Lab 6 Spectrophotometry Objectives 1 Understand light photon

Lab. 6 Spectrophotometry Objectives 1. Understand light, photon, and quantum. 2. Learn how to use a spectrophotometer. 3. Construct and use a standard curve to determine the unknown concentration of a dissolved chemical.

Review: Detection of Monomer and Polymer (Qualitative analysis) Using Benedict’s solution detects monossaccharides (sugars) Using Lugol’s iodine solution detects polysaccharides Using Biuret reagent detects proteins Using Sudan IV detects lipids What about Quantitative?
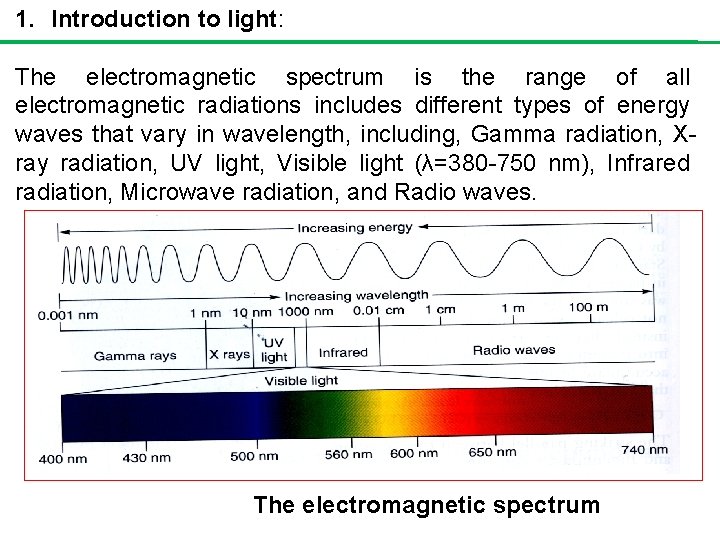
1. Introduction to light: The electromagnetic spectrum is the range of all electromagnetic radiations includes different types of energy waves that vary in wavelength, including, Gamma radiation, Xray radiation, UV light, Visible light (λ=380 -750 nm), Infrared radiation, Microwave radiation, and Radio waves. The electromagnetic spectrum

Light is an electromagnetic wave-that is, that light moved through the air as oscillating electric and magnetic fields. Photon: fundamental particle of light with an energy content that is inversely proportional to the wavelength of the light: shortwavelength light contains photons of higher energy than longwavelength light. Quantum: the unit of energy stored in photon. Each photon carries indivisible packet of energy-quantum. Energy: the energy amount is proportional inversely with wavelength of light
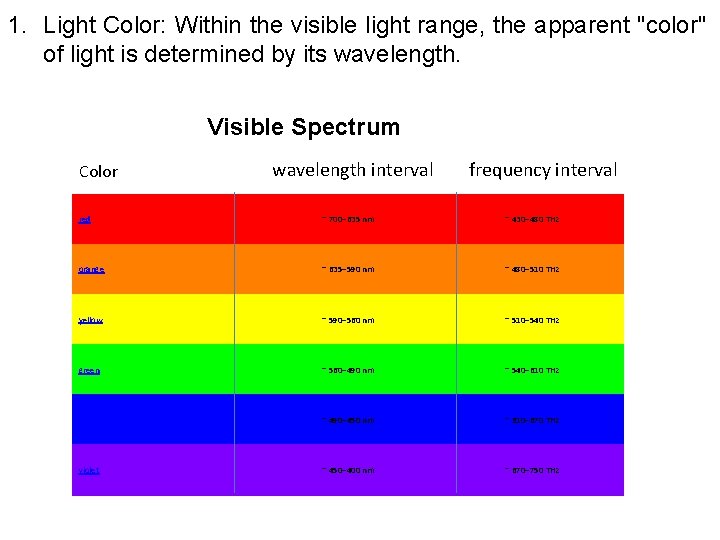
1. Light Color: Within the visible light range, the apparent "color" of light is determined by its wavelength. Visible Spectrum Color wavelength interval frequency interval red ~ 700– 635 nm ~ 430– 480 THz orange ~ 635– 590 nm ~ 480– 510 THz yellow ~ 590– 560 nm ~ 510– 540 THz green ~ 560– 490 nm ~ 540– 610 THz blue ~ 490– 450 nm ~ 610– 670 THz violet ~ 450– 400 nm ~ 670– 750 THz

