Lab Safety Lab Equipment Measurement Quiz Review Get

Lab Safety, Lab Equipment, & Measurement Quiz Review

Get out the safety scenarios from yesterday. Finish them if you have not yet. Also, grab the sheet from the front for today’s notes.
Test tube • Cylindrical-shaped glass tube used primarily in laboratories

Test tube brush • Used to clean test tubes

Pipet • Device used for measuring and transferring small volumes of liquid

Erlenmeyer flask • A cone shaped container used for holding liquids

Beaker • A wide mouth container used to hold liquids

Graduated cylinder • Used for measuring volume

Funnel • Hollow cone-shaped tool used for transferring liquids

Goggles • Used to protect eyes during experiments

Name the lab equipment

The basic unit of length in the SI system is the … • The basic unit of length in the SI system is the meter

The two units that measure the length of smaller objects are, … • millimeter • centimeter

The shorter lines on the metric ruler are called… • millimeters

The longer lines on the metric ruler are called… • centimeters

Measuring Length How many millimeters are in 1 centimeter? 1 centimeter = 10 millimeters What is the length of the line in centimeters? _______cm What is the length of the line in millimeters? _______mm What is the length of the line to the nearest centimeter? ____cm HINT: Round to the nearest centimeter – no decimals. Ruler: http: //www. k 12 math. com/math-concepts/measurement/ruler-cm. jpg
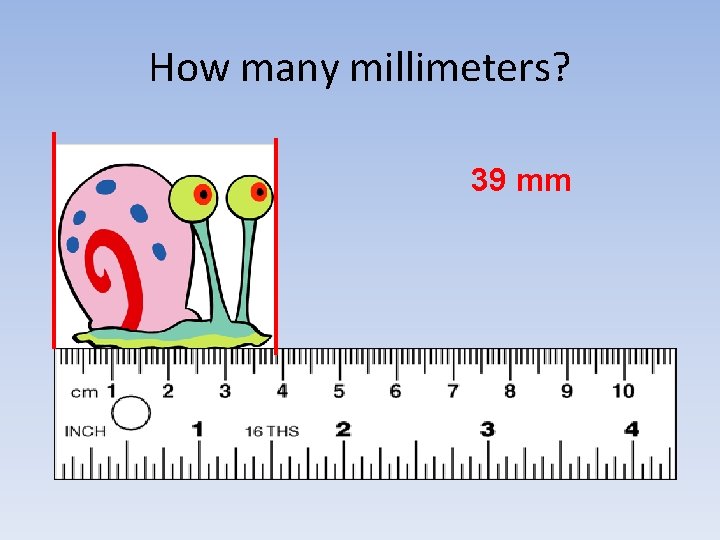
How many millimeters? 39 mm
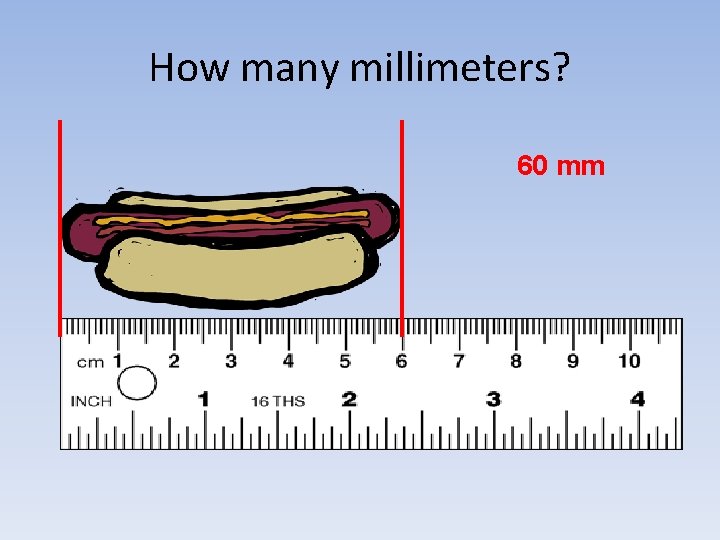
How many millimeters? 60 mm

