Lab 6 Cellular energetics Objective to examine respiration

Lab 6 - Cellular energetics Objective: to examine respiration in yeast and rat mitochondria Techniques: Measure effects of substrates and inhibitors on oxygen consumption in yeast and rat mitochondria using an oxygen polarograph
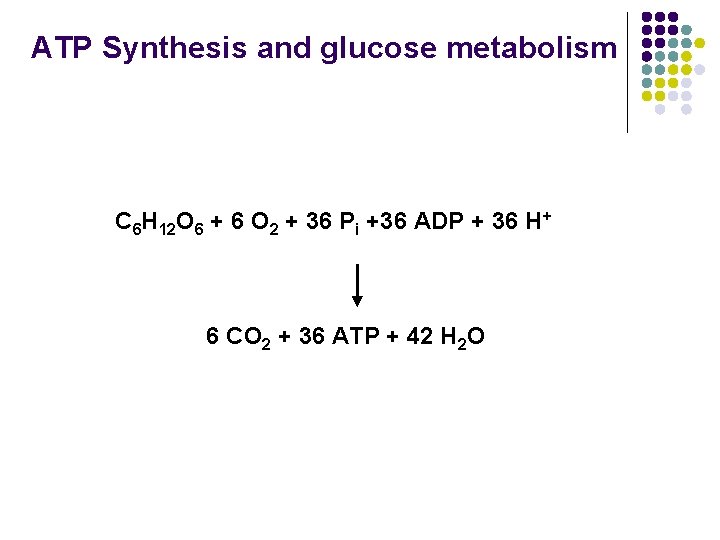
ATP Synthesis and glucose metabolism C 6 H 12 O 6 + 6 O 2 + 36 Pi +36 ADP + 36 H+ 6 CO 2 + 36 ATP + 42 H 2 O
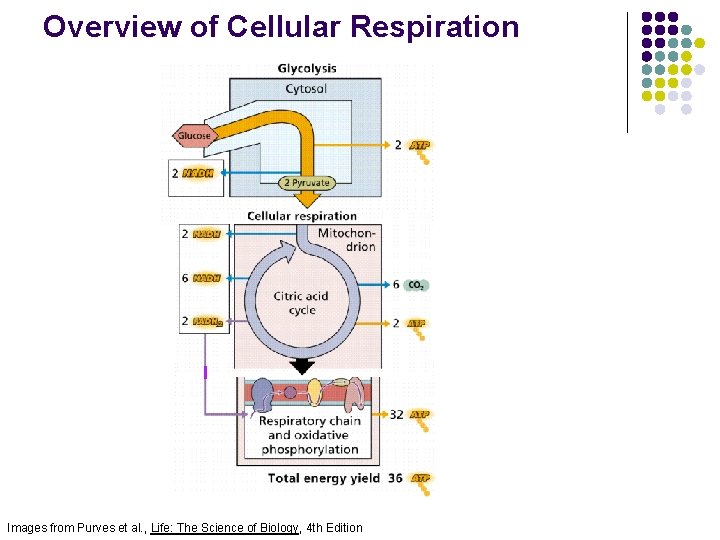
Overview of Cellular Respiration Images from Purves et al. , Life: The Science of Biology, 4 th Edition
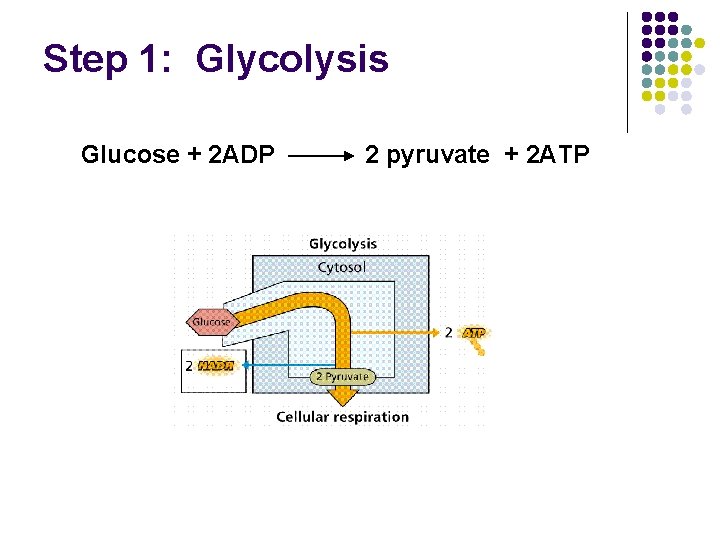
Step 1: Glycolysis Glucose + 2 ADP 2 pyruvate + 2 ATP
![Glycolysis • Occurs in the cytosol Hi [ATP] • Glucose metabolized to 2 pyruvate Glycolysis • Occurs in the cytosol Hi [ATP] • Glucose metabolized to 2 pyruvate](http://slidetodoc.com/presentation_image/24f85a12c85d3b300ac8ecfbc8810f96/image-5.jpg)