2. Substance color through our eyes Different substances absorb and reflect different wavelengths of light, and the light color on a substance is determined by reflecting wavelength and absorbance of the light. Some objects may also transmit light or emit light themselves, which also contribute to the color. § § A blue shirt appears to be blue because it absorbs all the wavelengths of white light except for those in the blue range, which it reflects. A glass of water looks transparent and clear because it does not absorb much visible light (however, it does absorb wavelengths outside of the visible range such as ultraviolet and infrared). A black cloth absorbs all the wavelengths of the visible light and gives black color. Can you guess why wearing white clothes in the sun will keep you slightly cooler than wearing black clothes ?
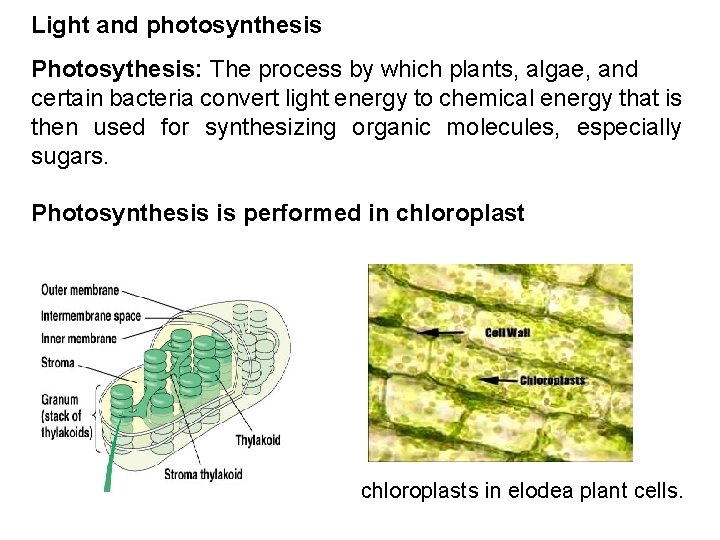
Light and photosynthesis Photosythesis: The process by which plants, algae, and certain bacteria convert light energy to chemical energy that is then used for synthesizing organic molecules, especially sugars. Photosynthesis is performed in chloroplasts in elodea plant cells.

Please explain why these flowers show different color and what possible arrange of wavelengths of light are responsible each of these flower colors? A B D E C F

2. Spectrophotometer: A spectrophotometer provides a quantitative way of measuring the absorbance and transmission properties of a solution. § § Light source: the light source in a spectropotometer produces white light, a combination of all visible wavelengths. Filter: the filter of spectrophotometer is adjusted to select the wavelength that you wish to pass through the sample Sample: sample is a solution contained in a clear test tube or cuvette made of glass or quartz. Blank: Blank is a test tube or cuvette containing only the solvent used to dissolve the chemical you are analyzing.

§ Absorbance: absorbance is the amount of radiation retained by the sample (Absorbance is expressed in units of optical density (O. D. ) Absorbance values are directly proportional to the concentration of the substance in solution: the more solutes, the higher absorbance and the less transmittance. § Transmittance (It): transmittance is the amount of radiation passing through the sample. Percent Transmittance (T) = (It /I 0) x 100 I 0 = original light intensity (entering light) It = transmitted light intensity (exiting light) Absorbance = log(1/T) = log 10 (I 0 /It )

3. Beer-Lambert’s Law The absorbance (A) is a better way to measure the concentration of a light-interfering solute because in dilute solutions, A is directly proportional to the solute’s molar concentration (there is linear relationship). This is known as Beer-Lambert’s Law: A=axbxc A = absorbance, a = coefficient, b = length of sample c = concentration of sample. When working in concentration units of molarity, the Beer-Lambert law is written as: A (absorbance) = ε x l x c ε = coefficient, l = length of sample (width of tube, usually 1 cm), c = molar concentration of sample
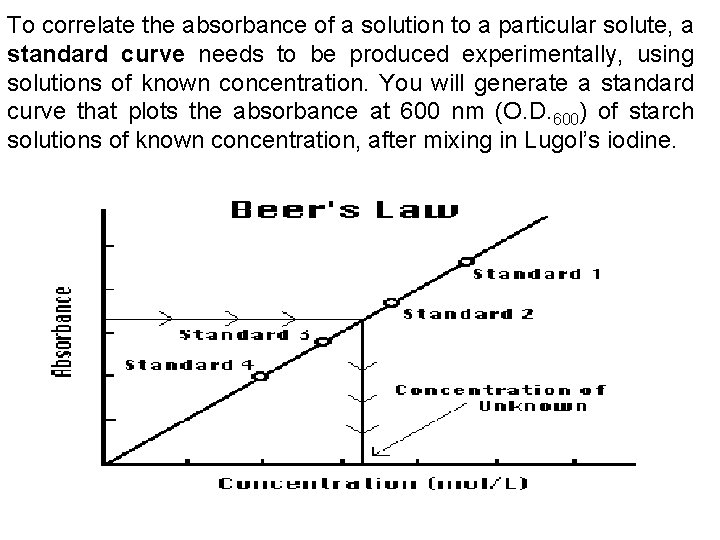
To correlate the absorbance of a solution to a particular solute, a standard curve needs to be produced experimentally, using solutions of known concentration. You will generate a standard curve that plots the absorbance at 600 nm (O. D. 600) of starch solutions of known concentration, after mixing in Lugol’s iodine.

1. Constructing a standard curve Turn on spectrophotometer, and select a wavelength of 600 nm (Step 1 and 2 of previous page). 1. Prepare 6 clean test tubes 2. Pipet the volumes of the solutions indicated in Table 1 (see handout). 3. Measure the absorbance of tubes 1 -5. 4. Plot the standard curve data (absorbance as a function of starch concentration) using the graph paper provided. Label the X-Y axis of the graph and give it a title. Using a ruler, draw a ‘best fit’ straight line, making sure that the line crosses the (0, 0) point and that it fits most data point (it’s ok if it doesn’t match the last point well). 2. Determine concentration of unknown starch solution
Absorbance Concentration

Calibrating the spectrophotometer: The spectrophotometer is first calibrated with the blank so the instrument indicates no absorbance (100% transmittance), as the light-interfering solutes we are interested in measuring are absent in the blank and do not contribute reduce light transmission. For very precise measurements, scientists will even use pairs of tubes (or cuvettes) that have been specially designed to transmit light in the same manner, so that everything is as similar as possible between the sample and blank solutions.
- Slides: 15