Lab Activity 8 Isolation of Cholesterol From Egg

Lab Activity 8 Isolation of Cholesterol From Egg Yolk IUG, 2016 -2017 Dr. Tarek M. Zaida 1
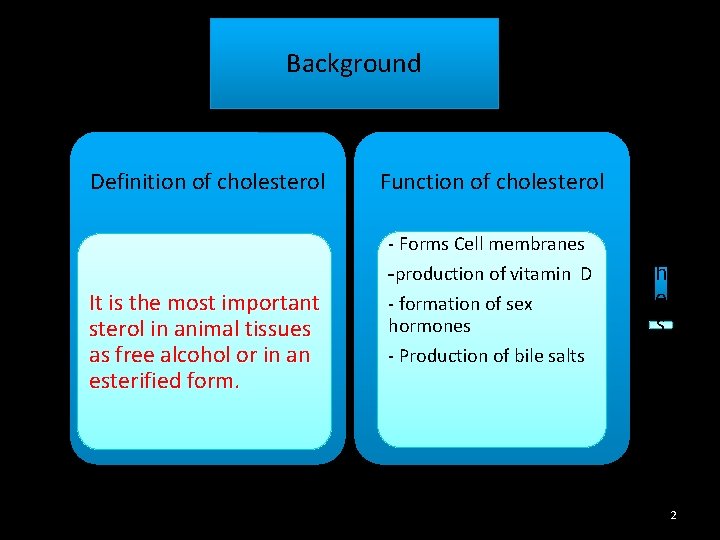
Background Function of cholesterol - Forms Cell membranes It is the most important sterol in animal tissues as free alcohol or in an esterified form. -production of vitamin D - formation of sex hormones - Production of bile salts e r Definition of cholesterol S y n t h e s i s 2
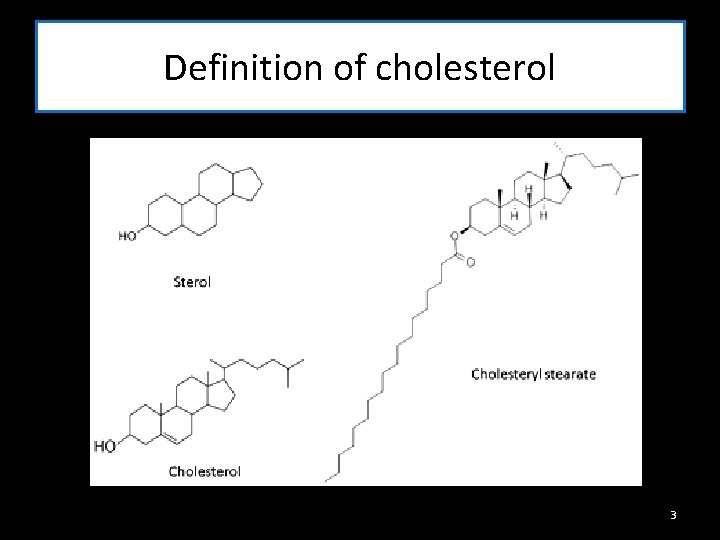
Definition of cholesterol 3

