LAB 6 Enzymes Introduction Kinetic reactions all chemical

LAB 6: Enzymes

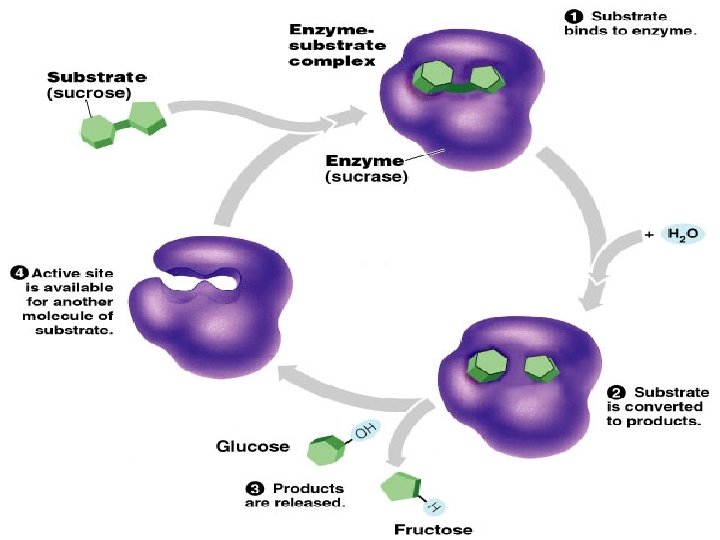
Introduction • Kinetic reactions: • all chemical reactions in living organisms require enzymes to work For building , breaking down molecules or speed up the reactions. • Enzymes are A biological catalyst. Usually a globular protein molecule produced by living organisms that can speed up a specific chemical reaction without itself being destroyed or changed in any way. • They are produced by the living organism and are usually present in only very small amounts in various cells. • Lock and Key Hypothèses • Active site: The region of an enzyme molecule which binds the substrate and carries out the catalytic reaction

Enzymes properities • Enzymes aren’t used up • Enzymes are not changed by the reaction • re-used again for the same reaction with other molecules. • Most enzymes are Proteins (tertiary and quaternary structures) • Act as Catalyst to accelerates a reaction • Are specific for what they will catalyze
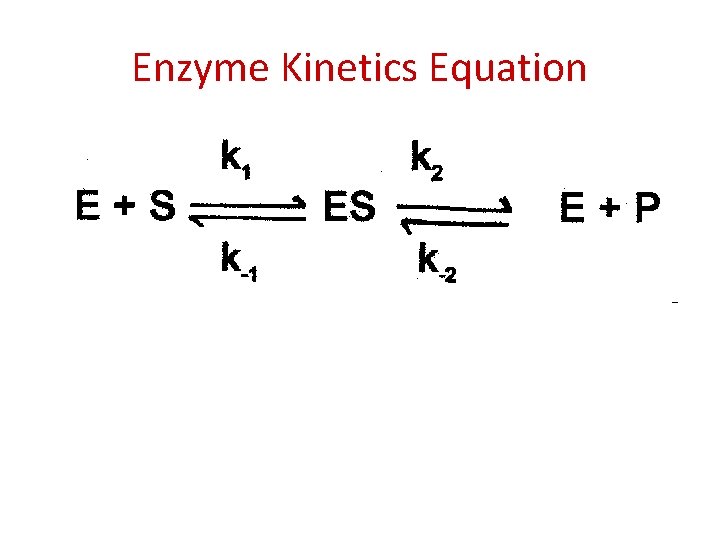
Enzyme Kinetics Equation
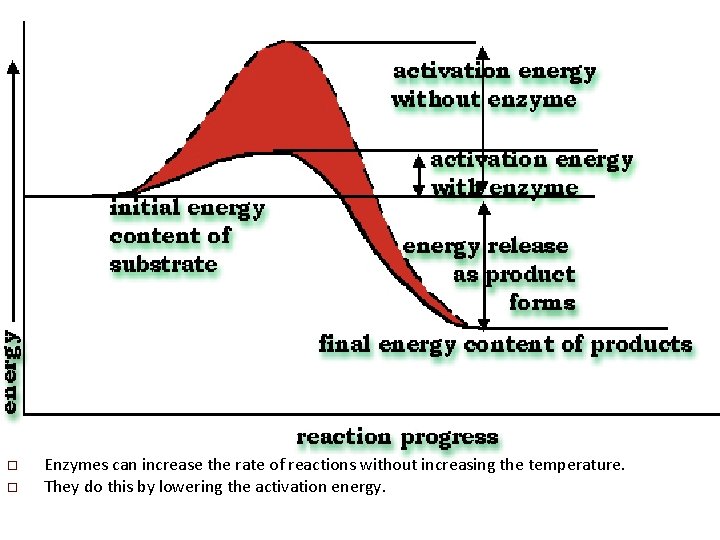
o o Enzymes can increase the rate of reactions without increasing the temperature. They do this by lowering the activation energy.

