LABELED IMMUNOASSAYS PART 2 Enzyme Linked Immunosorbent Assay

LABELED IMMUNOASSAYS PART 2 Enzyme Linked Immunosorbent Assay (ELISA) Lab. 3

Labeled Immunoassays • The basic underlying principles of indicator labeled immunoassays are the same • There are differences with respect to the detail of the protocols • The designation given to each test differs according to the label used to detect the antigen/ antibody complexes 2

Enzyme Linked Immunosorbent Assay (ELISA) • One of many assays collectively called enzyme immunoassays (EIA) • Can be used to detect both antibody and antigen • Very Sensitive (ng & pg/m. L), • Relies on Monoclonal Abs • An enzyme is used as an indicator molecule • The enzyme does not provide detection directly but through the break-down of a substrate. 3

Enzymes used in ELISA • Enzymes used as labels for immunoassay are chosen according to the following criteria: § Turnover number § Sensitivity § Ease and speed of detection § Stability § Absence of interfering factors in patient samples § Availability and cost of enzyme and substrate 4

Enzymes used in ELISA • The most commonly used enzymes are: § Horseradish peroxidase (HRP) and § Alkaline phosphatase (AP) • Each has a high turnover number (rapid conversion of substrate to a product) resulting in high sensitivity • Other enzymes have been used as well, but they have not gained widespread acceptance because of limited substrate options • These include beta-galactosidase, acetylcholinesterase and catalase 5

