Laboratoire de Physique de la Matire Condense Laboratoire

Laboratoire de Physique de la Matière Condensée Laboratoire mixte de l’école Polytechnique et du CNRS (UMR 7643) 3 Novembre 2005 L’interaction Agrégat métallique - Molécule au cœur des processus physicochimiques environnementaux D. Bazin 1
Objectiv of the lecture II Cluster Behaviour Adsorption Mode 2

PLAN I. X-ray Absorption Spectroscopy II. Interaction between nanometer scale metallic cluster & NO 3
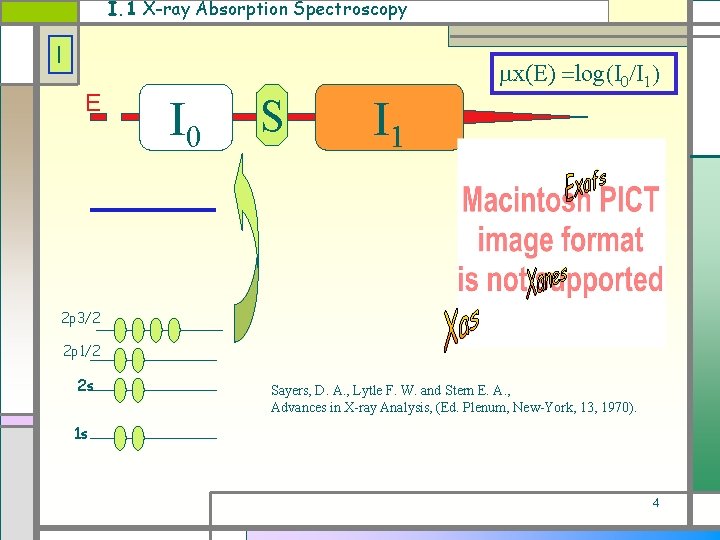
I. 1 X-ray Absorption Spectroscopy I E x(E) log I 0 S I 1 2 p 3/2 2 p 1/2 2 s Sayers, D. A. , Lytle F. W. and Stern E. A. , Advances in X-ray Analysis, (Ed. Plenum, New-York, 13, 1970). 1 s 4

I. 1 The associated formula I (k) = j Nj/k. R 2 fj(k)exp(-Rj/ )exp(-2 j 2 k 2)sin(2 k. Rj + j(k)) j Electronic terms fj(k), Structural terms Nj , Rj , j. 5