Factors affecting Enzymes o o substrate concentration p. H temperature inhibitors

PH • Extreme p. H levels will produce denaturation – most human enzymes = p. H 6 -8 • depends on where in body • pepsin (stomach) = p. H 3 • trypsin (small intestines) = p. H 8 • The structure of the enzyme is changed • The active site is distorted and the substrate molecules will no longer fit in it
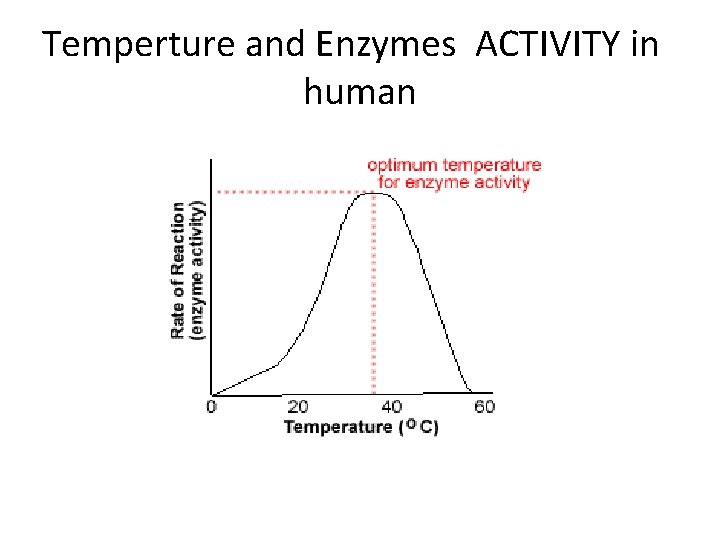
Temperture and Enzymes ACTIVITY in human

Applications o o o Many are a lot lower, cold water fish will die at 30°C because their enzymes denature A few bacteria have enzymes very high temperatures up to 100°C Most enzymes however are fully denatured at 70°C
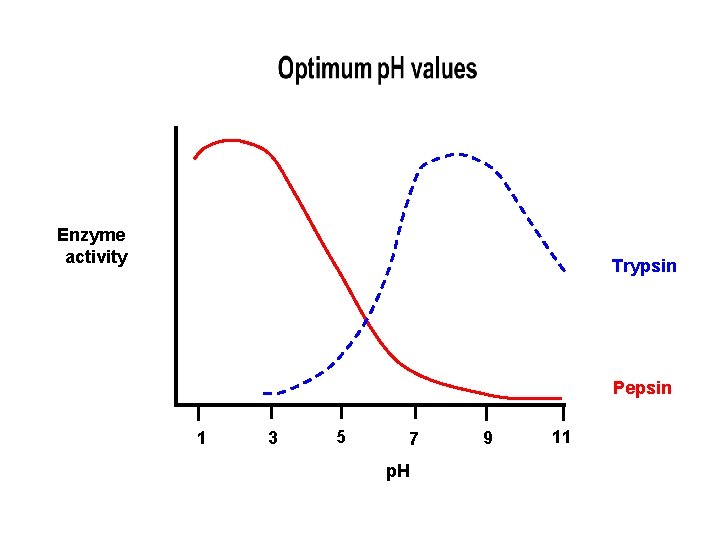
Enzyme activity Trypsin Pepsin 1 3 5 7 p. H 9 11