Enzyme Substrates • A large selection of substrates is available for performing the ELISA with an HRP or AP conjugate • Substrates for AP and HRP, depending upon the plate-reading equipment available and the level of sensitivity required in the ELISA • An array of, and substrates is available for use with either enzyme • Chromogenic Color • Fluorogenic Fluoroescence • Chemiluminescent spectrophotometer light fluorometer luminometer • Chemiluminescent and chemifluorescent substrates provide a stronger signal than Chromogenic substrates because of their: • sensitivity (less than 1 pg/ml “ 10 -12”), • large linear range for detection • and excellent antibody conservation 6

Enzyme Substrates 1. Chromogenic substrates • A common ELISA substrate for HRP is: • Tetramethylbenzidine (TMB), • TMB is oxidized to yield a blue product that is water-soluble and absorbs light at 650 nm • The reaction can be halted by addition of acid or another stop reagent • Using sulfuric acid turns TMB yellow color which may be read at 450 nm 7
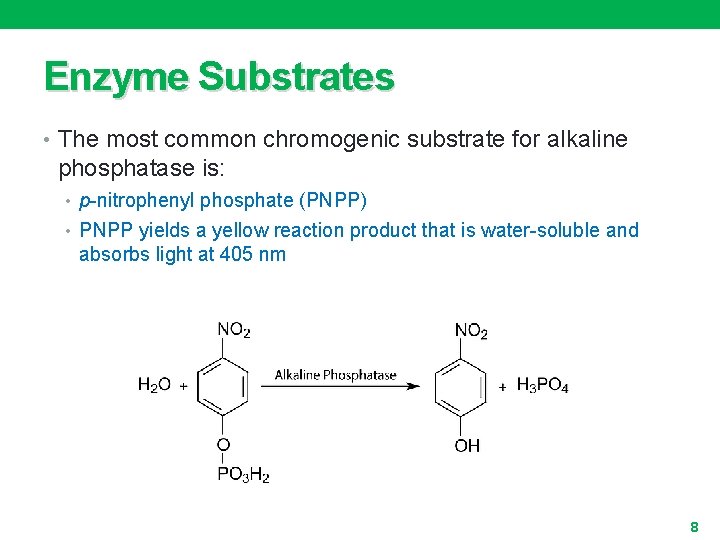
Enzyme Substrates • The most common chromogenic substrate for alkaline phosphatase is: • p-nitrophenyl phosphate (PNPP) • PNPP yields a yellow reaction product that is water-soluble and absorbs light at 405 nm 8
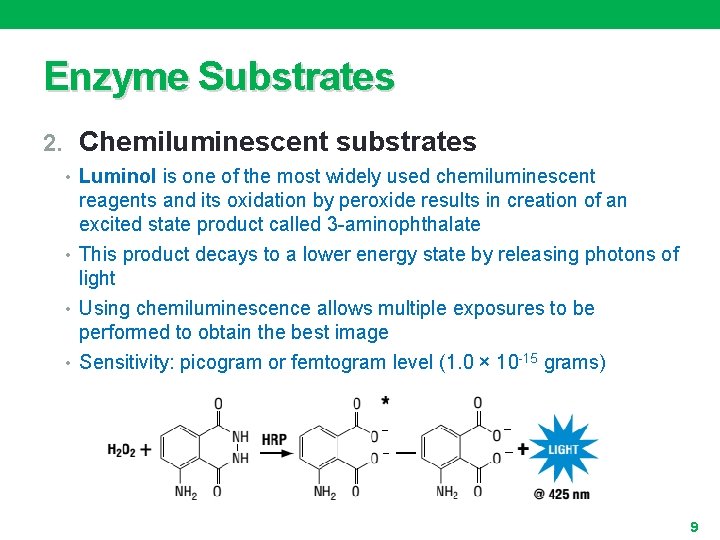
Enzyme Substrates 2. Chemiluminescent substrates • Luminol is one of the most widely used chemiluminescent reagents and its oxidation by peroxide results in creation of an excited state product called 3 -aminophthalate • This product decays to a lower energy state by releasing photons of light • Using chemiluminescence allows multiple exposures to be performed to obtain the best image • Sensitivity: picogram or femtogram level (1. 0 × 10 -15 grams) 9