I. 2 Nanometer scale metallic cluster & Xas I • D. Bazin, D. Sayers, J. Rehr, Comparison between Xas, Awaxs, Asaxs & Dafs applied to nanometer scale metallic clusters. J. Phys. Chem. B 101, 11040 (1997). • D. Bazin, D. Sayers, J. Rehr, C. Mottet Numerical simulation of the Pt LIII edge white line relative to nanometer scale clusters, J. of Phys. Chem. B 101, 5332 (1997). • D. Bazin, J. Rehr, Limits and advantages of X-ray absorption near edge structure for nanometer scale metallic clusters. J. Phys. Chem. B 107, 12398 (2003). 6
I. 3 Some examples I 1%wt Pt/oxide Impregnation Cl Pt O Calcination Reduction O Pt O O Pt Pt • Xas studies of bimetallic Pt-Re(Rh)/Al 2 O 3 catalysts in the first stages of preparation. D. Bazin et al. , J. of Cat. 110, 209 (1988). • Bimetallic reforming catalysts : Xas of the particle growing process during the reduction. D. Bazin et al. , J. Cat. 123, 86 (1990). • In situ high temperature and high pressure Exafs studies of Pt/ Al 2 O 3 catalysts : Part I. N. S. Guyot-Sionnest et al. , Cat. Let 8, 283 (1991). • In situ high temperature and high pressure Exafs studies of Pt/ Al 2 O 3 catalysts : Part II. N. S. Guyot-Sionnest et al. , Cat. Let 8, 297 (1991). • Investigation of dispersion and localisation of Pt species in mazzite using Exafs. A. Khodakov et al. J. of Phys. Chem. 101, 766 (1996). • In situ study by Xas of the sulfuration of industrial catalysts : the Pt & Pt. Re/Al 2 O 3 system. A. Bensaddik et al. , Applied Cat. A 162, 171, (1997). • Xas of electronic state correlations during the reduction of the bimetallic Pt. Re/Al 2 O 3 system. D. Bazin et al. , J. of Synchrotron Radiation 6, 465, 1999. • Influence of the H 2 S/H 2 ratio and the temperature on the local order of Pd atoms in the case of a highly dispersed multimetallic catalyst : Pd-Ni-Mo/Al 2 O 3. D. Bazin et al. , J. de Physique IV, 12 -6, 379, (2002). • Structure & size of bimetallic Pt. Pd clusters in an hydrotreatment catalyst. D. Bazin et al. , Accepted in Oil & Gas Science and Technology – Rev. IFP Reforming …Pt, Pt. Pd, Pt. Rh, Pt. Mo, Pt. Sn, Pt. Re, Pt. Ir, Fischer–Tropsch, Co. Pt, Co. Pd, Co. Ru, 7
I. 4 Nanometer scale metallic cluster & R. S. I • Comparison between Xas & Awaxs applied to monometallic clusters. D. Bazin, D. Sayers, Jpn J. Appl. Phys. 32 -2, 249, 1993. • Comparison between Xas & Awaxs applied to bimetallic clusters. Bazin, D. Sayers, Jpn J. Appl. Phys. 32 -2, 252, 1993. • AWAXS in heterogeneous catalysis Bazin, L. Guczi, J. Lynch, App. Cat. A 226, 87, 2002. D. D. J. D. Grunwaldt et al. , J. of Cat. 213, 291 (2003) L. Drozdova et al. J. Phys. Chem. B 106, 2240 (2002) • Real