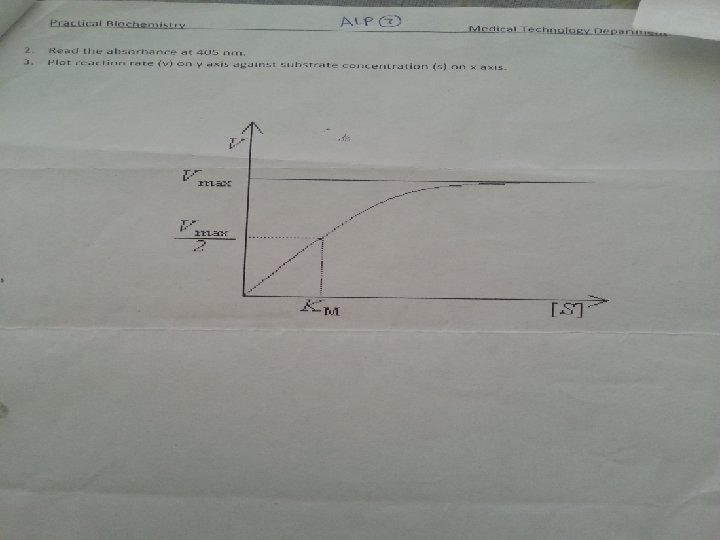
• Michaelis and menten at low substrate concentrations, the enzyme is not saturated with the substrate and the reaction is not proceeding at maximum velocity whereas when the enzyme is saturated with substrate, maximum velocity is observed.

Inhibitors are chemicals that reduce the rate of enzymic reactions. o The are usually specific and they work at low concentrations. o They block the enzyme but they do not usually destroy it. • Irreversible inhibitors: Combine with the functional groups of the amino acids in the active site, irreversibly. • reversible Competitive inhibitors: These compete with the substrate molecules for the active site. Non competitive inhibitors: when the inhibitor binds somewhere else on the enzyme molecules reducing its efficiency. o
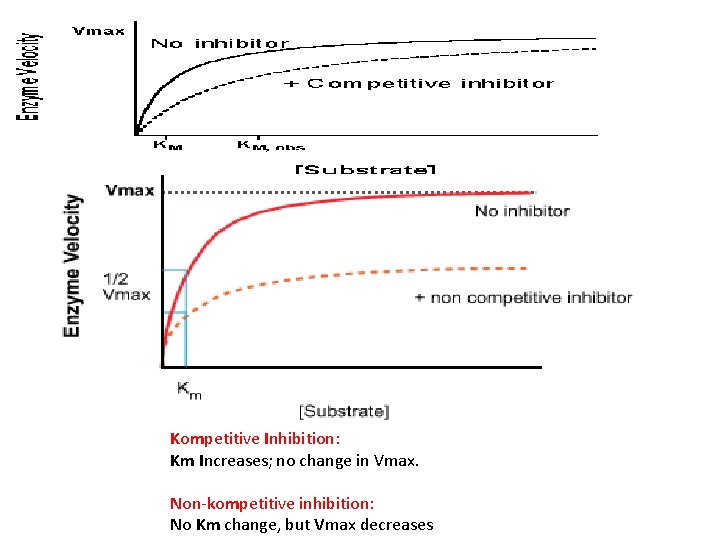
Kompetitive Inhibition: Km Increases; no change in Vmax. Non-kompetitive inhibition: No Km change, but Vmax decreases