Enzyme Substrates • Fluorogenic substartes • A Fluorogenic Substrate is a nonfluorescent material that is acted upon by an enzyme to produce a fluorescent compound • Different substartes are also available for AP and HRP enzymes • p-Hydroxphenylpropionic acid (HPPA) is used for HRP enzyme 10
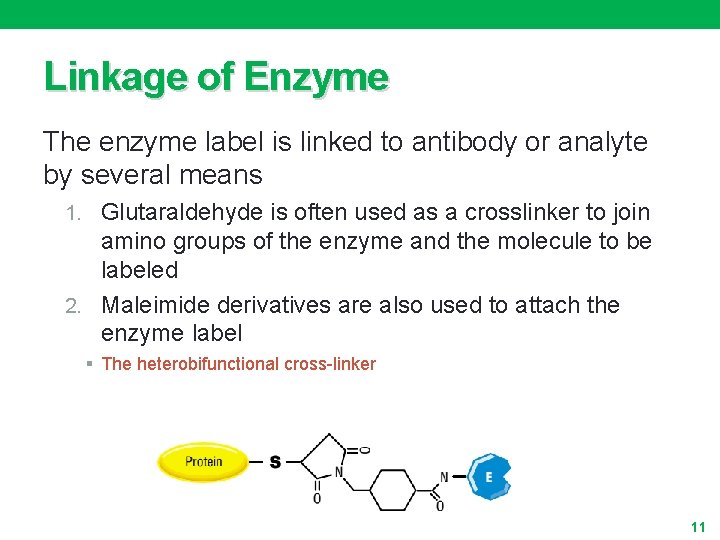
Linkage of Enzyme The enzyme label is linked to antibody or analyte by several means 1. Glutaraldehyde is often used as a crosslinker to join amino groups of the enzyme and the molecule to be labeled 2. Maleimide derivatives are also used to attach the enzyme label § The heterobifunctional cross-linker 11
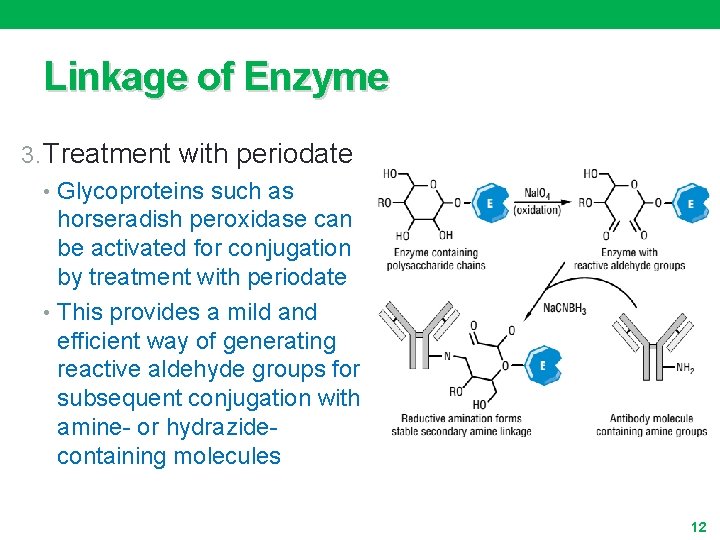
Linkage of Enzyme 3. Treatment with periodate • Glycoproteins such as horseradish peroxidase can be activated for conjugation by treatment with periodate • This provides a mild and efficient way of generating reactive aldehyde groups for subsequent conjugation with amine- or hydrazidecontaining molecules 12
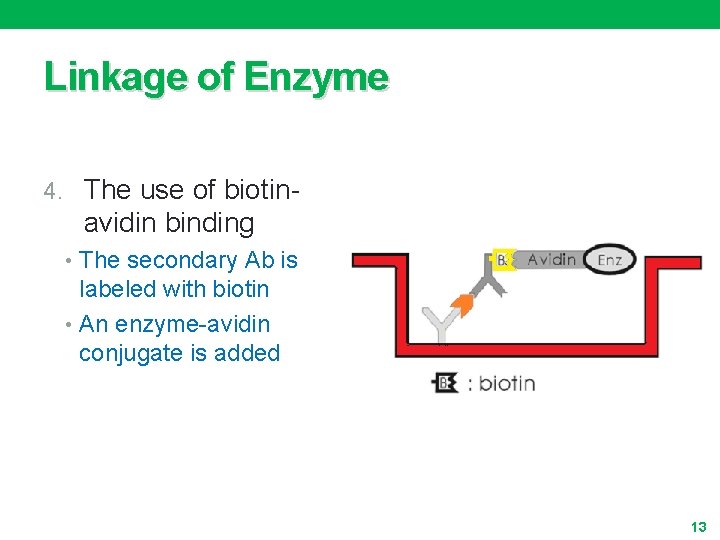
Linkage of Enzyme 4. The use of biotin- avidin binding • The secondary Ab is labeled with biotin • An enzyme-avidin conjugate is added 13

