Lab Safety Laboratory Safety Guidelines Directions 1 Follow

Lab Safety

Laboratory Safety Guidelines Directions 1. Follow ___________. – – – Follow both ______ and written verbal _______ instructions. Read _______ instructions BEFORE ____ proceeding with an experiment. all Check written instructions before _______. asking If verbal corrections must be made to a written questions note procedure, be sure to ____ them ______ on your lab handout. If verbal and written instructions seem to conflict, or if they seem vague, please ______for _______. ask clarification

Laboratory Safety Guidelines only 2. Use chemicals and equipment _____ as ______. directed ___ 3. Lab time is NOT play time. ALWAYS handle respect chemicals with _______. – – taste touch Never _____ chemicals or _____ chemicals with your bare hands, unless specifically instructed to do so. Never _____ chemicals directly. Instead, smell hold the container at arms length, slowly bring it closer and “waft” some of the fumes toward your nose.

Laboratory Safety Guidelines 4. Dress appropriately for lab. (We need to remember cycle days for P. E. , why not lab, too? ) loose clothing – Avoid wearing ______________ which may get caught on equipment or dragged in chemicals. closed toed shoes or sneakers. DO NOT wear – Wear _______-_______ sandals flip flops in lab. _______ or ______-______ tie ___ it ____ back while working in – If you have long hair ______ the lab. safety _____ glasses and – It is REQUIRED that you wear _____ RECOMMENDED that you wear ______ lab ____ apronswhen working with; __________, chemicals __________, or glassware __________. flames (For certain labs, you will be advised that aprons will be required. ) DO NOT WEAR CONTACT LENSES DURING LAB EXPERIMENTS. Contact lenses may trap chemicals and fumes in your eyes, making potential injuries even worse.

Laboratory Safety Guidelines Eating chewing ____ gum 5. ______ and _____ are strictly prohibited during experiments. Food and gum can absorb fumes and cause you to ingest harmful substances. 6. Be sure to know the locations and proper use safety equipment (fire extinguisher, of all _________. eyewash, safety shower, fume hoods, spill control center, etc. , ) 7. Keep your lab station ____. Arrange neat equipment in an organized fashion to reduce the possibility of ______. Do NOT breakage store ______ items in your lab station. unnecessary

Laboratory Safety Guidelines 8. Be sure all equipment is stored in its _____________. Your proper location group drawer is accessed by only your group, but common drawers are shared with other classes. (Click for Illustration) Dispose 9. _______ of materials ONLY AS DIRECTED. Do NOT discard solid ______ materials into the sink. liquids Dispose of ______ in the sink ONLY if instructed to do so. 10. Never heat a ______ system. closed

Laboratory Safety Guidelines aiming 11. Avoid _______ a container’s opening at yourself or someone else, especially when heating. 12. Never leave a ___________ lit burner unattended. 13. Always replace container _____. lids 14. Always add _____ to _____, acid water NOT water to acid, when diluting.

Laboratory Safety Guidelines aisles 15. Keep _____ clear of books, back packs, gym bags, etc. , 16. Do not _____ in lab. run 17. Students will ____ in lab. You should stand sitting particularly avoid _____ on lab tables. 18. If you spill acid or other chemicals on yourself, rinse with plenty of water (use ____ or sink eyewash station _______ if appropriate, use ___________ safety shower only for major spills) and notify your instructor.

Laboratory Safety Guidelines 19. Report all ______ (equipment accidents breakage, spills and injuries) to your instructor. 20. Students are not permitted in the ____________ without stock room permission from the instructor.

Laboratory Safety Guidelines 21. Know emergency procedures. – – right out of the classroom door, take For fire alarms turn ____ stairway “B” all the way down to the ground level, exit the building via the rear doors, move across the parking area into TEACHER the grassy area and FIND YOUR ________. In case of a tornado warning, exit the room from either door in an orderly fashion and find a seat along the _______. wall/lockers In any tornado situation, avoid ___________ and windows ceilings long-span ______________ (such as in the ________, _____ auditorium gym and ________. ) cafeteria If any type of evacuation occurs during a lab, be sure all _______ gas _______ jets and _____ water _______ faucets are turned off. The last person to leave the classroom (usually the instructor) lights must be sure to turn off the ______ and close the ________________. Door (should be UNlocked) 22. Always be prepared to help fellow students in an emergency.