Source of Cholesterol • It is synthesized in the body from acetyl-Co. A (1 gm/day, cholesterol does not exist in plants) mostly in Liver. • and is also taken in the diet (0. 3 gm/day as in, butter, milk, egg yolk, brain, meat and animal fat). 4
Good & Bad cholesterol LDL-C (bad) Cholesterol HDL-C (good) 5
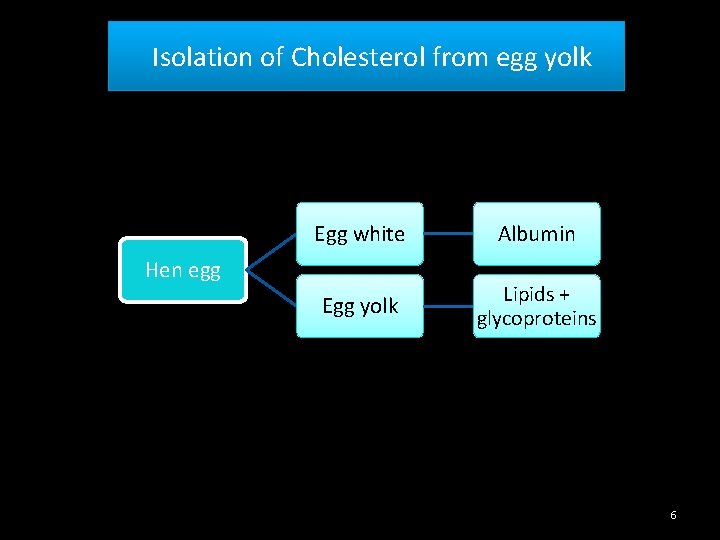
Isolation of Cholesterol from egg yolk Egg white Albumin Egg yolk Lipids + glycoproteins Hen egg 6
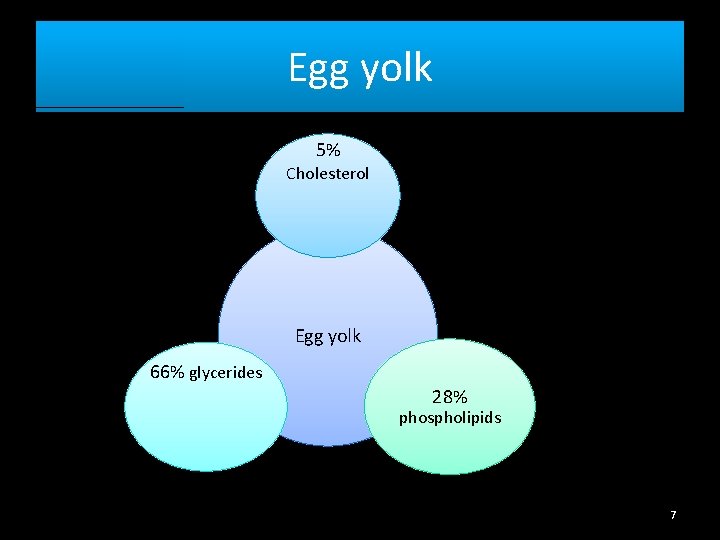
Egg yolk 5% Cholesterol Egg yolk 66% glycerides 28% phospholipids 7

Experiment Reagents and instruments • • A hard boiled hen egg Acetone, Ice, Water bath, Erlenmeyer flask, Funnel, Filter paper, 250 ml beaker 8

1. Extraction 1. Peal the hard boiled egg and remove the egg white. 2. In a 250 ml beaker weigh a ½ egg yolk and mash it. 3. To the mashed egg yolk add a 25 ml of acetone and keep stirring for 5 min. 9

4. Allow the solid to settle and carefully remove the acetone (the above liquid phase) and transfer it to a clean 100 ml Erlenmeyer flask and cover it tightly. 5. To the remained solid phase add a new 25 ml of acetone and repeat step 4. - Now you have collected twice the 25 ml of acetone extract (it contains the cholesterol). 10

2. Isolation & Purification 1. Using a funnel and a filter paper, filter the 50 ml of acetone extract into a clean 100 ml beaker. 2. Evaporate the acetone by placing the beaker on the steam bath in the hood until about 10 ml of extract is left. 11

3. Cover the warm acetone extract and immediately transfer it on ice. - Let it stand for 20 min. - A white precipitate will form (this is the crude cholesterol). - Remove the flask from ice. - Get rid of the liquid phase. 12

4. To the cholesterol precipitate add 15 ml of acetone and stir at room temp. Note that not all precipitate will dissolve (the contaminating phospholipids remain insoluble). 13

5. Weigh a clean, dry watch glass. Filter the dissolved cholesterol solution into the preweighed watch glass. Allow the acetone to evaporate in the hood. The remained crystals is the cholesterol. 6. Weigh the watch glass with cholesterol. Calculate the percentage of cholesterol in egg yolk. 14
- Slides: 14