Coating of Microplate • A key feature of the solid-phase ELISA is that antigens or • • • antibodies can be attached to surfaces easily by passive adsorption This process is commonly called coating Most proteins adsorb to plastic surfaces, probably as a result of hydrophobic interactions between nonpolar protein substructures and the plastic matrix Since most of proteins' hydrophilic residues are at the outside and most of the hydrophobic residues orientated towards the inside Partial denaturation of some proteins results in exposure of hydrophobic regions and ensures firmer interaction with the plastic This can be achieved by exposing proteins to low p. H or mild detergent 14

ELISA Protocol 1 - Coating antibody or antigen onto the microplate § § Dilute the protein to be coated in a buffer such as PBS or Carbonate-Bicarbonate and add 100 μl of this solution per well Incubate for 18 -20 hours at room temperature or 4°C Coated with Antibody when analysing antigen Coated with Antigen when analysing antibody Analyte = antigen Incubate, wash 15

ELISA Protocol 2 - Blockage of free binding sites § Block the unoccupied sites on the surface of the well to reduce the amount of nonspecific binding of proteins [blocking agent (200300 μl/well)] § § A variety of blocking buffers ranging from nonfat milk to highly purified proteins have been used to block unreacted sites The blocking buffer should improve the sensitivity of the assay by reducing the background interference Analyte = antibody Analyte = antigen Incubate, wash 16
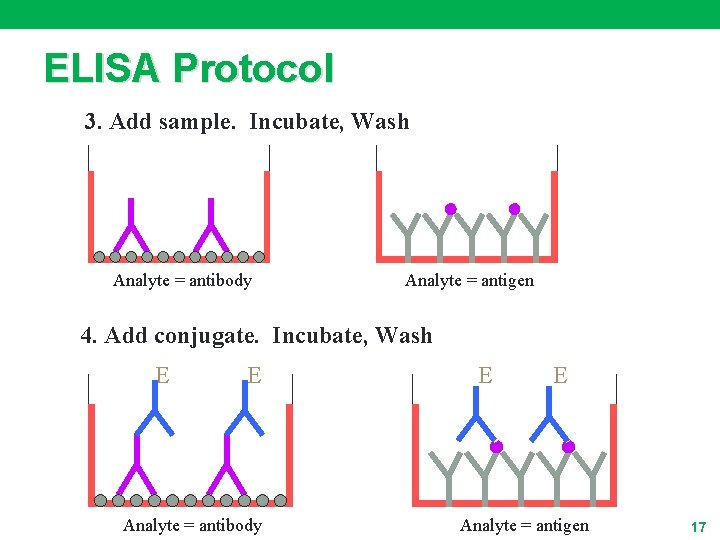
ELISA Protocol 3. Add sample. Incubate, Wash Analyte = antibody Analyte = antigen 4. Add conjugate. Incubate, Wash E E Analyte = antibody E E Analyte = antigen 17
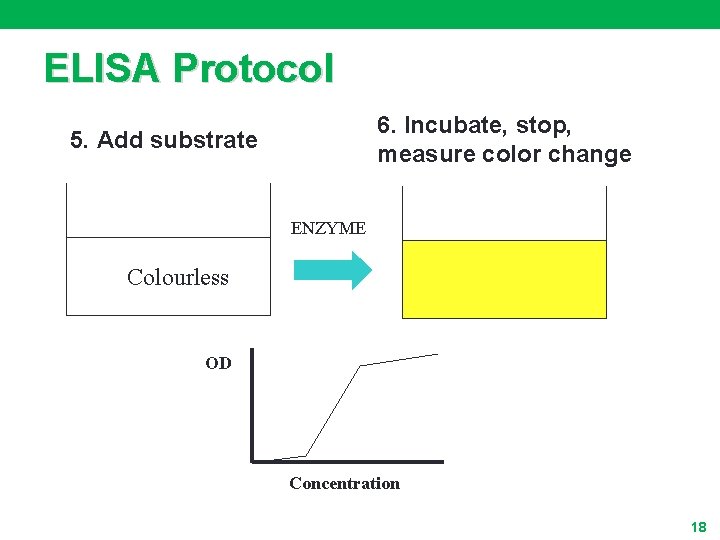
ELISA Protocol 6. Incubate, stop, measure color change 5. Add substrate ENZYME Colourless OD Concentration 18

Types of Enzyme Immunoassay § Three main types: 1. Competitive ELISA 2. Indirect ELISA 3. Sandwich ELISA 19