The Chemistry Lab (Room 313)

Group Drawers – each lab group will have one of these that ONLY their group will be using. Lab Stations Common Drawers – these four drawers contain equipment to be shared by lab groups in different classes. BACK TO THE SHOW
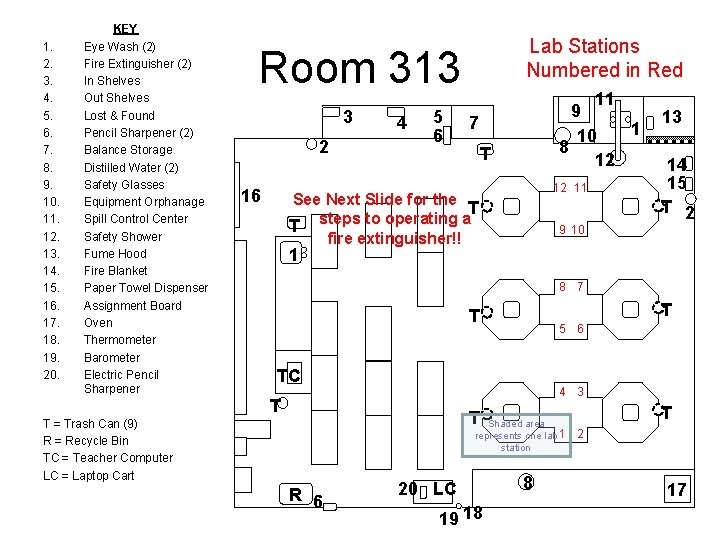
Lab Stations Numbered in Red Room 313 3 2 16 ROOM 313 -Chemistry 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. KEY Eye Wash (2) Fire Extinguisher (2) In Shelves Out Shelves Lost & Found Pencil Sharpener (2) Balance Storage Distilled Water (2) Safety Glasses Equipment Orphanage Spill Control Center Safety Shower Fume Hood Fire Blanket Paper Towel Dispenser Assignment Board Oven Thermometer Barometer Electric Pencil Sharpener 4 5 6 11 9 1 10 8 12 7 T 12 11 See Next Slide for the T steps to operating a T fire extinguisher!! 9 10 13 14 15 T 2 1 8 7 T T 5 6 TC 4 3 T T T = Trash Can (9) R = Recycle Bin TC = Teacher Computer LC = Laptop Cart Shaded area represents one lab 1 station R 6 20 LC 19 18 8 T 2 17

To use a fire extinguisher… 1. Pull the pin, 2. Aim at the BASE of the flames, and 3. Squeeze the handle. 4. Sweep side to side. (REMEMBER: P. A. S. S. )

Lab Equipment

Lab Equipment Beaker use to contain and/or heat liquids spout for pouring volume markings for approximation NOT measurement Erlenmeyer Flask contain/heat liquids with minimal evaporation shaped for swirling volume markings for approximation NOT measurement Graduated Cylinder set on level surface read at eye level bottom of the meniscus estimate last digit Volumetric Flask used to mix solutions of a single specific volume

Lab Equipment Buret Used to dispense a precisely measured volume of a reagent. Typically used in a process called “titration” in which the concentration of a solution in a flask is determined by measuring how much of a solution with KNOWN concentration is needed to react with the unknown solution Before using, clean thoroughly and rinse with a small amount of “titrant” (the KNOWN solution After using, clean thoroughly and disassemble the stopcock.

Lab Equipment Forceps tweezer-like utensils for grasping small, objects Beaker Tongs curved, ruber-coated jaws for gripping smooth straight sides of a beaker Do NOT use extensively over flames (will melt rubber) Crucible Tongs unique shape of jaws allows pinching or cradling designed for use with crucible and cover

Lab Equipment Test Tube Tongs Scissor-style tongs for grabbing test tubes (especially the large test tubes) Test Tube Holder Spring-loaded clamp for holding test tubes Use with care (suggestion – use two at a time) squeeze too much – drops out the bottom Test Tube Rack set test tubes in for holding and cooling Hot test tubes? Use the metal rack!