time in situ Xanes approach to characterise electronic state of nanometer scale entities D. Bazin, L. Guczi, J. Lynch, Rec. Res. Dev. Phys. Chem. 4, 259, 2000. • Soft X-ray absorption spectroscopy and heterogeneous catalysis. Bazin, L. Guczi, App. Cat. A 213/2, 147, 2001. D. • New opportunities to understand heterogeneous catalysis processes through S. R. studies and theoretical calculations of density of states : The case of nanometer scale bimetallic particles Bazin, C. Mottet, G. Treglia, Applied Catalysis A (1 -2), 47 -54, 2000. • New trends in heterogeneous catalysis processes on metallic clusters from S. R. & theoretical studies Bazin, C. Mottet, G. Treglia, J. Lynch, Applied Surf. Sci. 164, 140, 2000. D. D. 8
II. Catalyse De. NOx II II. 1 Introduction II. 2 Behaviours of the metallic clusters Ex : NO/Pt II. 3 NO adsorption on metallic surface II. 4 NO adsorption on metalic clusters (Ru & Pt) II. 5 Remarks from Prof. J. Friedel II. 6 Other experimental results Ir, Rh, Cu, Pt, Pd II. 7 Discussion : Support, Preparation, Cluster size, Temp. II. 8 Some explanations II. 9 Mechanisms Pt, Cu, Rh, Ru, Ir II. 10 CO on metallic surface II. 11 A bridge between surface science and nanoscience : Implications in heterogeneous catalysis : How to select a catalyst LMSPC 9
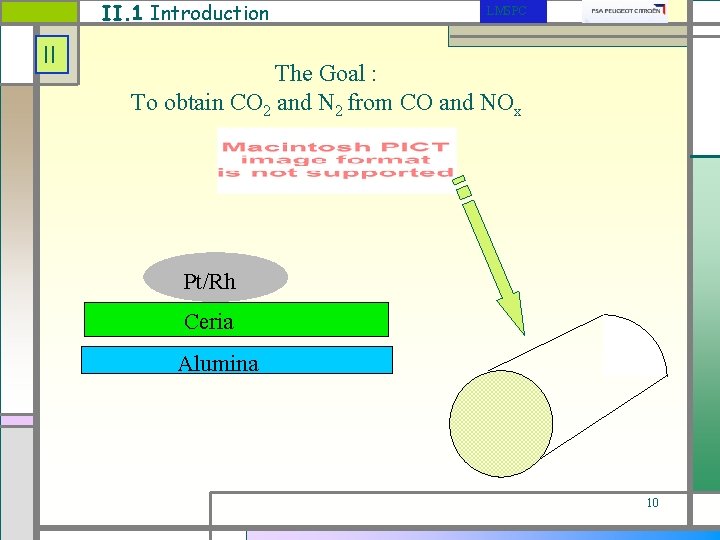
II. 1 Introduction II LMSPC The Goal : To obtain CO 2 and N 2 from CO and NOx Pt/Rh Ceria Alumina 10
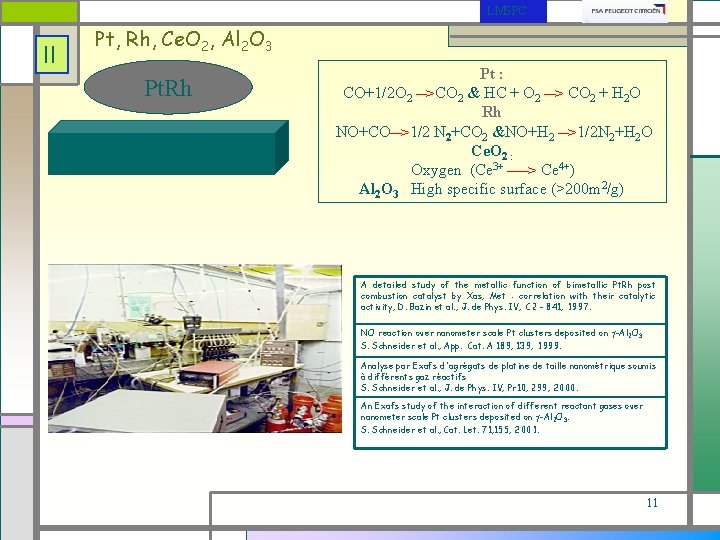
LMSPC II Pt, Rh, Ce. O 2, Al 2 O 3 Pt. Rh Pt : CO+1/2 O 2 __>CO 2 & HC + O 2 __> CO 2 + H 2 O Rh __ NO+CO >1/2 N 2+CO 2 &NO+H 2 __>1/2 N 2+H 2 O Ce. O 2 : Oxygen (Ce 3+ ____> Ce 4+) Al 2 O 3 : High specific surface (>200 m 2/g) A detailed study of the metallic function of bimetallic Pt. Rh post combustion catalyst by Xas, Met : correlation with their catalytic activity, D. Bazin et al. , J. de Phys. IV, C 2 - 841, 1997. NO reaction over nanometer scale Pt clusters deposited on -Al 2 O 3 S. Schneider et al. , App. Cat. A 189, 139, 1999. Analyse par Exafs d'agrégats de platine de taille nanométrique soumis à différents gaz réactifs S. Schneider et al. , J. de Phys. IV, Pr 10, 299, 2000. An Exafs study of the interaction of different reactant gases over nanometer scale Pt clusters deposited on -Al 2 O 3. S. Schneider et al. , Cat. Let. 71, 155, 2001. 11
II. 2 Behaviours of the metallic clusters Ex : NO/Pt II Initial state D. Bazin, L. Guczi, , Recent Res. Dev. Phys. Chem. 3, 387, 1999. D. Bazin, C. Mottet, G. Treglia, J. Lynch, Applied Surf. Sci. 164, 140, 2000. 12
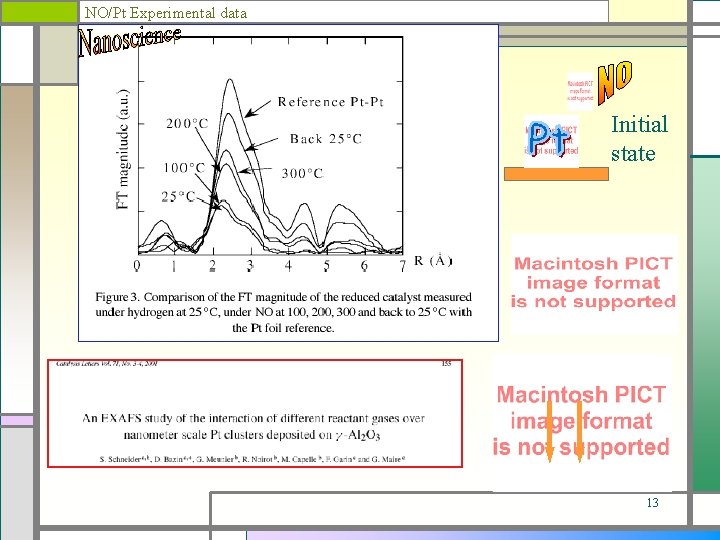
NO/Pt Experimental data Initial state 13