1 - Competitive ELISA • Enzyme activity is inversely proportional to the concentration of the test substance • A sensitivity of nanograms (10 -9 g)/ml can be achieved • This method can be used for measurement of small molecules that are relatively pure • such as insulin, and estrogen Competition ELISA to detect Antigens (Antibody-coated plate) Low [analyte] E 1. Anti-analyte Low [analyte] High [analyte] 2. Analyte-E + sample 1. Anti-analyte E E Low [analyte] High [analyte] E 3. Wash 2. Analyte- E E + sample 1. Anti-analyte 4. Substrate 3. Wash 2. Analyte-E + sample 1. Analyte E E High [analyte] 20
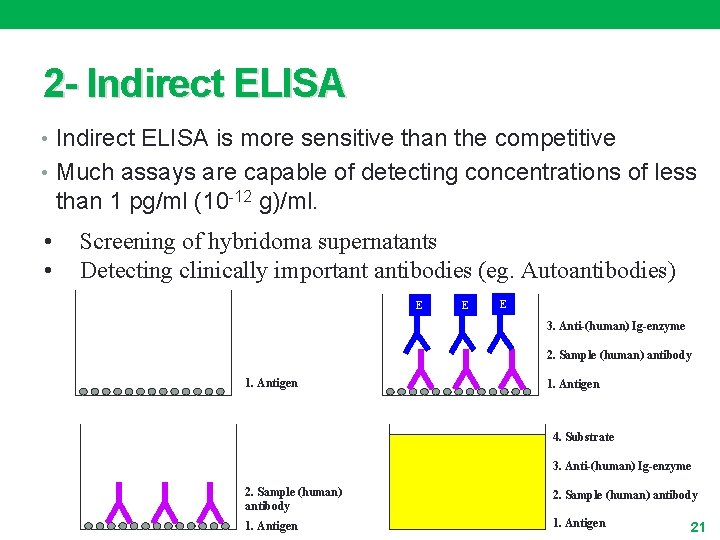
2 - Indirect ELISA • Indirect ELISA is more sensitive than the competitive • Much assays are capable of detecting concentrations of less than 1 pg/ml (10 -12 g)/ml. • • Screening of hybridoma supernatants Detecting clinically important antibodies (eg. Autoantibodies) E E E 3. Anti-(human) Ig-enzyme 2. Sample (human) antibody 1. Antigen E E 4. Substrate 3. Anti-(human) Ig-enzyme 2. Sample (human) antibody 1. Antigen 21
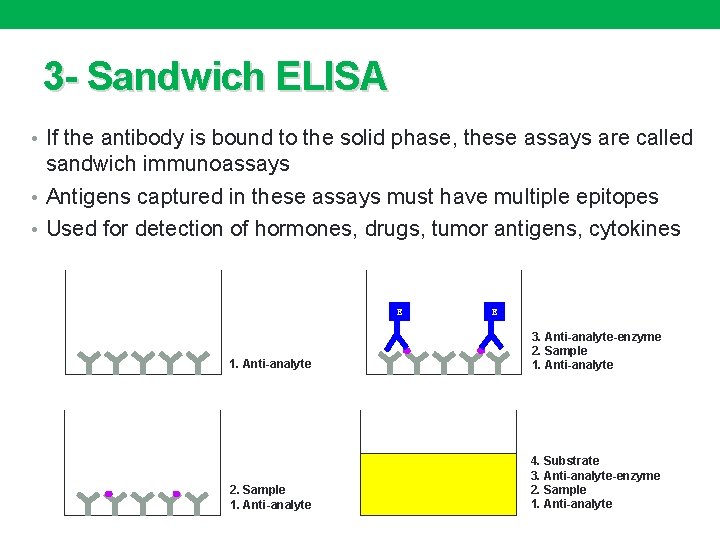
3 - Sandwich ELISA • If the antibody is bound to the solid phase, these assays are called sandwich immunoassays • Antigens captured in these assays must have multiple epitopes • Used for detection of hormones, drugs, tumor antigens, cytokines E E 1. Anti-analyte 3. Anti-analyte-enzyme 2. Sample 1. Anti-analyte 4. Substrate 3. Anti-analyte-enzyme 2. Sample 1. Anti-analyte

