LABELED IMMUNOASSAY Lab 2 Dr M Alzaharna 2015

LABELED IMMUNOASSAY Lab. 2 Dr. M. Alzaharna 2015
![Immunoassay • An immunoassay is a test that uses antibody and antigen complexes [immuno-complex] Immunoassay • An immunoassay is a test that uses antibody and antigen complexes [immuno-complex]](http://slidetodoc.com/presentation_image_h2/cd64b85f7d4eafcb0badaa36398a75b5/image-2.jpg)
Immunoassay • An immunoassay is a test that uses antibody and antigen complexes [immuno-complex] as a mean of generating a measurable result • Immuno refers to an immune response that causes the body to generate antibodies, • and assay refers to a test • The assay takes advantage of the specific binding of an antibody to its antigen • The antibodies used must have a high affinity for the antigen • Immunoassays derive their unique specificity, sensitivity, and flexibility from three important properties of antibodies: • Their ability to bind to an extremely wide range of natural and man-made chemicals, biomolecules, cells, and viruses • Exceptional specificity for the substance to which each antibody binds. • The strength of the binding between an antibody and its target Dr. M. Alzaharna 2015 2

Immunoassay • Both the presence of antigen or antibody can be measured • For measuring hormones such as insulin, the insulin acts as the antigen • When detecting infection the presence of antibody against the pathogen is measured • For numerical results the response of the object being measured must be compared to standards of a known concentration • This is usually done through the plotting of a standard curve on a graph paper, and then the quantity of the unknown is found from the curve Dr. M. Alzaharna 2015 3
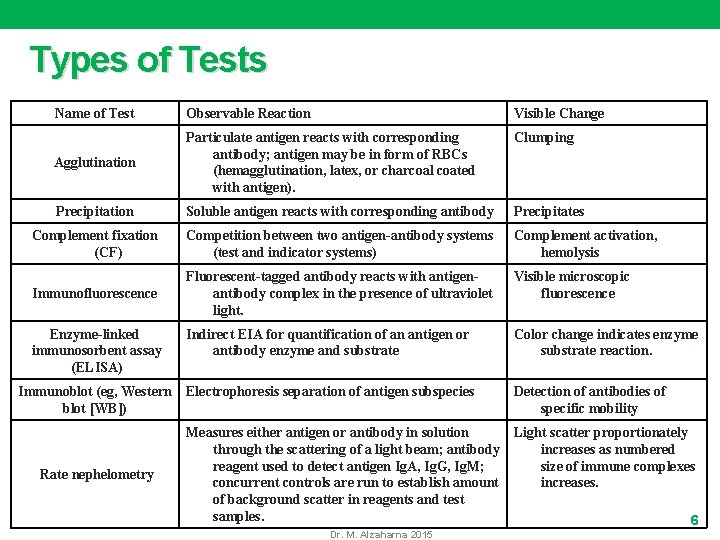
Types of Tests Name of Test Observable Reaction Visible Change Clumping Agglutination Particulate antigen reacts with corresponding antibody; antigen may be in form of RBCs (hemagglutination, latex, or charcoal coated with antigen). Precipitation Soluble antigen reacts with corresponding antibody Precipitates Complement fixation (CF) Competition between two antigen-antibody systems (test and indicator systems) Complement activation, hemolysis Immunofluorescence Fluorescent-tagged antibody reacts with antigenantibody complex in the presence of ultraviolet light. Visible microscopic fluorescence Enzyme-linked immunosorbent assay (ELISA) Indirect EIA for quantification of an antigen or antibody enzyme and substrate Color change indicates enzyme substrate reaction. Immunoblot (eg, Western blot [WB]) Electrophoresis separation of antigen subspecies Detection of antibodies of specific mobility Measures either antigen or antibody in solution through the scattering of a light beam; antibody reagent used to detect antigen Ig. A, Ig. G, Ig. M; concurrent controls are run to establish amount of background scatter in reagents and test samples. Light scatter proportionately increases as numbered size of immune complexes increases. Rate nephelometry Dr. M. Alzaharna 2015 6

