+ Samples and China • Majority of Scientists Hold Advanced Degrees Team & Facilities Proprietary Assays (Annual Capacity) • Mostly Proprietary Assays • 80+ Available Assays • 200, 000+ Samples • Ongoing Customized Assays and Therapeutic Focus Biologics Biomarker (Annual Capacity) • 58 LC-MS/MS Small Molecule 12

Small Molecule Capabilities Method Development, Transfer & Validation Sample Analysis • Nonclinical toxicokinetic/pharmac okinetic screening • Clinical PK/bioavailability studies • Bioequivalence studies • Therapeutic drug monitoring studies • Drug-drug interaction studies • Protein binding studies Expertise in New Sample Collection Procedure Development • DBS sampling • Micro-sampling techniques • Analyte stabilization procedures Expertise in Labile Compound Method Development • Enzymatic instability • Physical/chemical instability 13
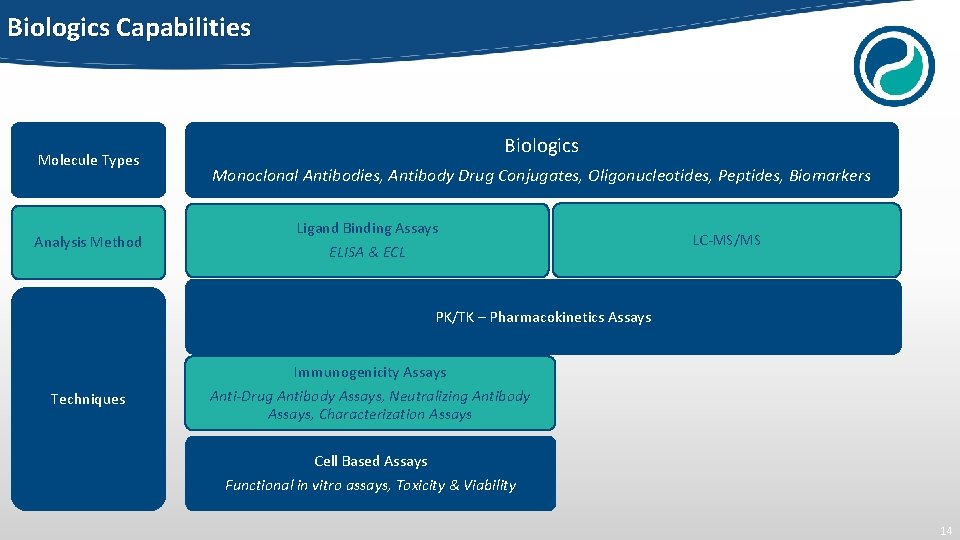
Biologics Capabilities Molecule Types Analysis Method Biologics Monoclonal Antibodies, Antibody Drug Conjugates, Oligonucleotides, Peptides, Biomarkers Ligand Binding Assays ELISA & ECL LC-MS/MS PK/TK – Pharmacokinetics Assays Techniques Immunogenicity Assays Anti-Drug Antibody Assays, Neutralizing Antibody Assays, Characterization Assays Cell Based Assays Functional in vitro assays, Toxicity & Viability 14

Laboratory Services CMC 15
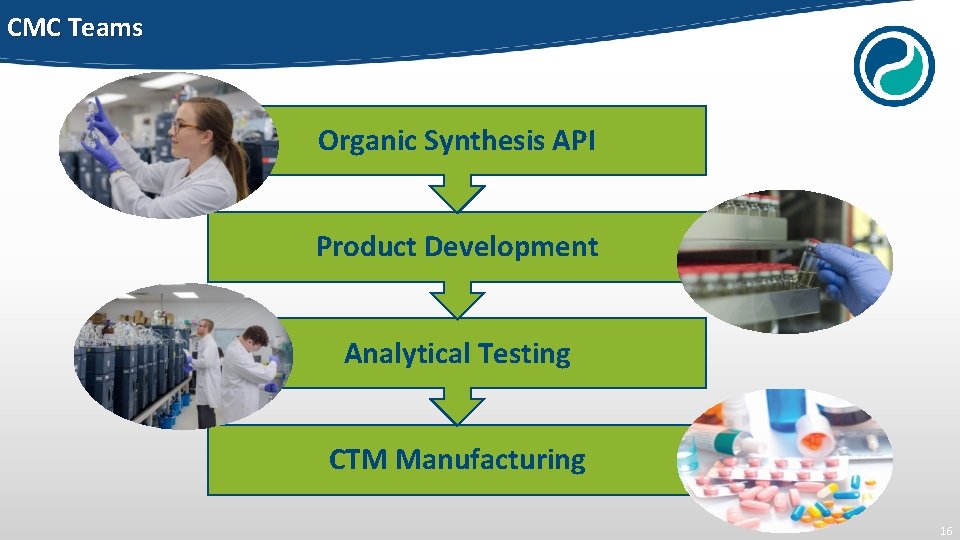
CMC Teams Organic Synthesis API Product Development Analytical Testing CTM Manufacturing 16