II. 3 NO adsorption on metallic surface II In particularly, in the case of NO, we can distinguish metals for which there is dissociative chemisorption and those for which molecular chemisorption occurs. Ability of transition metal surface to dissociate the NO molecule from G. Broden et al. Sc Ti V Cr Mn Fe Co Ni Cu Y Zr Nb Mo Tc Ru Rh Pd Ag Lu Hf Ta W Re Os Ir Pt Au Metals which are associated with a molecular chemisorption are in red style, the others give rise to a dissociative chemisorption. G. Broden et al. Surf. Sci. 59, 593 (1976). 14
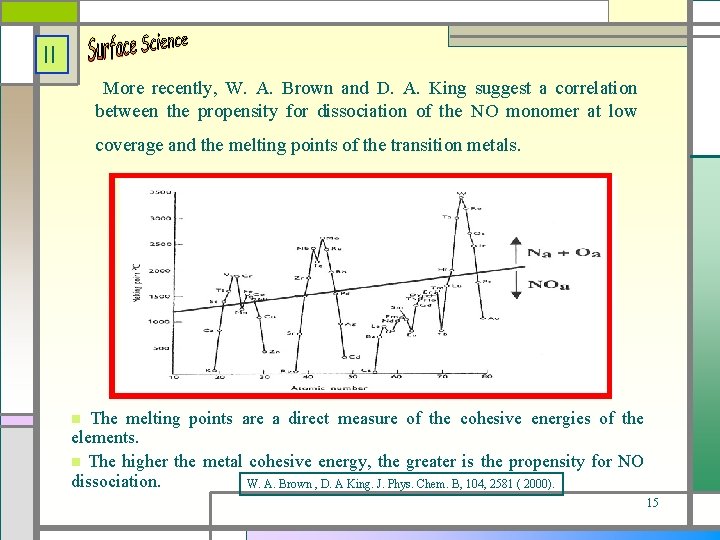
II More recently, W. A. Brown and D. A. King suggest a correlation between the propensity for dissociation of the NO monomer at low coverage and the melting points of the transition metals. The melting points are a direct measure of the cohesive energies of the elements. The higher the metal cohesive energy, the greater is the propensity for NO dissociation. W. A. Brown , D. A King. J. Phys. Chem. B, 104, 2581 ( 2000). 15
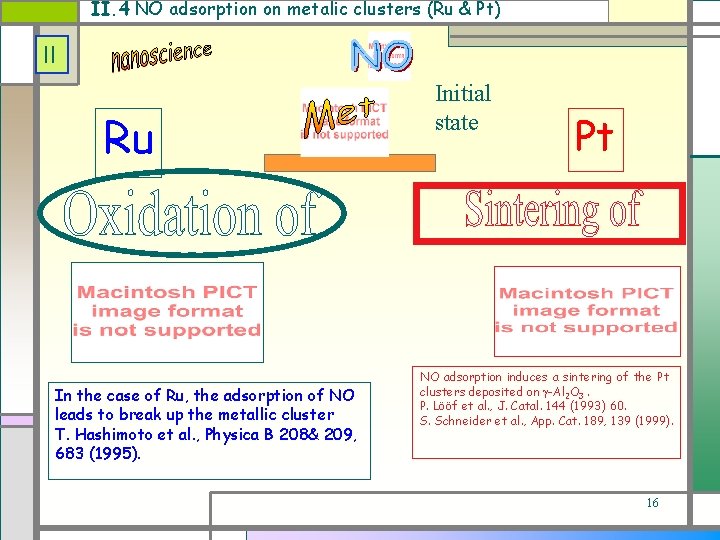
II. 4 NO adsorption on metalic clusters (Ru & Pt) II Ru In the case of Ru, the adsorption of NO leads to break up the metallic cluster T. Hashimoto et al. , Physica B 208& 209, 683 (1995). Initial state Pt NO adsorption induces a sintering of the Pt clusters deposited on -Al 2 O 3. P. Lööf et al. , J. Catal. 144 (1993) 60. S. Schneider et al. , App. Cat. 189, 139 (1999). 16
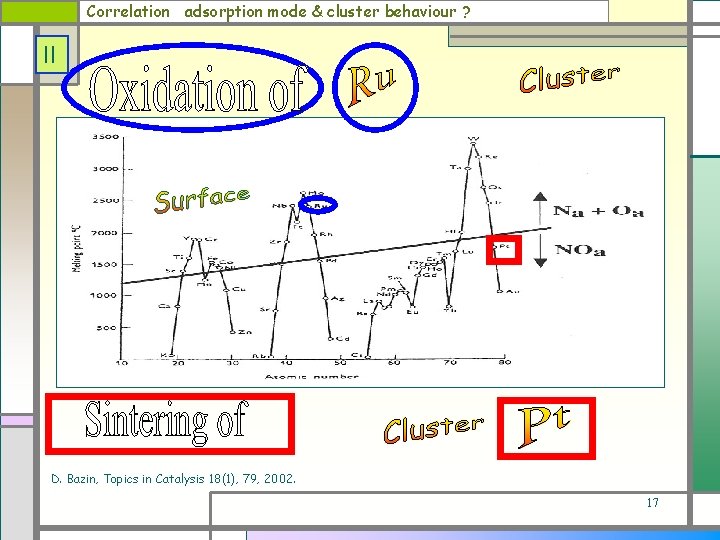
Correlation adsorption mode & cluster behaviour ? II D. Bazin, Topics in Catalysis 18(1), 79, 2002. 17