• Therefore, the amount of enzyme inhibition depends upon the inhibitor concentration, substrate concentration, and the relative affinities of the inhibitor and substrate for the active site. • The net effect of a non competitive inhibitor is to change the shape of the enzyme and thus the active site, so that the substrate can no longer interact with the enzyme to give a reaction.
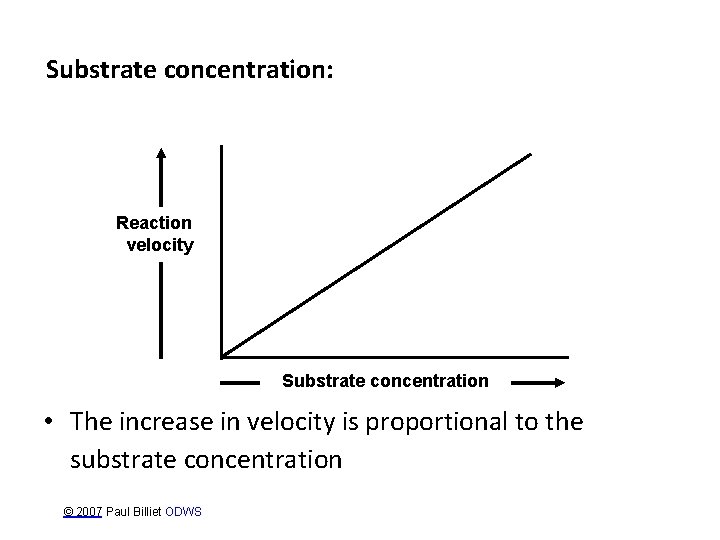
Substrate concentration: Reaction velocity Substrate concentration • The increase in velocity is proportional to the substrate concentration © 2007 Paul Billiet ODWS
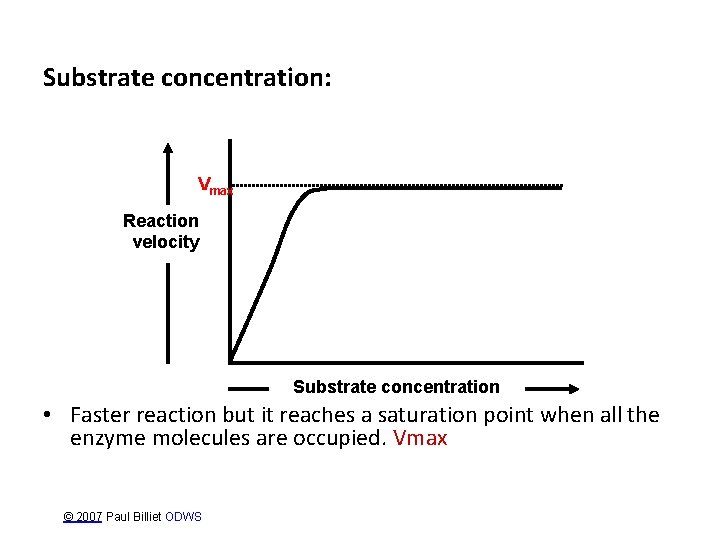
Substrate concentration: Vmax Reaction velocity Substrate concentration • Faster reaction but it reaches a saturation point when all the enzyme molecules are occupied. Vmax © 2007 Paul Billiet ODWS

K m: (Michaelis constant) The substrate concentration at which an enzyme catalyzed reaction proceeds at half the maximum velocity. Or the concentration of the substrate at which half the active sites of the enzyme are occupied by the substrate. a high Km value indicates weak binding between the enzyme and substrate, whereas a low Km indicates strong binding.

• V max: (Maximum velocity) The maximum initial velocity of an enzyme catalysed reaction; determined by increasing the substrate [S] until a constant rate of product formation is achieved (i. e. saturating substrate levels).