Product Development and CTM Manufacturing Work with > 170 clients for > 170 compounds and >370 GMP batches IND: Formulation Development /CTM/IND (OSD, Topical, Sterile) High Potency Suite for CTM Manufacturing ANDA: Reverse Engineering/Formulation Dev/Tech Transfer • Formulation and Process Development (Qb. D format) Sterile Suite for CTM Manufacturing (Phase 1) Proof of Concept Studies in Animals via Frontage and 3 rd Party Facilities Consumer Products: OTC reformulation DEA Manufacturing License for Controlled Substance (CI-V) Integrated Services with Frontage Clinical Center 17

Analytical Services Method Development, Validation and Transfer • Developed and/or validated over 1, 350 methods (~100 methods/year) • HPLC (20), UPLC (22), GC (2), GC/MS, IC, HPLC/MS, UPLC/MS, ICP/OES, ICP/MS • Dissolution (16) • Diffusion IVRT and Skin Permeation IVPT testing (48 Franz cells) Unknown Impurity Identifications • Fraction collection, lyophilization, LC-MS-MS, NMR Extractable/Leachable Studies • GC/MS, LC/MS, ICP-OES or MS API and Finished Product Release Testing Commercial Product Stability Testing and Storage GLP Dosing Sample Analysis DEA License for Testing Controlled Substances Schedule I-V 18

GMP Sample Stability Testing FDA-Inspected Labs for commercial stability study Over 4, 500 cubic feet of storage (6 walk-in 8’x 8’ rooms; 6 reach-in chambers) Typical ICH storage conditions & WHO (Zone IVb hot/higher humidity) • 25 o. C/60%RH, 30 o. C/65%RH, 40 o. C/75%RH, 30 o. C/75%RH Low Humidity for Semi Permeable Containers (Ophthalmic Products) • 5 o. C, 25 o. C/40%RH, 40 o. C/25%RH Low Temperature for Parenteral & Lyophilized Products • -70 o. C, -20 o. C, 5 o. C, 25 o. C/60%RH ICH compliant photo-stability chamber 24/7 e-monitoring system/Emergency back-up power generator for continuous operations 19

Clinical Services Clinical 20

Clinical Research Facility – Secaucus, NJ • State of the Art Facility • Less than 5 miles from NYC • ½ mile from local medical center • Near major airports and mass transit • 36, 000 sq. ft. 160 Beds • Multiple units can be combined to accommodate any size study • Telemetry Unit • Safe drug handling including controlled substance management (Schedule 2 -5 license) 21

