Lab 8 CENTRIFUGATION KAUFaculty of Science Biochemistry department

Lab (8): CENTRIFUGATION KAU-Faculty of Science- Biochemistry department Analytical biochemistry lab (Bioc 343) 2012 T. A Nouf Alshareef nf. shareef@hotmail. com

Introduction • particles or cells in a liquid suspension, will settle at the bottom of tube due to gravity (1 x g) during long period to time. • However, the length of time required for such separations is impractical. • Other particles which are very small in size, will not separate at all • So, high centrifugal force though method called centrifugation must be used in these cases.
Simply, Centrifugation >>>>> Increasing the effect of gravity

Sedimentation • Is the motion of molecules in solutions or, particles in suspensions, in response to an external force such as gravity, centrifugal force or electric force. • Centrifugation increase the rate of sedimentation by spinning the samples and creating a centrifugal force acts on particles in an instrument called a centrifuge. The rate of sedimentation depends on: 1. Properties of molecules: size, shape, density 2. Properties of solvent or gradient material: density, viscosity, temperature 3. Interaction between solute molecules and solvent or gradient material

Centrifugation Theoretical bases of centrifugation: • When suspension is rotated at certain speed or Revolution Per Minute (RPM) centrifugal force causes the particles to move away from the axis of rotation. • The force on particles is called relative central force (RFC)

Relative centrifugal force: • Relative centrifugal force (RCF) is the measurement of the force applied to a sample within a centrifuge. • Centrifugal force is measured as X g gravity. • An RCF of 500 Xg indicates that the centrifugal force applied is 500 times greater than earth gravitational force • This can be calculated from the speed (RPM) and the rotational radius (cm) using the following calculation.

It is important to remember that : • Centrifugal force or (RCF) is measured as X g gravity. • Speed of the rotation in centrifuge is expressed as Revolutions Per Minutes (RPM).

Centrifuge • is apparatus that rotates at high speed and by centrifugal force to separates substances of different densities (two liquids or liquid-solid) • materials with the highest density travel towards the bottom of centrifuge tube at higher rate faster than normal gravity. • Centrifuge is considered as device that increase the effects of earth's gravitational pull.

• Sample is placed in a centrifuge tube which is then placed in rotor. • Separates into: pellet (precipitate) supernatant or supernatant (clarified liquid above pellet) Terms: • Pellet: (denser material) is settled at bottom of tube. • supernatant (liquid) is then either decanted from tube without disturbing the precipitate , or withdrown with pasteurpipette.

Common examples of the use of centrifugal force 1. washing machine: centrifugal force generated in the "spin" cycle that forces water out of the fabric to facilitate faster drying 2. To separate cream from whole milk 3. To separate uranium 235, to produce nuclear energy

Centrifuge components: Every centrifuge contains : 1. Drive shaft ﻋﻤﻮﺩ ﺍﻟﺪﻭﺭﺍﻥ 2. Electric mortor ( Rotating part) 3. Rotor

Centrifuge Rotors • rotor is positioned centrally on the drive shaft of centrifuge • samples tubes are inserted in rotor. • Is dense metal containing 2, 4, 6 or more numbered wells. • stored in refrigeration units to keep them at or near operating temperature. Two types of rotors: 1 - fixed angle Rotor 2 - Swinging Bucket Rotor.

1. Fixed Angle Rotor: • Is most commonly used, consist of a solid piece of metal with 6 -12 holes at angle between 20 o -45 o. • tubes are fixed at the angle of rotor cavity. • It can hold larger volumes than swinging bucket rotor but the pellet forms less evenly.
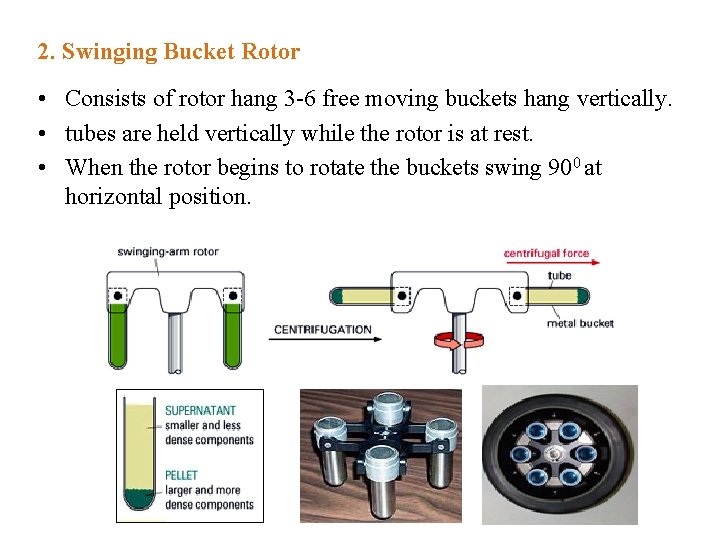
2. Swinging Bucket Rotor • Consists of rotor hang 3 -6 free moving buckets hang vertically. • tubes are held vertically while the rotor is at rest. • When the rotor begins to rotate the buckets swing 900 at horizontal position.

