Laboratory 06 MOLECULAR GEOMETRY AND POLARITY Background Lewis
Laboratory 06 MOLECULAR GEOMETRY AND POLARITY

Background- Lewis structure Diagrams that show the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule A Lewis structure can be drawn for any covalently bonded molecule, as well as coordination compounds.

Construction of Lewis Structures Two Rules 1. Total # of valence electrons – the total number of valence electrons must be accounted for, no extras, none missing. 2. Octet Rule – every atom should have an octet (8) electrons associated with it. Hydrogen should only have 2 (a duet).

Determining the number of valence electrons Full d-orbitals do not count as valence electrons. They belong to the inner shell. For example: Pb [Xe]4 f 145 d 106 s 26 p 2 This is four (4) valence electrons. The 5 d is part of the inner shell (n=5) which is full.

The total number of available valence electrons is just the sum of the number of valence electrons that each atom possesses (ignoring d-orbital electrons) For H 2 O, The total number of valence electrons = 2 x 1 (each H is 1 s 1) + 6 (O is 2 s 22 p 4) =8 For CO 2 The total number of valence electrons = 4 (C is 2 s 22 p 2) + 2 * 6 (O is 2 s 22 p 4) = 16

Central Atom In a molecule, there are only 2 types of atoms: 1. 2. “central” – bonded to more than one other atom. “terminal” – bonded to only one other atom. Almost always the least electronegative atom is the central atom. For example, in Cl. O 2, the Cl is the central atom; in SF 5 the S is the central atom. Hydrogen never is the central atom. It forms only one bond, so it must generally be in the outer layer of atoms. You can have more than one central atom in a molecule.

Bonds are pairs of shared electrons. Each bond has 2 electrons in it. You can have multiple bonds between the same 2 atoms. For example: C-O C=O C O Each of the lines represents 1 bond with 2 electrons in it.

Lewis Dot Structure Each electron is represented by a dot in the structure. : Cl: ¨ That symbol with the dots indicate a chlorine atom with 7 valence electrons.

Drawing Lewis Dot Structures 1. Determine the total number of valence electrons. 2. Determine which atom is the “central” atom. 3. Stick everything to the central atom using a single bond. 4. Fill the octet of every atom by adding dots. 5. Verify the total number of valence electrons in the structure. 6. Add or subtract electrons to the structure by making/breaking bonds to get the correct # of valence electrons. 7. Check the “formal charge” of each atom.

Formal Charge of an Atom Formal charge = number of valence electrons – number of bonds – number of non-bonding electrons.

Dot structure for H 2 O 1. Total number of valence electrons: 6 + (2 x 1) =8 2. Central Atom – O 3. Stick all terminal atoms to the central atom using a single bond.
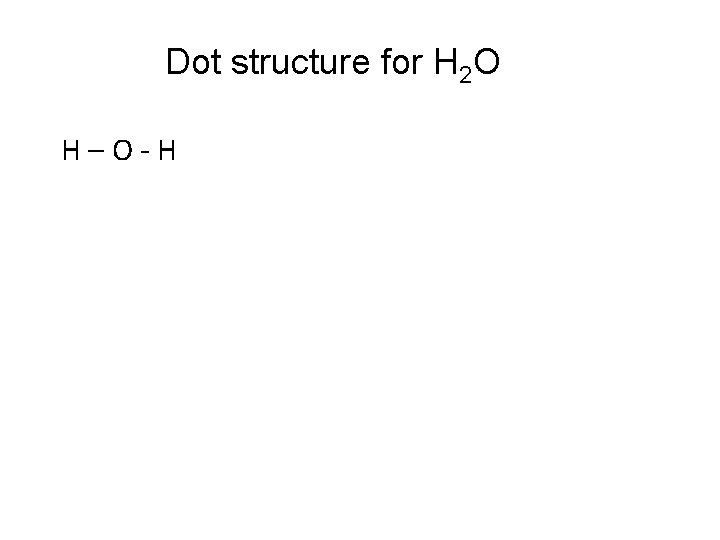
Dot structure for H 2 O H–O-H
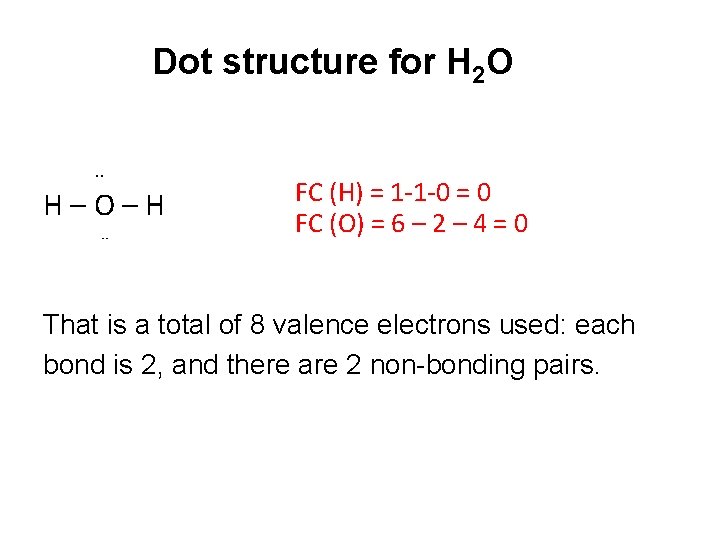
Dot structure for H 2 O. . H–O–H ¨ FC (H) = 1 -1 -0 = 0 FC (O) = 6 – 2 – 4 = 0 That is a total of 8 valence electrons used: each bond is 2, and there are 2 non-bonding pairs.