Lab Equipment Pipestem Triangle Set on iron ring to hold crucible while heating May be used to support funnel, if funnel is too small for ring Crucible & Cover Acorn-shaped porcelain container Use to heat materials to a very high temperature Evaporating Dish Wide, shallow procelain dish used to evaporate liquid from a mixture over heat Watch Glass Used as a cover for evaporating dish or beaker Use with edges up, bulge down

Lab Equipment Spatula Use to scoop granulated solids (also called a “scoopula”) Clean thoroughly between uses (May or may not have wooden handle) Microspatula Same as spatula, but used for amounts <1 g Do not use as a stirring rod! Liquid Funnel Narrow neck – used in filtering and for directing the flow of liquids Powder Funnel Has a wider spout Use to pour granulated solids into containers with small openings

Name That Lab Equipment!! Bea ker

Name That Lab Equipment!! Forc eps

Name That Lab Equipment!! Cruci ble

Name That Lab Equipment!! Test Tube

Name That Lab Equipment!! Bea ker

Name That Lab Equipment!! Crucibl e and

Name That Lab Equipment!! Wat ch

Name That Lab Equipment!! Liqui d

Name That Lab Equipment!! Powd er

Name That Lab Equipment!! Pipest em

Name That Lab Equipment!! Evapora ting

Name That Lab Equipment!! Micr o-

Name That Lab Equipment!! Test Tube

Name That Lab Equipment!! Spat ula

Name That Lab Equipment!! Test Tube

Name That Lab Equipment!! Gradua ted

Name That Lab Equipment!! Buret

Name That Lab Equipment!! Volume tric

Name That Lab Equipment!! Erlenme yer

Name That Lab Equipment!! Barom eter

Name That Lab Equipment!! Thermom

Lab Technique

Proper Lab Techniques 1. Precision Principle § Estimate one digit between the smallest markings. § (In other words – “Read between the lines. ”) § If a measurement falls directly on a line, record your value with a final zero. § (Ex. A measurement which falls “exactly” on the third line between 7 and 8 would be recorded as 7. 30)

Proper Lab Techniques 2. Graduated Cylinder § Set on a level surface. § Read at eye level. § Read at the BOTTOM of the MENISCUS. § Don’t forget to apply the precision principle. § A white sheet of paper behind the cylinder may help you see the meniscus better.

Proper Lab Techniques 3. Triple Beam Balance 1. Zero the balance § Clear the pan § Set all riders to zero § Adjust the thumbscrew to align the pointer 2. Mass an empty container or piece of massing paper § Place paper or container on pan. § Move riders (largest first, smallest last) until the pointer aligns. § Read the hundreds, tens, ones and tenths and estimate the hundredths of a gram. Record mass to two decimal places. 3. Mass your object with the container or paper. (Same as step 2) 4. Subtract. ALWAYS MASS BY DIFFERENCE!!!!

Proper Lab Techniques Why mass by difference? Hypothetical Situation… You are massing an object and your balance is off by +0. 02 g, but you don’t know it… If you mass by difference… If you mass directly… The object’s actual mass: 6. 08 g You measure: 6. 10 g The paper’s actual mass: 1. 16 g You measure: 1. 18 g Paper + Object (Actual = 6. 08+1. 16): 7. 24 g Paper and Object (Measured): 7. 26 g Mass by Difference = 7. 26 – 1. 18 = 6. 08 g