How many centimeters? 8. 5 cm
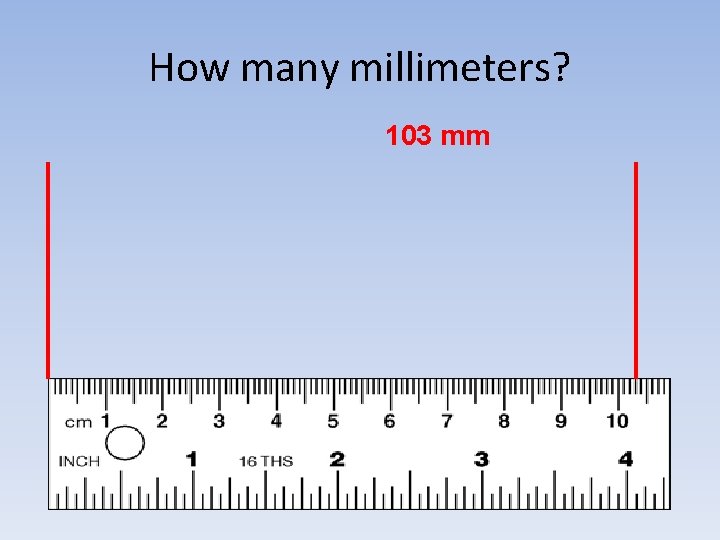
How many millimeters? 103 mm
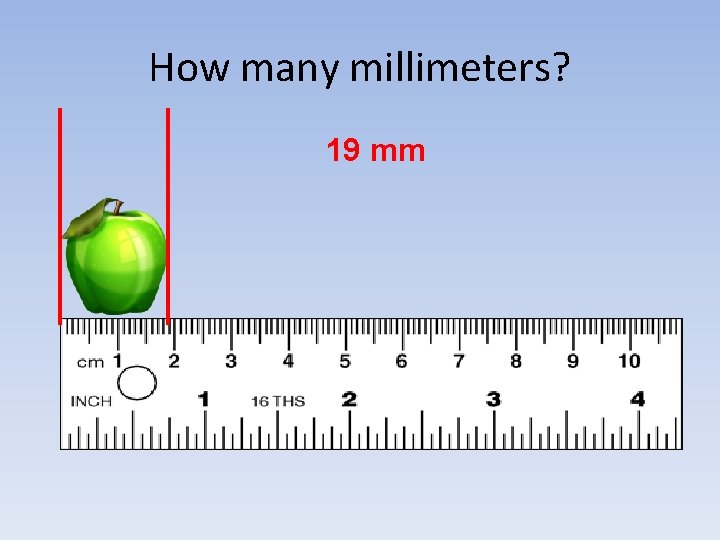
How many millimeters? 19 mm
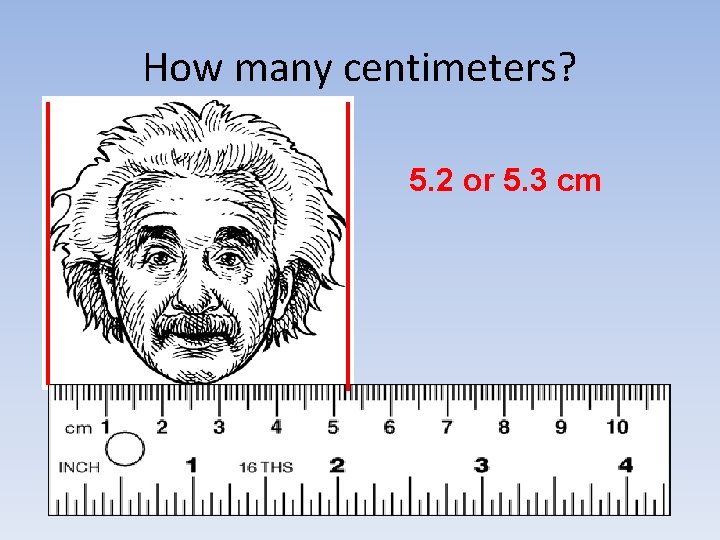
How many centimeters? 5. 2 or 5. 3 cm
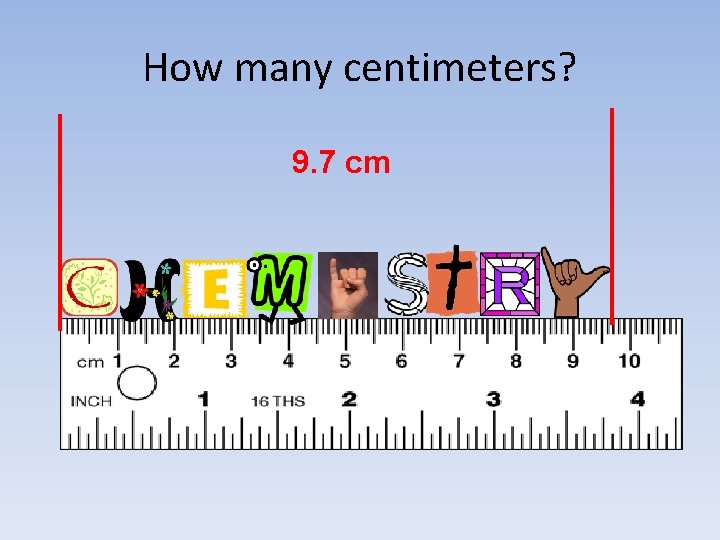
How many centimeters? 9. 7 cm