Antigen-antibody interaction • Antigens are substances that stimulate and subsequently react with the products of an immune response. They may be: • Enzymes, • Toxins, • Microorganisms (eg, bacterial, viral, parasitic, fungal), • Tumors, • Autoimmune factors • Antibodies are proteins produced by the body's immune system in response to an antigen or antigens. • The antigen-antibody response is the body's natural defense against invading organisms Dr. M. Alzaharna 2015 7
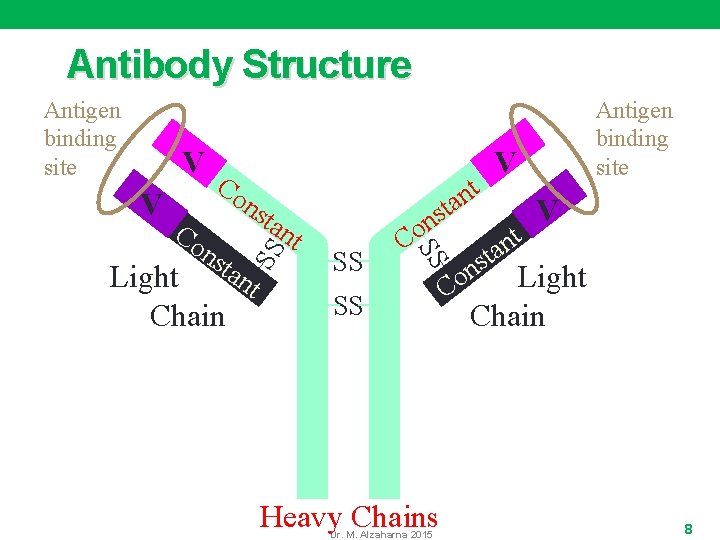
Antibody Structure V V Co ns t tan Light t Chain t n a t s on SS Co ns tan SS Antigen binding site SS Heavy Chains Dr. M. Alzaharna 2015 n o C Antigen binding site V t n a t s V Light Chain 8
Antigen Binding tig An b a i r a V eavy H Antigen 3 2 le en Antigen 1 e l b a i r a V ght Li Dr. M. Alzaharna 2015 9

• There are now hundreds of immunoassays for dozens of analytes including: • Hormones • Tumor markers • Antibodies • Drugs • Cardiac markers • covering the fields of: • Endocrinology • Oncology • Hematology • Toxicology • Immunology • Infectious diseases • Developments in antibodies, labels, and automation have resulted in highly specific and sensitive assays Dr. M. Alzaharna 2015 8

Constituents of a Labeled Immunoassay • For detection of an analyte, the following are usually a part of the assay: 1. Labeled analytes 2. Specific antibody 3. Standards or calibrators 4. A method to separate the bound from free components 5. A method for detection of the label Dr. M. Alzaharna 2015 9

1 - Labeled Analytes • A labeled analyte is used to detect whether or not specific binding has taken place • The label used in immunoassay: • must not alter the reactivity of the molecule • and it should remain stable for the shelf life of the reagent • Labels attached to analytes and antibodies can be: • Radioactive • usually iodine-125 (radioimmunoassay), • Enzymes • such as alkaline phosphatase and horseradish peroxidase, (enzyme immunoassay or enzyme-linked immunosorbent assay [ELISA]), • Chemiluminescent • e. g. acridinium ester • Fluorescent • e. g. fluorscein Dr. M. Alzaharna 2015 10