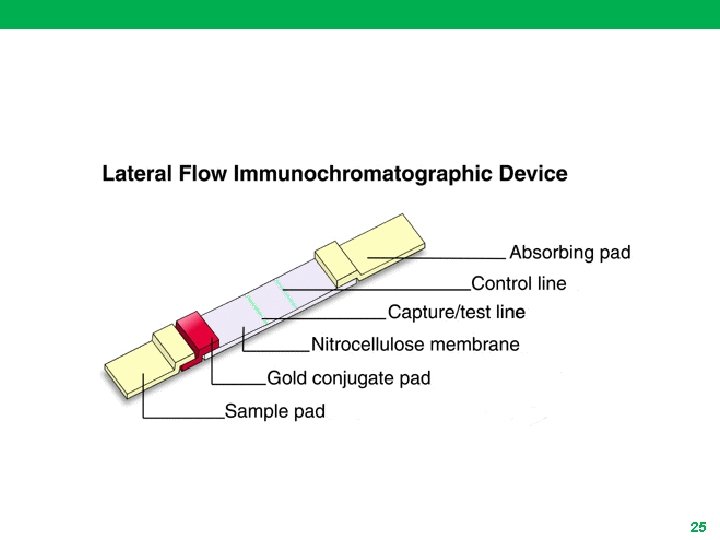
Lateral Flow Immunoassay (LFIA) (Immunochromatography System Assays) (Membrane based immunodiagnostic assay) 23

Principle • Are based on the migration of nano or micro particles on strips for analytes detection in several areas • In principle, any colored particle can be used, however latex (blue color) or nanometer sized particles of gold (red color) are most commonly used • The technology is based on a series of capillary beds, such as pieces of porous paper • These elements has the capacity to transport fluid (e. g. , urine, plasma, . . ) spontaneously 24
25
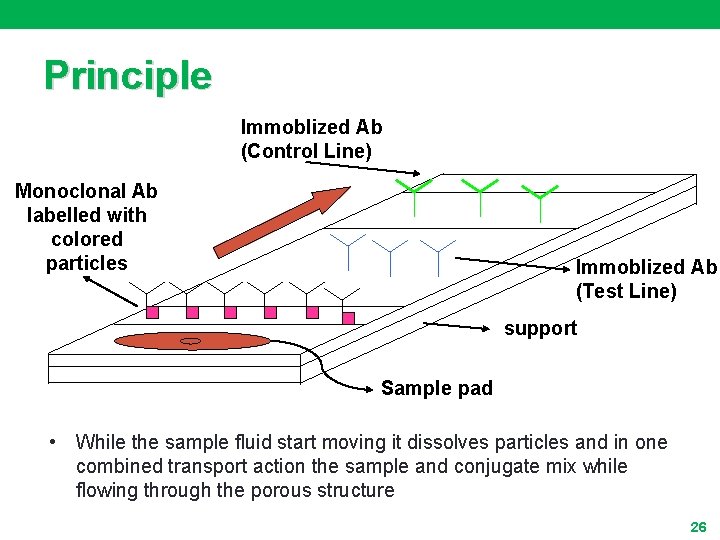
Principle Immoblized Ab (Control Line) Monoclonal Ab labelled with colored particles Immoblized Ab (Test Line) support Sample pad • While the sample fluid start moving it dissolves particles and in one combined transport action the sample and conjugate mix while flowing through the porous structure 26
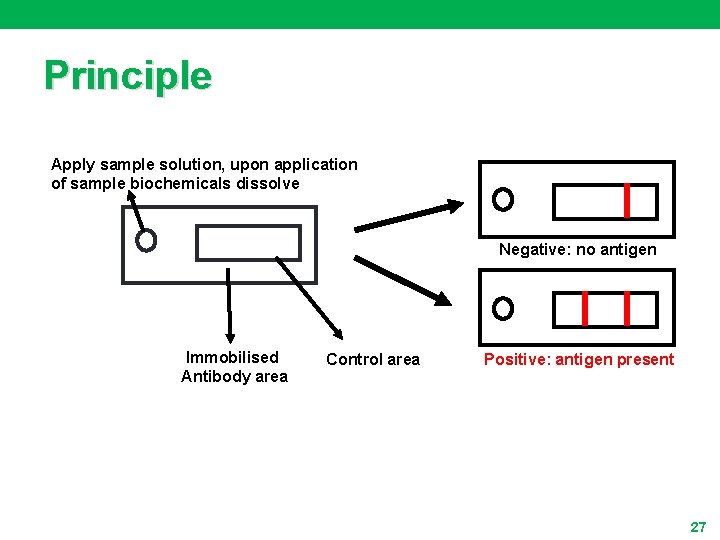
Principle Apply sample solution, upon application of sample biochemicals dissolve Negative: no antigen Immobilised Antibody area Control area Positive: antigen present 27