Mass refers to the amount of matter in an object. Triple Beam Balance

The basic unit of mass in the SI system is the… • The basic unit of mass in the SI system is the gram.

A smaller object’s mass such as a paper clip would be measured in • Smaller objects such as a paper clip would be measure in milligrams.

Measuring Mass We will be using triple-beam balances to find the mass of various objects. The objects are placed on the scale and then you move the weights on the beams until you get the lines on the right-side of the scale to match up. Once you have balanced the scale, you add up the amounts on each beam to find the total mass. What would be the mass of the object measured in the picture? _______ + _______ = ____ g Top Image: http: //www. southwestscales. com/Ohaus_Triple_Beam_750 -SO. jpg Bottom Image: http: //www. regentsprep. org/Regents/biology/units/laboratory/graphics/triplebeambalance. jpg

Measuring Mass – Triple-Beam Balance 1 st – Place the film canister on the scale. 2 nd – Slide the large weight to the right until the arm drops below the line. Move the rider back one groove. Make sure it “locks” into place. 3 rd – Repeat this process with the top weight. When the arm moves below the line, back it up one groove. 4 th – Slide the small weight on the front beam until the lines match up. 5 th – Add the amounts on each beam to find the total mass to the nearest tenth of a gram.

https: //www. wisc-online. com/learn/naturalscience/chemistry/gch 202/reading-a-triplebeam-balance

• https: //www. wisc-online. com/learn/naturalscience/chemistry/gch 302/measuringvolume-using-a-graduated-cylinder#