Methods of coupling indicator labels to antigen or antibody • The Methods include: A. Binding to amino acids • The radioactive isotope iodine is covalently linked to tyrosine residues present on antibodies and most antigens Dr. M. Alzaharna 2015 11
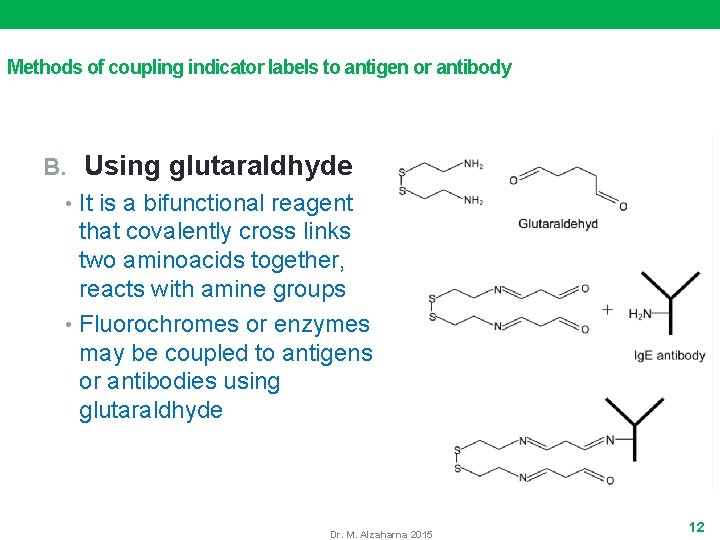
Methods of coupling indicator labels to antigen or antibody B. Using glutaraldhyde • It is a bifunctional reagent that covalently cross links two aminoacids together, reacts with amine groups • Fluorochromes or enzymes may be coupled to antigens or antibodies using glutaraldhyde Dr. M. Alzaharna 2015 12

Methods of coupling indicator labels to antigen or antibody C. Biotin-Streptavidin system • Biotin is a vitamin that can bind tightly to either avidin or streptavidin (proteins) • The natural attraction of biotin to these two proteins is a property that has been exploited to facilitate coupling of indicator molecules to antigens or antibodies • At the end of the assay, a conjugate of streptavidin linked to a signalgenerating substance is added • Examples of suitable conjugates are streptavidin-alkaline phosphatase, streptavidin-HRP, streptavidin-125 I, streptavidin fluorescein Dr. M. Alzaharna 2015 13

