Label the different parts of a microscope Biology
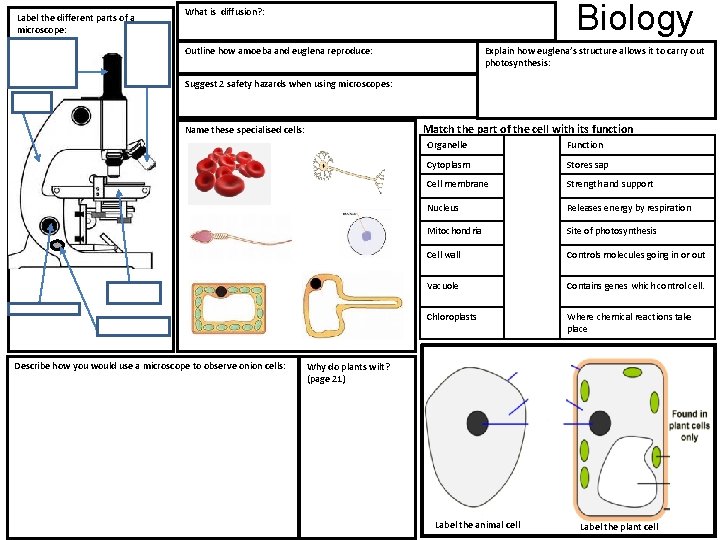
Label the different parts of a microscope: Biology What is diffusion? : Outline how amoeba and euglena reproduce: Explain how euglena’s structure allows it to carry out photosynthesis: Suggest 2 safety hazards when using microscopes: Match the part of the cell with its function Name these specialised cells: Describe how you would use a microscope to observe onion cells: Organelle Function Cytoplasm Stores sap Cell membrane Strength and support Nucleus Releases energy by respiration Mitochondria Site of photosynthesis Cell wall Controls molecules going in or out Vacuole Contains genes which control cell. Chloroplasts Where chemical reactions take place Why do plants wilt? (page 21) Label the animal cell Label the plant cell
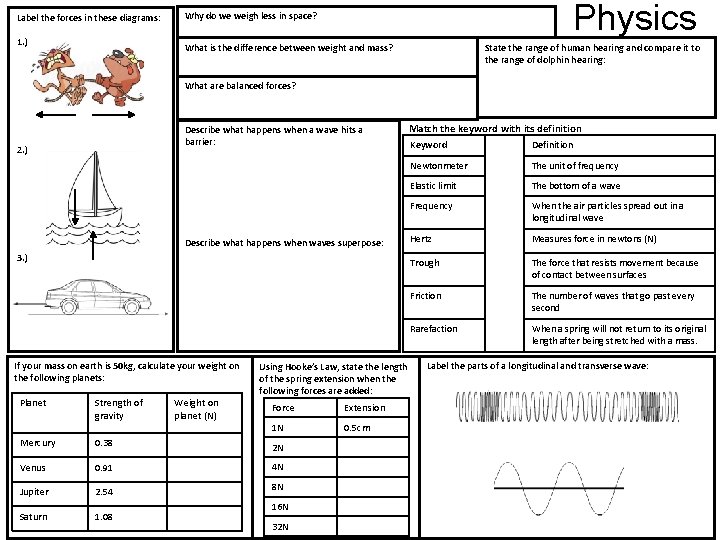
Label the forces in these diagrams: 1. ) Physics Why do we weigh less in space? What is the difference between weight and mass? State the range of human hearing and compare it to the range of dolphin hearing: What are balanced forces? Describe what happens when a wave hits a barrier: 2. ) Describe what happens when waves superpose: 3. ) If your mass on earth is 50 kg, calculate your weight on the following planets: Planet Strength of gravity Weight on planet (N) Using Hooke’s Law, state the length of the spring extension when the following forces are added: Force Extension 1 N 0. 5 cm Mercury 0. 38 2 N Venus 0. 91 4 N Jupiter 2. 54 8 N Saturn 1. 08 16 N 32 N Match the keyword with its definition Keyword Definition Newtonmeter The unit of frequency Elastic limit The bottom of a wave Frequency When the air particles spread out in a longitudinal wave Hertz Measures force in newtons (N) Trough The force that resists movement because of contact between surfaces Friction The number of waves that go past every second Rarefaction When a spring will not return to its original length after being stretched with a mass. Label the parts of a longitudinal and transverse wave:
Draw particles in the solid, liquid and gas states: Solid Chemistry Why does gas pressure increase when more particles are added? How can you check the purity of a substance using its melting point? Compare the processes of boiling and evaporation: Which 3 factors affect diffusion speed? Liquid Gas This table shows the boiling and melting points of 4 elements Substance Boiling point (°C) Melting point (°C) Bromine -7 59 Mercury -39 357 Silver 961 2210 Neon -249 -246 a. ) Which has the highest boiling point? Match the keyword with its definition Keyword Definition Gas pressure The change of state from a gas to liquid. Melting The tiny things that substances are made from Condensation The change of state from liquid to gas when particles leave the surface of a liquid Particle The change of state from solid to liquid Evaporation The force exerted by gas particles per unit area of a surface b. ) Name one substance that is a gas at 20°C? c. ) Name one substance that is a liquid at 100°C? Estefano says that gas pressure is the result of particles colliding with each other. Is he correct? Explain your answer. Tommy went camping. He put a can of baked beans on the fire without opening the lid. The can exploded. Using your knowledge on particles, explain why this happened. Explain what happens in sublimation: Using your knowledge of states of matter, complete this table: State Solid Liquid Gas Can you compress it? Does it flow? Shape
- Slides: 3