Proper Lab Techniques 4. Electronic Balance 1. 2. 3. 4. Make sure there is nothing on the balance pan. Turn on the balance by pressing the power button. Wait for the balance display to read all zeroes. Mass an empty container or piece of massing paper… § Place paper or container on pan. § Wait for the display to settle. Note that the digits may drift and you may need to take the reading anyway after the first few seconds when the change in the numbers is not significant. Record the mass of the paper or container to 2 decimal places. § 5. Mass your object with the container or paper. 6. (Same as step 4) Subtract. (Note that the TARE button will zero the balance WITH the container on it, but sometimes we can’t do that because we need the mass of the container later in the experiement!) ALWAYS MASS BY DIFFERENCE!!!!
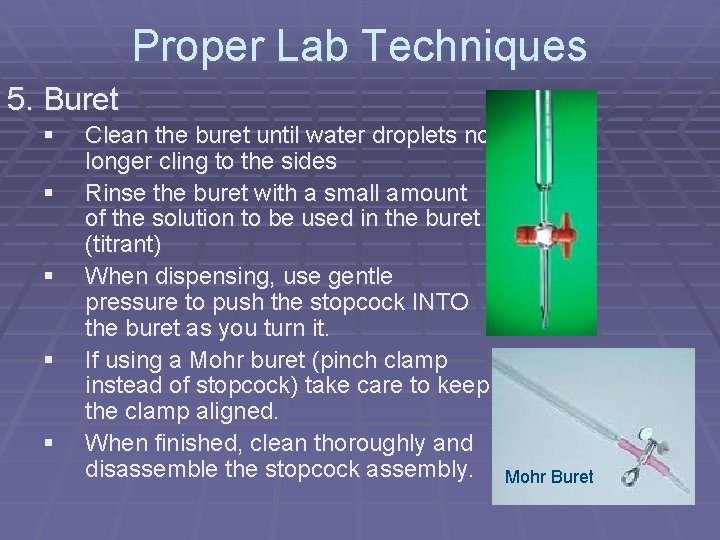
Proper Lab Techniques 5. Buret § § § Clean the buret until water droplets no longer cling to the sides Rinse the buret with a small amount of the solution to be used in the buret (titrant) When dispensing, use gentle pressure to push the stopcock INTO the buret as you turn it. If using a Mohr buret (pinch clamp instead of stopcock) take care to keep the clamp aligned. When finished, clean thoroughly and disassemble the stopcock assembly. Mohr Buret

Proper Lab Techniques 6. Bunsen Burner § 3 -point safety check – hose, stem, adjustments (open gas adjustment about one turn, close air collar) § Connect burner to gas jet § Announce “I’m lighting the burner!” § Turn on gas jet (parallel) § Strike flint lighter above the burner. § Adjust to about 6” using gas knob and to a smooth, pale blue flame using the air collar. (Yellow or orange flame has too little oxygen Rough, sputtering flame has too much oxygen. )
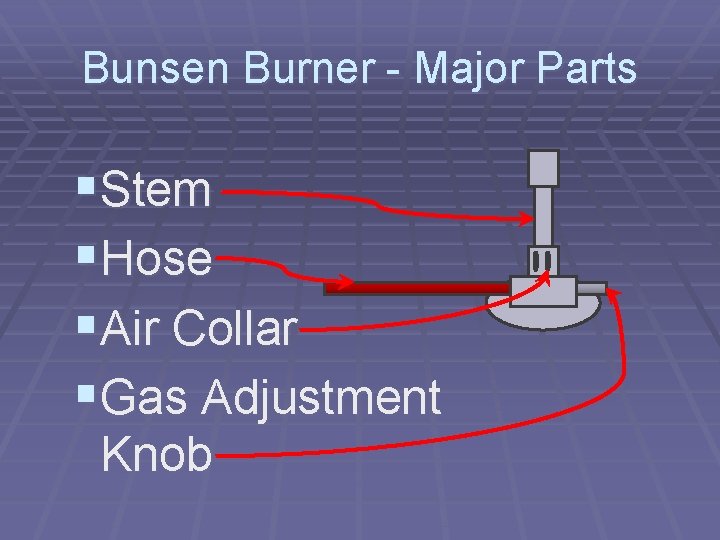
Bunsen Burner - Major Parts §Stem §Hose §Air Collar §Gas Adjustment Knob

ALWAYS TURN THE BUNSEN BURNER OFF AT THE SOURCE, NOT WITH THE GAS KNOB

Proper Lab Techniques 7. Cross Contamination § Clean containers and utensils thoroughly between uses. § Handle lids properly; § Set lids down with the open end up. § Replace lids immediately after use. § NEVER return excess chemicals to the original stock container. (Pass extras on to another lab group or ask your instructor what to do with them. )
- Slides: 53