2 - Production of Antibodies • The production of antibodies is an important process in the use of immunoassays because it is the antibody-antigen complexes that form the basic • Antibodies can be called depending upon the technique used to produce them either: a) Monoclonal or b) Polyclonal Dr. M. Alzaharna 2015 14
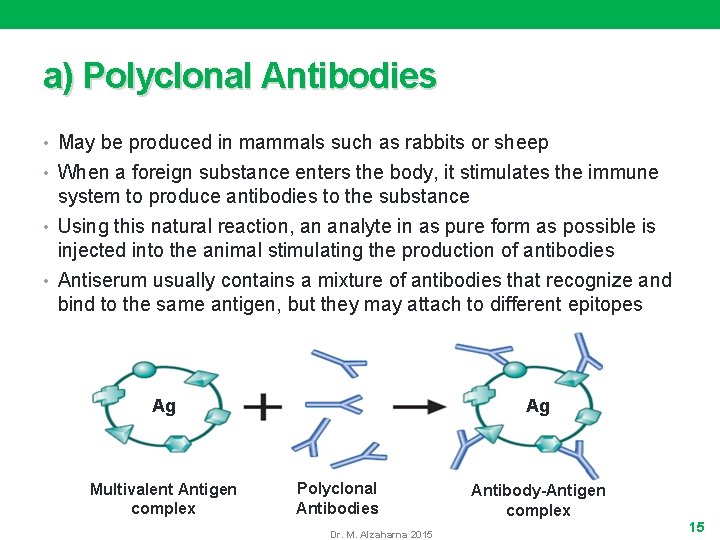
a) Polyclonal Antibodies • May be produced in mammals such as rabbits or sheep • When a foreign substance enters the body, it stimulates the immune system to produce antibodies to the substance • Using this natural reaction, an analyte in as pure form as possible is injected into the animal stimulating the production of antibodies • Antiserum usually contains a mixture of antibodies that recognize and bind to the same antigen, but they may attach to different epitopes Ag Ag Multivalent Antigen complex Polyclonal Antibodies Dr. M. Alzaharna 2015 Antibody-Antigen complex 15
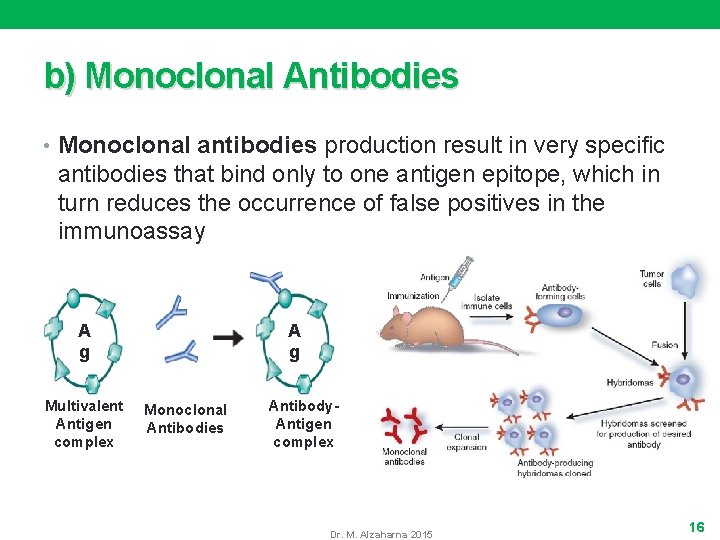
b) Monoclonal Antibodies • Monoclonal antibodies production result in very specific antibodies that bind only to one antigen epitope, which in turn reduces the occurrence of false positives in the immunoassay A g Multivalent Antigen complex A g Monoclonal Antibodies Antibody. Antigen complex Dr. M. Alzaharna 2015 16
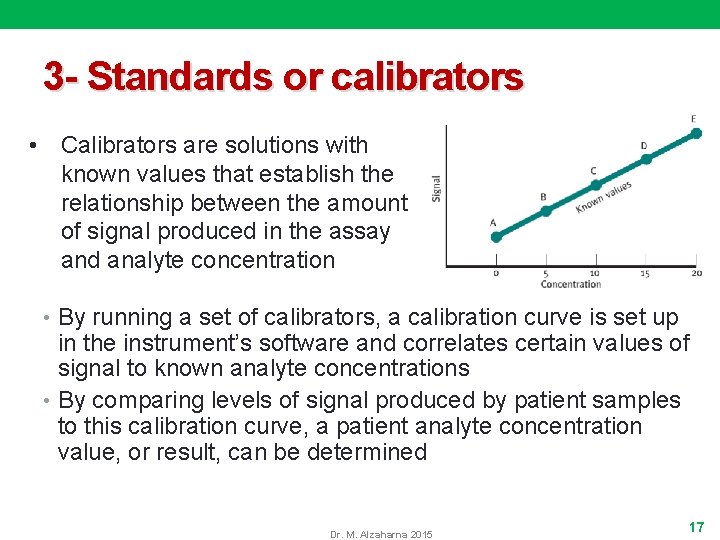
3 - Standards or calibrators • Calibrators are solutions with known values that establish the relationship between the amount of signal produced in the assay and analyte concentration • By running a set of calibrators, a calibration curve is set up in the instrument’s software and correlates certain values of signal to known analyte concentrations • By comparing levels of signal produced by patient samples to this calibration curve, a patient analyte concentration value, or result, can be determined Dr. M. Alzaharna 2015 17

4 - Separation Methods • In most assays, once the reaction between antigen and antibody has taken place, there must be a way of separating reacted from unreacted analyte • Unreacted analyte can be removed by: A. Adsorption on particles such as dextran-coated charcoal • These adsorb out the smaller unbound molecules, which are then separated from bound molecules by centrifugation or filtration • The amount of label remaining in the supernatant provides an indirect measure of analyte present in the patient's sample B. Another means of separation involves precipitation of antigenantibody complexes • Complexes can be precipitated by adding concentrated solutions of ammonium sulfate, or ethanol Dr. M. Alzaharna 2015 18

4 - Separation Methods C. Currently, most immunoassays use a solid-phase stage for separation • Numerous substances, such as polystyrene test tubes, microtiter plates are used for this purpose • Antigen or antibody is attached by physical adsorption, and when specific binding takes place, complexes remain attached to the solid phase Dr. M. Alzaharna 2015 19