• Uses • Human and animal diagnostics Point of Care testing (Po. CT) • Forensics • Environmental testing • Advantages • Rapid test (<15 minutes) • Reliable and easy-to-use (no special equipment required nor trained staff) • Semi-quantitative or quantitative results • Non-refrigerated storage 28

Detection of HBs. Ag Lateral Flow Immunoassay (LFIA) 29

Intended Use • HBs. Ag One Step Hepatitis B Surface Antigen Test cassette (Serum/Plasma) is a rapid chromatographic immunoassay for the qualitative detection of Hepatitis B Surface Antigen in serum or plasma
Summary and explanation • Viral hepatitis is a systemic disease primarily involving the liver, and in most cases is caused by one of three viruses: • Hepatitis A (HAV), • Hepatitis B (HBV) • or Hepatitis C (HCV). • The antigen found on the envelope of HBV is designated Hepatitis B Surface antigen (HBs. Ag) and its presence in serum or plasma indicates active HBV infection. • In a typical Hepatitis B infection, HBs. Ag will be detected 2 – 4 weeks before ALT levels become abnormal and 3 -5 weeks before symptoms or jaundice develop.

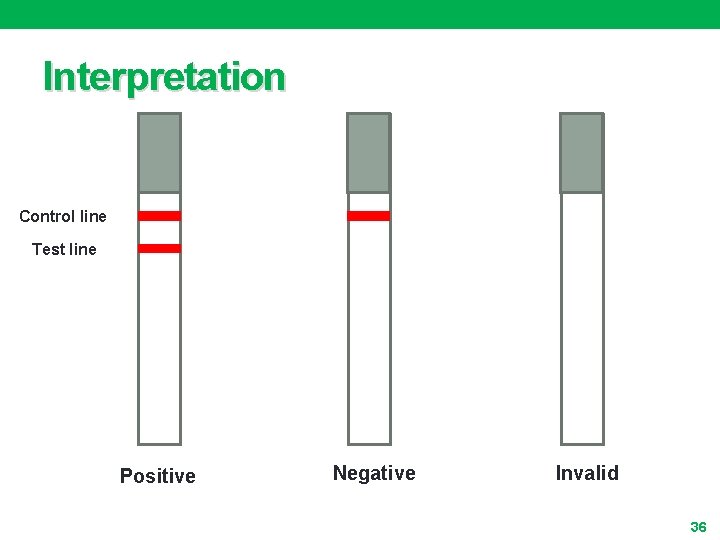
Principle • The membrane is precoated with anti-HBs. Ag antibodies on the test line region of the test • During testing, the serum or plasma specimen reacts with the particle coated with anti-HBs. Ag antibody • The mixture migrates upward on the membrane chromatographically by capillary action to react with anti-HBs. Ag antibodies on the membrane and generate a colored line • The presence of this colored line in the test region indicates a positive result, while its absence indicates a negative result 32

Quality Control • A procedural control is included in the test • A red line appearing in the control region (C) is the internal procedural control • It confirms sufficient specimen volume and correct procedural technique (membrane wicking has occurred) 33

Limitations • LINEAR HBs. Ag cannot detect less than 1 ng/m. L of HBs. Ag in specimens • If the test result is negative and clinical symptoms persist, additional follow-up testing using other clinical methods is suggested • A negative result at any time does not preclude the possibility of Hepatitis B infection 34

Procedure Allow the test strip to equilibrate to room temperature prior to testing 2. Place the test device on a clean and level surface. Add 100 µl of serum or plasma to the specimen well (S) and then start the timer. 3. Wait for the red line (s). The result should be read at 15 minutes 1. 35
Interpretation Control line Test line Positive Negative Invalid 36
- Slides: 36