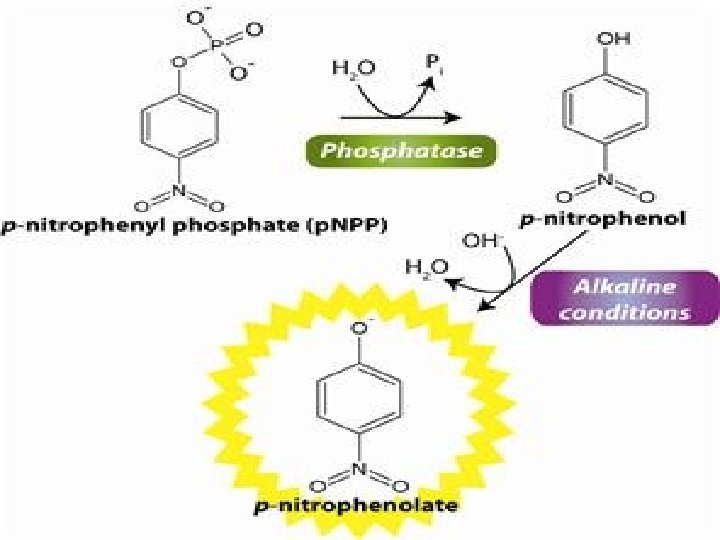
ALKALINE PHOSPHATASE • Is a member of the phosphomonoestrases group. Which are highly speicfic amd act an only one substrate, alkaline phosphatase has a broad substrate specificity and is named alkaline phosphatase because its ph optimum is usually around 9 but other broad spectrum phosphoesterases with ph optima less than 7 are termed acid phosphatases.

Determination ALP in vitro • Alkaline phosphatase activity by following the rate of formation of products. • ALP catalyzes in alkaline medium the transfer of the phosphate group from 4 nitrophenylphosphate to 2 amino 2 methyle propanol, librating 4 nitrophenol • The catalytic concentration is determined from the rate of 4 nitrophenol formation, measured at 400 nm. • The 4 nitrophenol is bright yellow but other reactants and products are colorless in aqueous solution.

• The measurement of ALP activity in vitro is based on artificial substrate pnitrophenylphosphate. • The intensity of yellow color in the reaction solution thus indicates the degree to which enzyme has acted upon substrate. (how much enzyme acted on substrate).

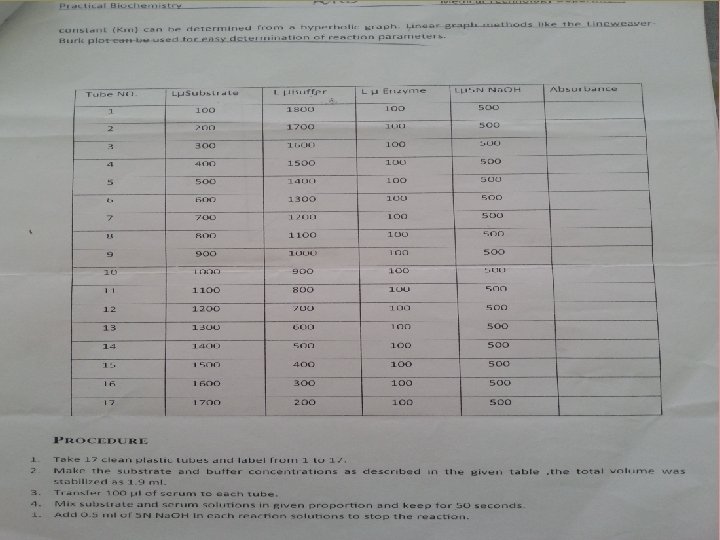
Study enzyme kinetics • Enzyme kinetics: the study of the rate at which an enzyme works. • To examine it, when the substrate available to the enzyme one would do the following: 1. set up the series of tubes containing graded concentrations of substrate 2. At time zero, a fixed amount of the enzyme preparation is added 3. Over the next few minutes, the concentration of product formed, is measured. If the product absorbs light, we can easily do this in a spectrophotometer 4. Early in the run, when the amount of substrate is in substantial excess to the amount of enzyme, the rate we observed is the initial of velocity Vi.
- Slides: 26