5 - Methods for detection of label • The last step common for all immunoassays is detection of the labeled analyte • The method depends on the label; • e. g. 125 I is easily detected by a γ-counter • Enzymes are generally used to produce coloured products from colourless substrates that can be determined easily using a spectrophotometer • Automated plate readers are commercially available which make reading large numbers of samples relatively easy Dr. M. Alzaharna 2015 20

Quality Control in Immunoassays • It is essential that quality control procedures be established • This is done to limit random errors, such as • temperature fluctuations • minor changes in the concentration of reagents • and changes in detector efficiency • A negative control and a positive control should be run • This serves as a check on the quality of the reagents to make sure that the label is readily detectable under current testing conditions Dr. M. Alzaharna 2015 21

Methodological Principles 1. Competitive Immunoassays 2. Noncompetitive Immunoassays 3. Heterogeneous Immunoassays 4. Homogeneous Immunoassays Dr. M. Alzaharna 2015 22
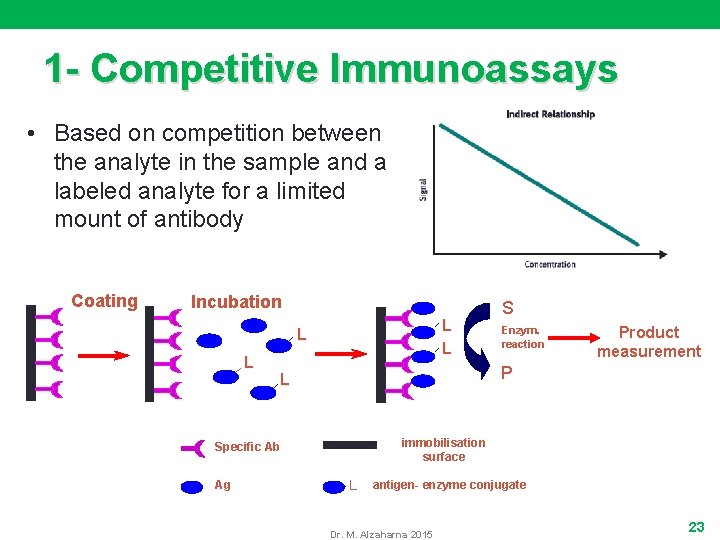
1 - Competitive Immunoassays • Based on competition between the analyte in the sample and a labeled analyte for a limited mount of antibody Coating Incubation L L Enzym. reaction Product measurement P L immobilisation surface Specific Ab Ag S L antigen- enzyme conjugate Dr. M. Alzaharna 2015 23
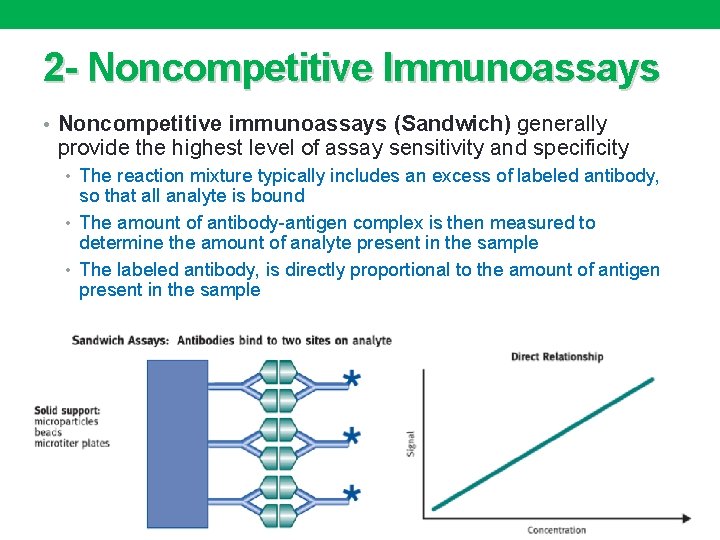
2 - Noncompetitive Immunoassays • Noncompetitive immunoassays (Sandwich) generally provide the highest level of assay sensitivity and specificity • The reaction mixture typically includes an excess of labeled antibody, so that all analyte is bound • The amount of antibody-antigen complex is then measured to determine the amount of analyte present in the sample • The labeled antibody, is directly proportional to the amount of antigen present in the sample Dr. M. Alzaharna 2015 24