Measuring Volume We will be using graduated cylinders to find the volume of liquids and other objects. Read the measurement based on the bottom of the meniscus or curve. When using a real cylinder, make sure you are eye-level with the level of the water. What is the volume of water in the cylinder? _____m. L What causes the meniscus? A concave meniscus occurs when the molecules of the liquid attract those of the container. The glass attracts the water on the sides. Top Image: http: //www. tea. state. tx. us/student. assessment/resources/online/2006/grade 8/science/images/20 graphicaa. gif Bottom Image: http: //morrisonlabs. com/meniscus. htm
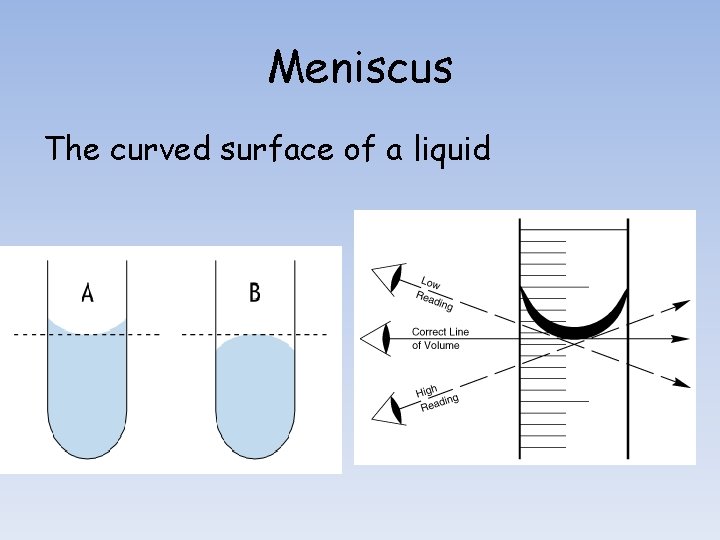
Meniscus The curved surface of a liquid
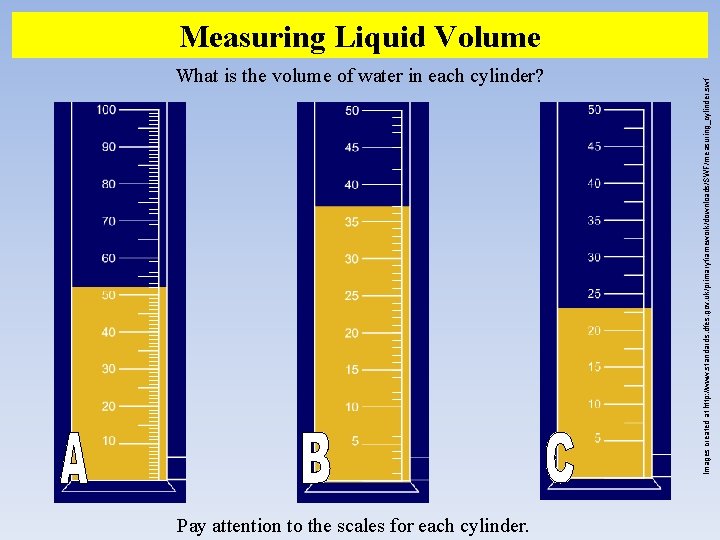
What is the volume of water in each cylinder? Pay attention to the scales for each cylinder. Images created at http: //www. standards. dfes. gov. uk/primaryframework/downloads/SWF/measuring_cylinder. swf Measuring Liquid Volume

Measuring Solid Volume 9 cm We can measure the volume of regular object using the formula length x width x height. 8 cm _____ X _____ = _____ We can measure the volume of irregular object using water displacement. Amount of H 2 O with object = ______ About of H 2 O without object = ______ Difference = Volume = ______ Click here for an online activity about volume. Choose Lessons Volume & Displacement http: //resources. edb. gov. hk/~s 1 sci/R_S 1 Science/sp/e n/syllabus/unit 14/new/testingmain 1. htm 10 cm



Measuring Temperature • http: //science. wonderhowto. com/how-to/use -thermometer-determine-temperature 212256/

• Thermometers are very fragile and must be handled with care. Read the following rules for the proper use of a thermometer. • 1. Never "shake down" a thermometer to reset it. • 2. Never use a thermometer to stir a liquid. • 3. Never allow a thermometer to touch the bottom of a container that is being heated.

Safety Video • http: //www. shmoop. com/video/chemistry-11 -labsafety? tag=6 FDC 1073429 B 473 D 9451 D 819837 82473

False • You only need to use safety goggles when working with acids.

False • If you are unsure about the identity of a chemical, it is okay to taste and smell it.

False • Only major accidents should be reported to the teacher.

True • All test tubes need to be thoroughly cleaned with soap and water before re-using them.

True • The proper way to smell a chemical is to carefully waft the vapors towards your nose.
- Slides: 44