Expanded Octets Example PCl 5: . . : Cl: . . : Cl – P - Cl : ¨ | ¨ : Cl: ¨ Total valence e- = 40 FC(P) = 5 – 0 =0 FC (Cl) = 7 – 1 – 6 = 0

Background - Covalent Bonds • The simplest covalent bond is that in H 2 – the single electrons from each atom combine to form an electron pair – the shared pair functions in two ways simultaneously; it is shared by the two atoms and fills the valence shell of each atom • The number of shared pairs – one shared pair forms a single bond – two shared pairs form a double bond – three shared pairs form a triple bond

Background – Polarity Polar and Nonpolar Covalent Bonds • Although all covalent bonds involve sharing of electrons, they differ widely in the degree of sharing • We divide covalent bonds into – nonpolar covalent bonds – polar covalent bonds

An example of a polar covalent bond is that of H-Cl – the difference in electronegativity between Cl and H is 3. 0 - 2. 1 = 0. 9 – Polarity can be shown by using the symbols d+ and -, or dby using an arrow with the arrowhead pointing toward the negative end a plus sign on the tail of the arrow at the positive end d+ d. H Cl
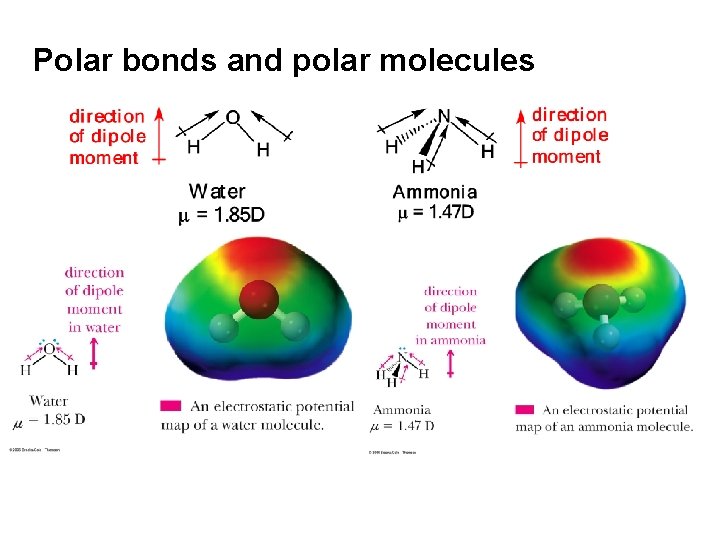
Polar bonds and polar molecules