Heterogeneous versus Homogeneous Immunoassays 3 - Heterogeneous Immunoassay • Methods that require separation of bound Ab-Ag* complex 4 - Homogeneous Immunoassay • Those that do not require separation. • Homogeneous methods have been generally applied to the measurement of small analytes such as abused and therapeutic drugs • Since homogeneous methods do not require the separation of the bound Ab-Ag* from the free Ag*, they are generally much easier and faster to perform Dr. M. Alzaharna 2015 25

Types of Immunoassays (Heterogeneous) • Within the categories of competitive, noncompetitive, homogenous, and heterogeneous, there are specific types, which include: 1. Radioimmunoassays (RIAs) • utilize a radioactive label (usually 125 I, 3 H or 14 C), which emits radiation that can be measured with a beta or gamma counter Dr. M. Alzaharna 2015 26

Types of Immunoassays (Heterogeneous) 2. Enzyme linked immunosorbant assay (ELISA): • Direct, sandwich and competitive • Reaction components are absorbed or bound to the surface of a solid phase, commonly a well of a microtiter plate • Absorbance is measured using a micro-plate reader Dr. M. Alzaharna 2015 27
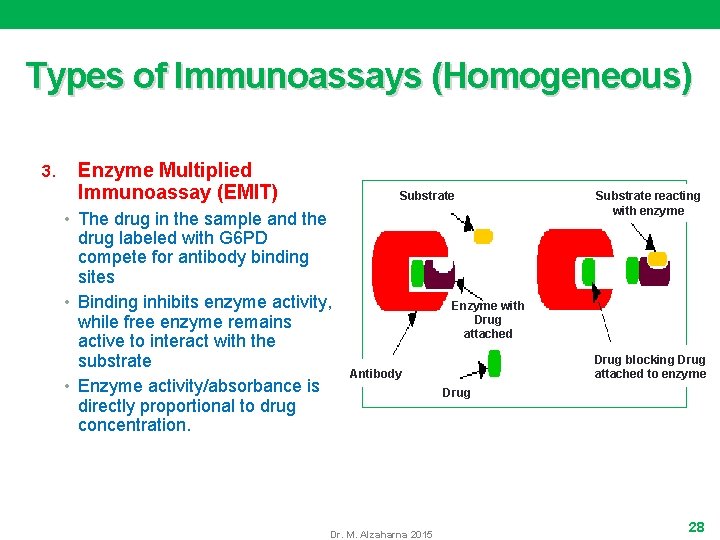
Types of Immunoassays (Homogeneous) 3. Enzyme Multiplied Immunoassay (EMIT) Substrate • The drug in the sample and the drug labeled with G 6 PD compete for antibody binding sites • Binding inhibits enzyme activity, while free enzyme remains active to interact with the substrate • Enzyme activity/absorbance is directly proportional to drug concentration. Substrate reacting with enzyme Enzyme with Drug attached Drug blocking Drug attached to enzyme Antibody Dr. M. Alzaharna 2015 Drug 28

Immunoassay Results • Qualitative • Single point calibration at a specific cutoff • Results are either ‘positive’ or ‘negative’ (i. e. above or below the cutoff) • Quantitative • Provides numeric results that are an estimate the analyte concentration based on the measurement standards Dr. M. Alzaharna 2015 29

Rubella Ig. M Test Kit Dr. M. Alzaharna 2015

Intended Use • Rubella Ig. M ELISA test is an enzyme linked immunosorbent assay (ELISA) for the detection of Ig. M class antibodies to Measles (Rubella) in human serum or plasma. Dr. M. Alzaharna 2015 31