II. 5 Remarks from Prof. J. Friedel II 18
II. 6 Other experimental results Ir, Rh, Cu, Pt, Pd II At 500°C, almost all NO in contact with Ir 0 was decomposed to N 2 and oxidized Ir 0 to Ir. O 2. C. Wögerbauer, et al. J. of Catalysis, 205, 157 -167 (2002). 19
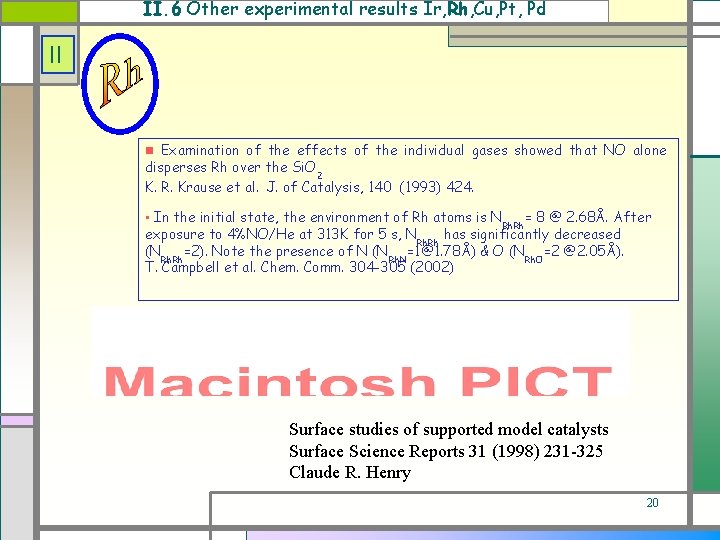
II. 6 Other experimental results Ir, Rh, Cu, Pt, Pd II Examination of the effects of the individual gases showed that NO alone disperses Rh over the Si. O 2 K. R. Krause et al. J. of Catalysis, 140 (1993) 424. • In the initial state, the environment of Rh atoms is N = 8 @ 2. 68Å. After exposure to 4%NO/He at 313 K for 5 s, N has significantly decreased Rh. Rh (N =2). Note the presence of N (N =1@1. 78Å) & O (N =2 @2. 05Å). Rh Rh. N Rh. O T. Campbell et al. Chem. Comm. 304 -305 (2002) Rh. Rh Surface studies of supported model catalysts Surface Science Reports 31 (1998) 231 -325 Claude R. Henry 20
II. 6 Other experimental results Ir, Rh, Cu, Pt, Pd II The adsorption and reaction of NO on Cu clusters deposited on a 5 Å thick Al 2 O 3 film shows strong similarities to its behaviour on Cu single crystals. The STM results show that the Cu clusters grow according to the Volmer-Weber mechanism. Nitric oxide reduction by Cu nanoclusters supported on thin Al 2 O 3 films J. of Catalysis, Volume 22 ( 2004) 204. S. Haq, A. Carew and R. Raval 21
II. 6 Other experimental results Ir, Rh, Cu, Pt, Pd II X. Wang et al. Cat. Today 96 (2004) 11 -20. 22
II. 6 Other experimental results Ir, Rh, Cu, Pt, Pd II Pd(110) Pd(100) Pd(111) In Xafs of the Pd/Mg. O catalyst indicates that neither Pd oxidation nor particle sintering occurs during heating in flowing 1%/NO/He to 300°C. 23
II. 7 Discussion II Discussion • Nature of the support • Preparation procedure • Cluster size • Temperature Combining Solid State Physics Concepts & X-Ray Absorption Spectroscopy to understand heterogeneous catalysis, D. Bazin, D. Sayers, J. Lynch, L. Guczi, G. Treglia, C. Mottet 24
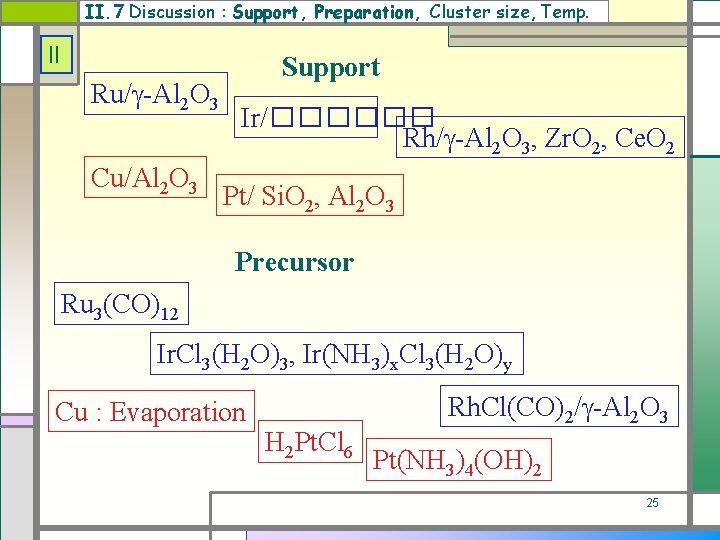
II. 7 Discussion : Support, Preparation, Cluster size, Temp. II Ru/ -Al 2 O 3 Cu/Al 2 O 3 Support Ir/������ Rh/ -Al 2 O 3, Zr. O 2, Ce. O 2 Pt/ Si. O 2, Al 2 O 3 Precursor Ru 3(CO)12 Ir. Cl 3(H 2 O)3, Ir(NH 3)x. Cl 3(H 2 O)y Cu : Evaporation H 2 Pt. Cl 6 Rh. Cl(CO)2/ -Al 2 O 3 Pt(NH 3)4(OH)2 25