Centrifuge tubes

Types of centrifuges • The major differences between centrifuge types are: speed and capacity. • There at least five types of centrifuge: 1. Tabletop/clinical centrifuge or microcentrifuge (13. 000 rpm) 2. Low-speed centrifuge ( maximum speed 5000 rpm ) 3. High-speed centrifuge ( speed up to about 25, 000 rpm ), 4. Ultracentrifuge (70, 000 -100. 000 rpm) 5. Geotechnical centrifuge

Typical biochemistry lab have three different centrifuges: 1. Microcentrifuge: (microfuge centrifuges or mini centrifuge or bench top centrifuge) • • Capacity: spinning 1 -2 ml centrifuge tubes (small quantities) Speeds: up to 12000 - 13000 rpm. (low to medium) It has small light rotor speed up and slow down rapidly. used for: separation of small samples, separation of DNA, RNA and proteins that need isolation.

2. High-speed centrifuge: • Speed: up to about 25, 000 rpm • Capacity: various sizes, depending on the rotors (the larger the rotor, the slower the maximum speed). • Usually rotors at 4 o. C, so called cold centrifuge

3. Ultracentrifuge: • Capacity: up to 60 ml • Speeds: up to 600, 000 rpm (high speed). • can be very dangerous if maintenance is not performed regularly because of high speeds. • centrifuge chamber must be refrigerated and evacuated so that friction does not heat up the suspension.

Types of Centrifugal Separations: 1. Differential centrifugation: 2. Density gradient centrifugation:
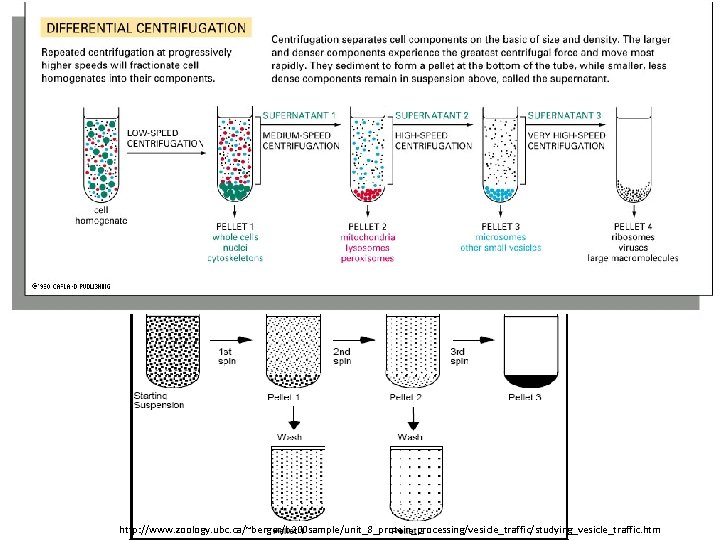
http: //www. zoology. ubc. ca/~berger/b 200 sample/unit_8_protein_processing/vesicle_traffic/studying_vesicle_traffic. htm
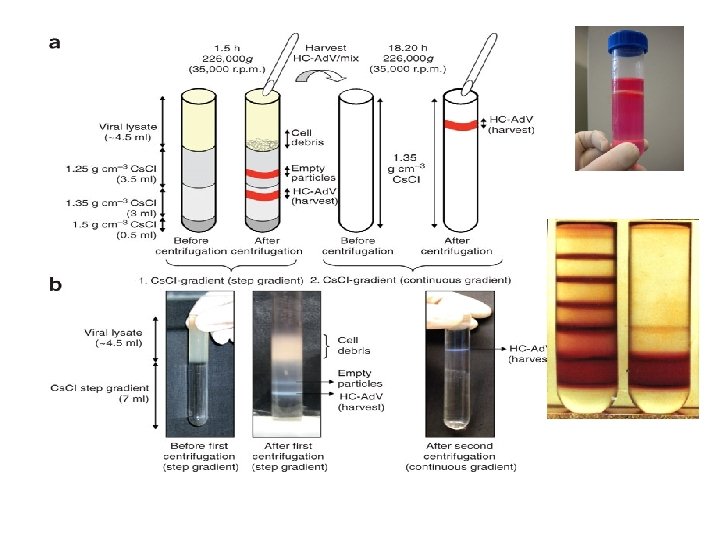

DNA Extraction from 0. 22 μM Sterivex Filters and Cesium Chloride Density Gradient Centrifugation
Lab practice separation of liver proteins by centrifugation Aim: • Liver proteins extraction using centrifugation techniques

5 g + 10 ml cold water Homogenate using mortar • Filter using muslin cylinder complete volume to 20 ml by cold water (homogenate) 0. 1 ml (Fraction 0) • Centrifuge 3500 rpm, 15 min • Collect supernante, measure its volume 0. 1 ml (Fraction 1) • Add 10 ml of (0. 2 M) Na. Cl, Centrifuge 3500 rpm, 15 min • Collect supernante, measure its volume 0. 1 ml (Fraction 2) • Add 10 ml of (1 M) Na. Cl Centrifuge 3500 rpm, 15 min • Collect supernante, measure its volume 0. 1 ml (Fraction 3) • Add 10 ml of (1 M) Na. Cl+detergent, Centrifuge 3500 rpm, 15 min • Collect supernante, measure its volume 0. 1 ml (Fraction 4)

Protein Biuret assay • • 0. 1 ml sample complete it to 4 ml by H 2 O Add 6 ml biuret reagent Incubates for 5 min at 37 o. C Read abs. at 540 nm
- Slides: 26