Clinical Research at A Glance Seven Early Phase Clinical Facilities in the U. S. and China Data Management and Biometrics EDC support & statistical programming, including e. Submission and CDISC support Experience with novel compounds and access to a diverse range of subject populations and patients Clinical Study Design and Execution Medical Writing and Regulatory Support Early phase focus, monitoring and study management for multicenter studies (PK/PD) Clinical protocol development, Investigator’s brochures, CSR 22
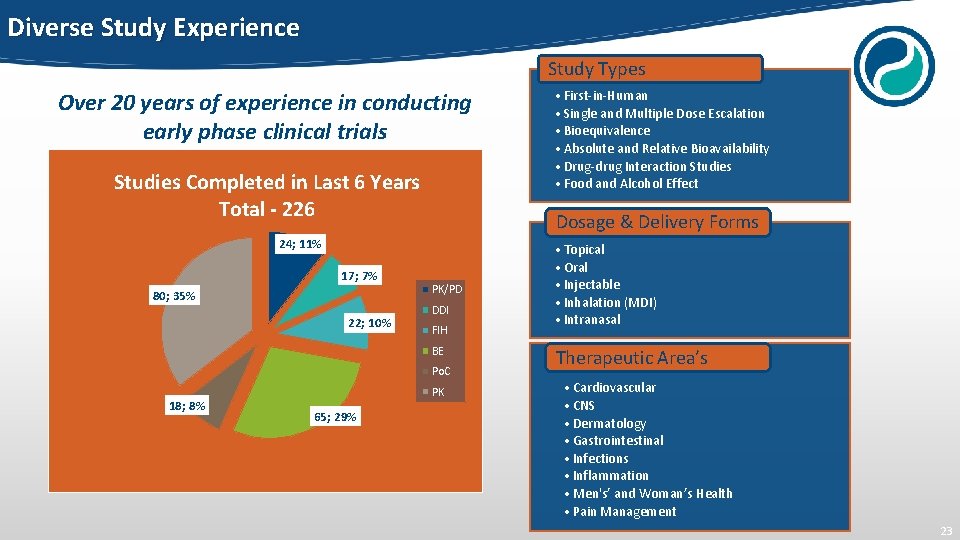
Diverse Study Experience Study Types Over 20 years of experience in conducting early phase clinical trials Studies Completed in Last 6 Years Total - 226 Dosage & Delivery Forms 24; 11% 17; 7% 80; 35% 22; 10% PK/PD DDI FIH BE Po. C 18; 8% PK 65; 29% • First-in-Human • Single and Multiple Dose Escalation • Bioequivalence • Absolute and Relative Bioavailability • Drug-drug Interaction Studies • Food and Alcohol Effect • Topical • Oral • Injectable • Inhalation (MDI) • Intranasal Therapeutic Area’s • Cardiovascular • CNS • Dermatology • Gastrointestinal • Infections • Inflammation • Men's’ and Woman’s Health • Pain Management 23

Clinical Services Biometrics 24

Biometrics Services to Rapidly Compile the Clinical Study Report Data Management • Rigorous SOPs for Regulatory Standards Adherence • Optimized DM Solutions - Pair Technology to Study • Reliable, High. Quality Delivery Supported by Experienced Teams Biostatistics and Programming • Biostatistics Consultation and Analyses by Staff statisticians with more than 20 years of industry experience • Statistical Programming Medical Writing Regulatory • Pharmacokinetic (PK) Analysis led by Ph. D Clinical Pharmacologist with 25 years of experience • Insight into Clinical Development Plans, Study Designs and the Regulatory • Medical Writing from to Clinical study reports • Approval Path Led by former FDA reviewer 25

Snapshot of China Operations Clinical CMC Bioanalytical 26

Frontage China Operations Headquartered in Zhangjiang Hi-Tech Park, Shanghai Labs in function from 2006 (the first global standard GLP lab in China) Director and above levels are trained abroad and have multiple years working experience in Pharmaceutical industries Multiple times successfully passed FDA and WHO site inspections (GLP/GMP/GCP) 27

Frontage China Services Bioanalytical • • • >26, 000 sqf facilities located in Shanghai and Suzhou >50 Scientists; >70% have advanced degrees 16 LC/MS/MS with Watson. LIMS system Clinical CMC/CTM • >30, 000 sqf facilities located in Suzhou • • US-China method cross transfer and validation abilities • Support sample analysis for small molecules drugs and biologics • • >40 Scientists; >70% have advanced degrees Hospital-based early phase clinical centers in different regions • Analytical method dev/val; stability and release testing >250 beds phase I capacity currently; about 600 beds mi 2017 • Various therapeutic area experience • DM/Bio. Stat fully support functions • Late phase clinical trial capabilities Formulation development – various dosage forms Clinical trial material manufacturing 28

Global Headquarter Locations U. S. Headquarters China Headquarters 29
- Slides: 29