II. 7 Discussion : Support, Preparation, Temp. , Cluster size II Effect on the NO adsorption mode Rh NO adsorbs molecularly on Rh at low temperatures and dissociatively at higher temperatures. On Rh[100], molecular NO dominates upon adsorption at 100 K, but heating leads to N 2 and O 2 production in TPD, suggesting dissociation. Ni molecular adsorption takes place on Ni at low temperature and at higher temperatures both molecular and dissociative adsorption are observed. 26
II. 7 Discussion : Support, Preparation, Temp. , Cluster size II 27

Preliminary conclusion II Discussion • Nature of the support • Preparation procedure • Cluster size • Temperature • Pressure • Cluster morphology 28

II. 8 Some explanations : Cohesion energy II How change the Brown diagram when we consider not metallic surface but metallic cluster ? As it can be seen, a significant decrease of the cohesive energy, around 30%, is observed independently the nature of the metal and the morphology of the cluster [a-c]. [a] A. Khoutami, Ph. D thesis, Paris XI University (1993). [b] F. Baletto et al. , Phys. Rev. Let. 84, 5544 (2000). [c] R. A. Guirado-Lopez, Ph. D thesis, Paris XI University (1999). 29
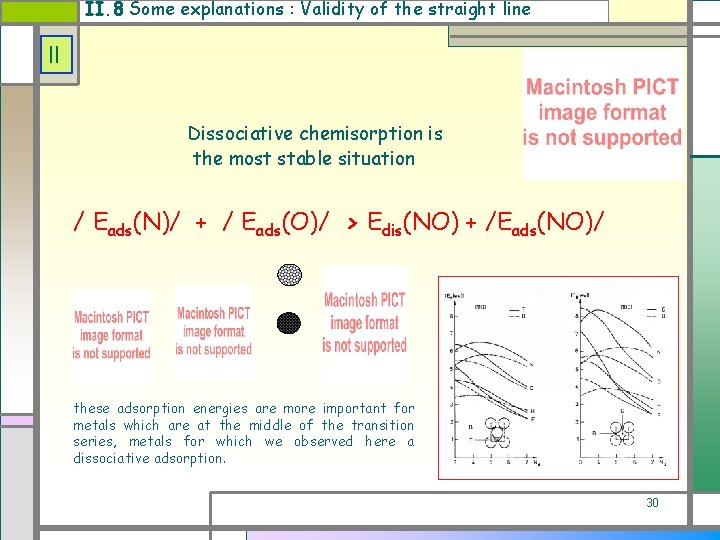
II. 8 Some explanations : Validity of the straight line II Dissociative chemisorption is the most stable situation / Eads(N)/ + / Eads(O)/ > Edis(NO) + /Eads(NO)/ these adsorption energies are more important for metals which are at the middle of the transition series, metals for which we observed here a dissociative adsorption. 30
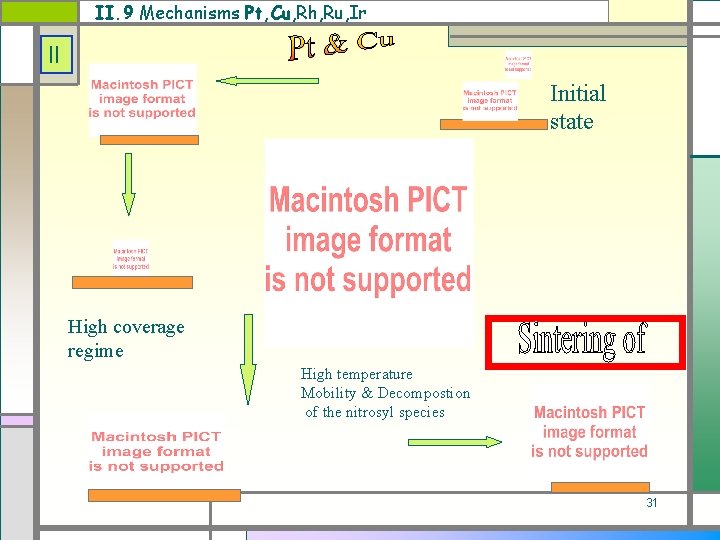
II. 9 Mechanisms Pt, Cu, Rh, Ru, Ir II Initial state High coverage regime High temperature Mobility & Decompostion of the nitrosyl species 31