Summary and explanation • Measles is an acute, highly contagious viral disease. • Although measles is usually considered a childhood disease, it can be contracted at any age. • Measles is spread by direct contact with nasal or throat secretions of infected people or, less frequently, by airborne transmission. Dr. M. Alzaharna 2015 32

Summary and explanation • The presence of Ig. G antibody to Rubella virus is indicative of previous exposure or vaccination. • In individuals with acute measles, a significant increase in measles Ig. G antibody level is indicative of recent infection. • Ig. M antibodies to Rubella virus are often detectable with onset of the rash and typically persist for 4 weeks. • At least 80% of patients will be positive for Rubella Ig. M at 6 days and 100% at 16 days after onset of symptoms. Dr. M. Alzaharna 2015 33

Principle • Diluted patient serum is added to wells coated with purified • • • Rubella antigen. Rubella Ig. M specific antibody, if present, binds to the antigen. All unbound materials are washed away and the enzyme conjugate is added to bind to the antibody-antigen complex, if present. Excess enzyme conjugate is washed off and substrate is added. The plate is incubated to allow the hydrolysis of the substrate by the enzyme. The intensity of the color generated is proportional to the amount of Ig. M specific antibody in the sample. Dr. M. Alzaharna 2015 34

Procedure 1. Bring all specimens and kit reagents to room temperature (18 -26 °C) and gently mix. 2. Place the desired number of coated strips into the holder. 3. Negative control, positive control, and calibrator are ready to use. Prepare 1: 21 dilution of test samples, by adding 10 μl of the sample to 200 μl of sample diluent. Mix well. 4. Dispense 100 μl of diluted sera, calibrator and controls into the appropriate wells. For the reagent blank, dispense 100μl sample diluent in 1 A well position. Tap the holder to remove air bubbles from the liquid and mix well. Incubate for 20 minutes at room temperature. 5. Remove liquid from all wells. Wash wells three times with 300 μl of 1 X wash buffer. Blot on absorbance paper or paper towel. Dr. M. Alzaharna 2015 35

Procedure 6. Dispense 100 μl of enzyme conjugate to each well and incubate for 20 minutes at room temperature. 7. Remove enzyme conjugate from all wells. Wash wells three times with 300 μl of 1 X wash buffer. Blot on absorbance paper or paper towel. 8. Dispense 100 μl of TMB substrate and incubate for 10 minutes at room temperature. Add 100 μl of stop solution. 9. Read O. D. at 450 nm using ELISA reader within 15 min. 10. A dual wavelength is recommended with reference filter of 600 -650 nm. Dr. M. Alzaharna 2015 36

Calculation of results 1. 2. Check Calibrator Factor (CF) value on the calibrator bottle. This value might vary from lot to lot. Make sure you check the value on every kit. Calculate the cut-off value: • Calibrator OD x Calibrator Factor (CF). 3. Calculate the Ab (Antibody) Index of each determination by dividing the O. D. value of each sample by cut-off value. • Example of typical results • • Calibrator mean OD = 0. 8 Calibrator Factor (CF) = 0. 5 Cut-off Value = 0. 8 x 0. 5= 0. 400 Positive control O. D. = 1. 2 • Ab Index = 1. 2 / 0. 4 = 3 • Patient sample O. D. = 1. 6 • Ab Index = 1. 6 / 0. 4 = 4. 0 Dr. M. Alzaharna 2015 37

Validation requirement and quality control 1. The O. D. of the Calibrator should be greater than 0. 250. 2. The Ab index for Negative control should be less than 0. 9. 3. The Ab Index for Positive control should be greater than 1. 2. Dr. M. Alzaharna 2015 38

Interpretation of results • The following is intended as a guide to interpretation of Measles Ig. M test results; each laboratory is encouraged to establish its own criteria for test interpretation based on sample populations encountered. • Antibody Index Interpretation • < 0. 9 No detectable Ab to Rubella Ig. M by ELISA. • 0. 9 – 1. 1 Borderline positive, follow up testing is recommended if clinically indicated. • > 1. 1 Detectable antibody to measles Ig. M by ELISA. Dr. M. Alzaharna 2015 39
- Slides: 39