Laboratory 07 QUALITATIVE ANALYSIS : TESTING THE SOLUBILITY RULES

Background : Ionic Compounds 1. Most ionic compounds are also called salts. 2. Most ionic compounds exist as solids and many dissolve to form aqueous solutions. Example : Ag. Cl insoluble in water but Ag. NO 3 is soluble 3. An ionic compound is made up of a metal and a nonmetal; metals are located on the left side of the periodic table and nonmetals are on the right side. 4. The cation (positive ion) is written first followed by the anion (negative ion).
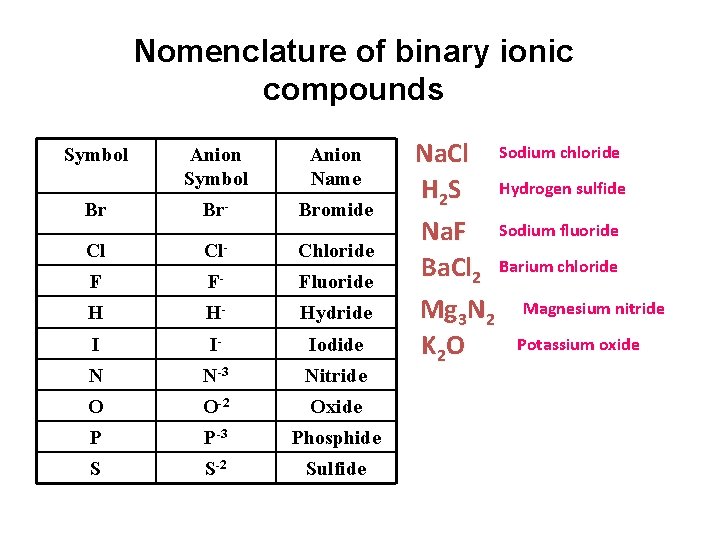
Nomenclature of binary ionic compounds Symbol Anion Name Br Br- Bromide Cl Cl- Chloride F F- Fluoride H H- Hydride I I- Iodide N N-3 Nitride O O-2 Oxide P P-3 Phosphide S S-2 Sulfide Na. Cl Sodium chloride H 2 S Hydrogen sulfide Na. F Sodium fluoride Ba. Cl 2 Barium chloride Mg 3 N 2 Magnesium nitride Potassium oxide K 2 O

Polyatomic anions NO 3 - = nitrate NO 2 - = nitrite SO 4 2 - = sulfate SO 32 - = sulfite PO 43 - = phosphate CO 32 - = carbonate HCO 3 - = hydrogen carbonate or bicarbonate OH- = hydroxide CN- = cyanide C 2 H 3 O 2 - = acetate C 2 O 42 - = oxalate

Na. HCO 3 = sodium hydrogen carbonate or sodium bicarbonate K 2 SO 3 = potassium sulfite Mg. SO 4 = magnesium sulfate KCN = potassium cyanide H 2 PO 4 = hydrogen phosphate Ca(OH)2 = calcium hydroxide NH 4 NO 3 = ammonium nitrate Zn(NO 3)2 = zinc nitrate Li 3 PO 4 = lithium phosphate HNO 3 = hydrogen nitrate

Solubility Rules for Ionic Compounds in Water Soluble Ionic Compounds Insoluble Ionic Compounds 1. All common compounds of Group 1 A 1. All common metal hydroxides are ions (Li+, Na+, K+, etc. ) and ammonium insoluble, except those of Group 1 A and ion (NH 4+) are soluble. the larger members of Group 2 A (beginning with Ca 2+). 2. All common nitrates (NO 3 -), acetates 2. All common carbonates (CO 32 -) and (CH 3 COO-), and most perchlorates (Cl. O 4 phosphates (PO 43 -) are insoluble, except -) are soluble. those of Group 1 A and NH 4+. 3. All common chlorides (Cl-), bromides 3. All common sulfides are insoluble (Br-), and iodides (I-) are soluble, except those of Group 1 A, Group 2 A, those of Ag+, Pb 2+, Cu+, and Hg 2+. All and NH 4+. common fluorides (F-)are soluble, except those of Pb 2+ and Group 2 A. 4. All common sulfates (SO 42 -) are soluble, except those of Ca 2+, Ba 2+, Ag+, and Pb 2+.

Precipitation reactions General form: Solution A + Solution B → Insoluble Solid C + Solution D. In a precipitation reaction two solutions are mixed together to produce an insoluble solid which is called the precipitate. This type of reaction is also called a double displacement reaction Lead nitrate(aq) + Potassium iodide(aq) → Lead iodide(s) + potassium nitrate(aq) Pb(NO 3)2(aq) + 2 KI(aq) → Pb. I 2(s) + KNO 3(aq)

Barium chloride(aq) + Sodium sulfate(aq) → Barium sulfate(s) + Sodium chloride(aq) Ba. Cl 2(aq) + Na 2 SO 4(aq) → Ba. SO 4(s) + Na. Cl(aq) Copper sulfate(aq) + Sodium hydroxide(aq) → Copper hydroxide(s) + Sodium sulfate(aq) Cu. SO 4(aq) + 2 Na. OH(aq) → Cu(OH)2(s) + Na 2 SO 4(aq)

Mercury(II) nitrate(aq) + Potassium iodide(aq) → Mercury iodide(s) + Potassium nitrate Hg(NO 3)2(aq) + 2 KI(aq) → Hg. I 2(s) + KNO 3(aq) Silver nitrate(aq) + sodium chloride(aq) →Silver chloride(s) + sodium nirate(aq) Ag. NO 3(aq) + Na. Cl(aq) → Ag. Cl(s) + Na. NO 3(aq)
- Slides: 27