II. 9 Mechanisms Pt, Cu, Rh, Ru, Ir II Initial state N 2 desorption High temperature Decomposition process 32

Other molecules II / Eads(N)/ + / Eads(O)/ > Edis(NO) + /Eads(NO)/ NO : 6. 50 e. V N 2: 9. 76 e. V Sc Ti V Cr Mn Fe Co Y Zr Nb Mo Tc Ru Rh Lu Hf Ta W Re Os Ir Ni Cu Pd Ag Pt Au O 2 : 5. 12 e. V, N 2 O : 4. 4 e. V 33

II. 10 CO on metallic surface II In particularly, in the case of CO, we can distinguish metals for which there is dissociative chemisorption and those for which molecular chemisorption occurs. Ability of transition metal surface to dissociate the CO molecule from R. B. Anderson Sc Ti V Cr Mn Fe Co Ni Cu Y Zr Nb Mo Tc Ru Rh Pd Ag Lu Hf Ta W Re Os Ir Pt Au Dissociativ T=20°C Non-Dissociativ T=200°C-300°C CO: 11. 09 e. V R. B. Anderson ”The fischer-Tropsch Synthesis”, Chap 4, Academic press, New York, 1984 34

II. 10 Behaviours of the metallic cluster II Initial state High coverage High Temperature On Pt clusters, A. Berko et al. [1] found that CO adsorption induces a significant increase in the initial size of the Pt nanoparticles of 1– 2 nm even at room temperature. This agglomeration preceded by disruption of the smaller Pt nanoclusters at lower CO pressures is explained by CO-assisted Ostwald ripening, in which the mass transport proceeds via surface carbonyl intermediates. Similar results have been obtained in the case of Ru [2], Rh[3, 4], Cu[5] and Pd [6, 7]. [1] [2] [3] [4] [5] [6] [7] A. Berko et al. Surface Science 566568, 337 (2004). T. Mizushima et al. J. Phys. Chem. 94, 4980 (1990). H. F. J. Van't Blik et al. J. Phys. Chem. 87, 2264 (1983). A. Suzuki et al. Angew. Chem. Int. Ed. , 42, 4795 (2003). X. Wang et al. J. Phys. Chem. B 2004, 108, 13667. S. L. Anderson et al. J. Phys. Chem. , 95, 6603(1991) W. Vogel et al. J. Phys. Chem. B 102, 1750 (1998). For all these metals, we are in the case of a non dissociative adsorption mode and it seems that the structural evolution of the nanometer scale metallic atoms follows the same scheme. Based on all these experimental facts, we can suppose that CO adsorption leads to a disruption of metal-metal atoms and to the formation of Metal-(CO)n species. Then, metallic clusters can be observed and as supposed in the case of Pt, mass transport proceeds via surface carbonyl intermediates. This general schema is quite close to the one proposed in the case of NO adsorption process. 35
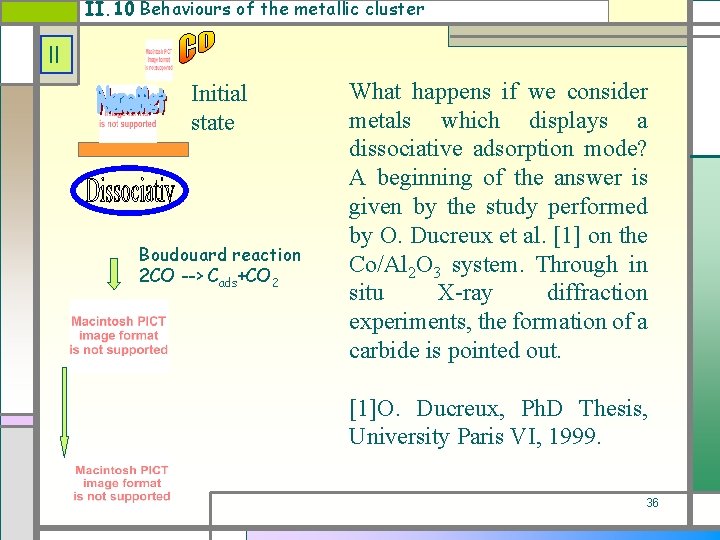
II. 10 Behaviours of the metallic cluster II Initial state Boudouard reaction 2 CO --> Cads+CO 2 What happens if we consider metals which displays a dissociative adsorption mode? A beginning of the answer is given by the study performed by O. Ducreux et al. [1] on the Co/Al 2 O 3 system. Through in situ X-ray diffraction experiments, the formation of a carbide is pointed out. [1]O. Ducreux, Ph. D Thesis, University Paris VI, 1999. 36
II. 11 A bridge between surface science and nanoscience II Adsorption Mode Cluster Behaviour 37

II. 11 a the metal-support interaction ? II the metal-support interaction ? • As we have seen the relationship we have proposed between the adsorption mode of NO and the behaviour of the metallic cluster seems to be independant of the nature of the support, except for Pd. • For this metal, a great difference exists between Mg. O and Ce. O 2. We have linked this dependency to the position of the metal versus the line which separates the two adsorption modes. • If this assumption is correct, such dependency is not so significant for other metals such Rh or Pt. 38

II. 11 b NO on monometallic cluster II Catalytic activity of metallic clusters ? Ø For metals above the stability line, NO adsorption leads to the formation of a metal oxide. Thus, the catalytic activity tends to decrease. Ø For metals below the line, large metallic clusters are finally generated and evolution of the catalytic activity will follow these structural modifications. N 2 NO t 39

II. 11 c Bimetallic systems II This simple model leads to a complete rejection of some bimetallic systems. For example, if we consider a Rh. Ru bimetallic cluster, the NO adsorption process conducts to the formation of a metal oxide i. e. the dissociation of NO will stop. N At the opposite, if we consider a Pt. Cu bimetallic system, the NO adsorption will lead to some large clusters. • A guideline for the choice of bimetallic systems is to add to Pt (or Cu) a second metal such Rh, Ru or Ir. If we consider the Ce. O 2 support, the Pt. Pd bimetallic seems to be acceptable while the Pt. Pd bimetallic system supported on alumina has to be rejected. NO Ce. O 2 2 t Al 2 O 3 40
II. 11 d a mixture of NO+O 2 II For metals above the stability line, NO adsorption leads to the formation of a metal oxide. The presence of O 2 will not change significantly this simple scheme. For these metals, it is necessary to add to a NO+O 2 mixing, a reductor agent in order to retablish the metallic state of the atoms. Thus, the presence of NO allowed the Pt particles to conserve a metallic character. In this case, we can probably play with the relative concentration of the two gases NO and O 2 to keep a metallic state. 41
II. 12 Conclusion & Perspectives II • Nature of the support • Preparation procedure • Cluster size • Temperature D. Bazin, (2003) Solid State Physics and Synchrotron Radiation Techniques to Understand Heterogeneous Catalysis in nanotechnology, Ed. G. A. Somorjai, S. Hermans, B. Zhou. D. Bazin, J. Lynch, M. Ramos-Fernandez, X-ray absorption spectroscopy and Anomalous Wide Angle X-ray Scattering Two basic tools in heterogeneous catalysis, Oil & Gas Science and Technology – Rev. IFP, 58 (2003), No. 6, pp. 667 -683 Séminaires 7 Avril 2005 : Modélisation de l’interaction entre le monoxyde d’azote et un agrégat métallique de dimension nanométrique, Centre de Recherche en Matière Condensée et Nanosciences, Marseille, France 11 Avril 2005 : L’interaction Agrégat métallique - Molécule au cœur des processus physico-chimiques environnementaux, Laboratoire de Chimie Théorique, Ivry sur Seine, France 20 Avril 2005 : L’interaction Agrégat métallique - Molécule au cœur des processus physico-chimiques environnementaux, Groupe de Physique des solides (GPS), Paris, France 42

II. 13 Remarks from Prof. J. Friedel 43
II. 12 Conclusion & Perspectives Structural Characterization @ the atomic scale Catalytic Activity 44
- Slides: 44