Glycolysis • Occurs in the cytosol Hi [ATP] • Glucose metabolized to 2 pyruvate + 2 ATP • High [ATP] inhibits phosphofructokinase (PFK) • High [ADP] stimulates PFK • Pasteur Effect: Increase in the rate of carbohydrate breakdown that occurs when switched from aerobic to anaerobic conditions Fig. 16 -3
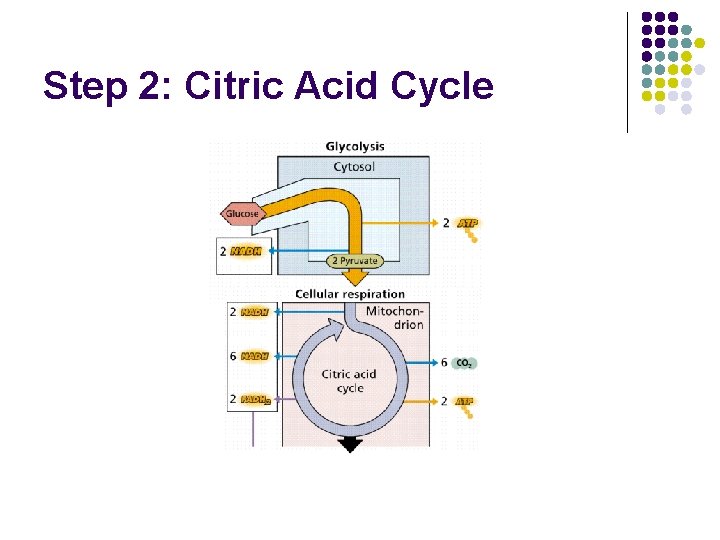
Step 2: Citric Acid Cycle
Mitochondria
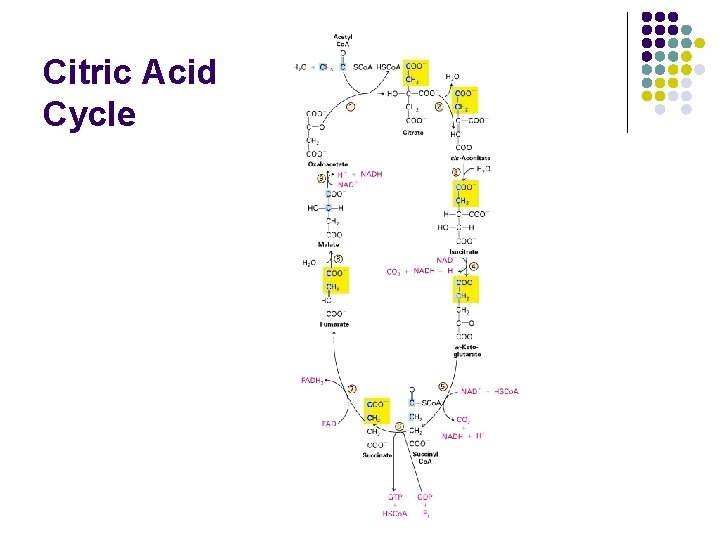
Citric Acid Cycle
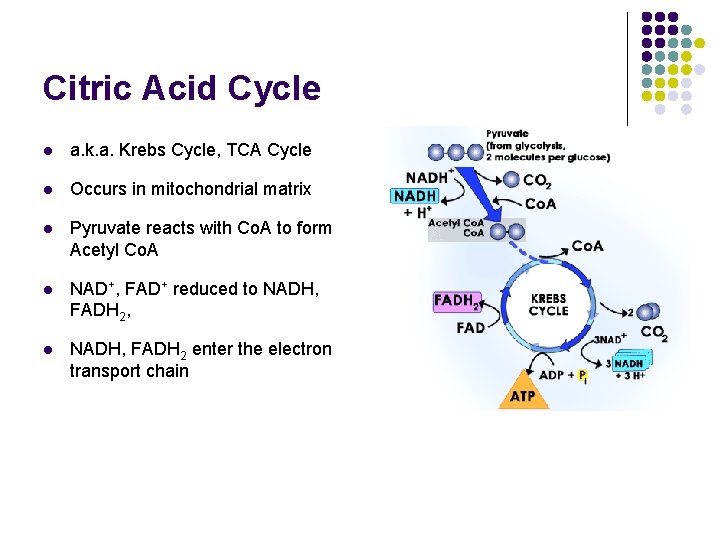
Citric Acid Cycle l a. k. a. Krebs Cycle, TCA Cycle l Occurs in mitochondrial matrix l Pyruvate reacts with Co. A to form Acetyl Co. A l NAD+, FAD+ reduced to NADH, FADH 2, l NADH, FADH 2 enter the electron transport chain
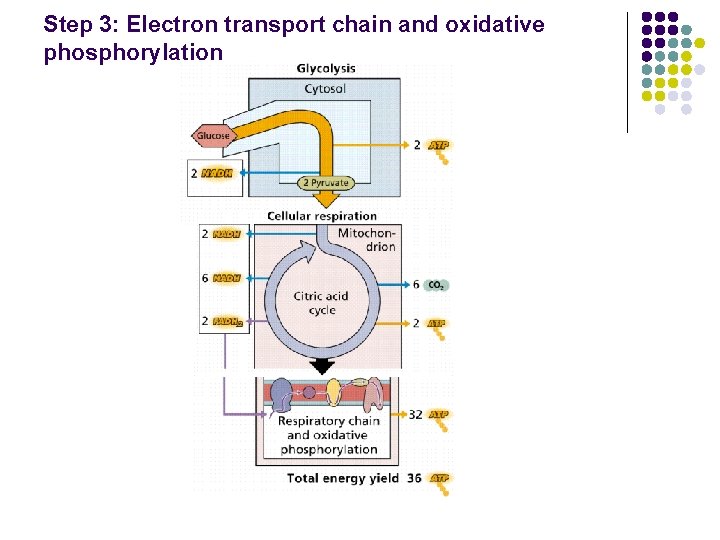
Step 3: Electron transport chain and oxidative phosphorylation

Oxidative Phosphorylation Oxidative phosphorylation is the process by which the energy stored in NADH and FADH 2 is used to produce ATP. A. Oxidation step: electron transport chain 1 O 2 2 1 FADH 2 + O 2 2 NADH + H+ + NAD+ + H 2 O FAD + H 2 O B. Phosphorylation step ADP + Pi ATP
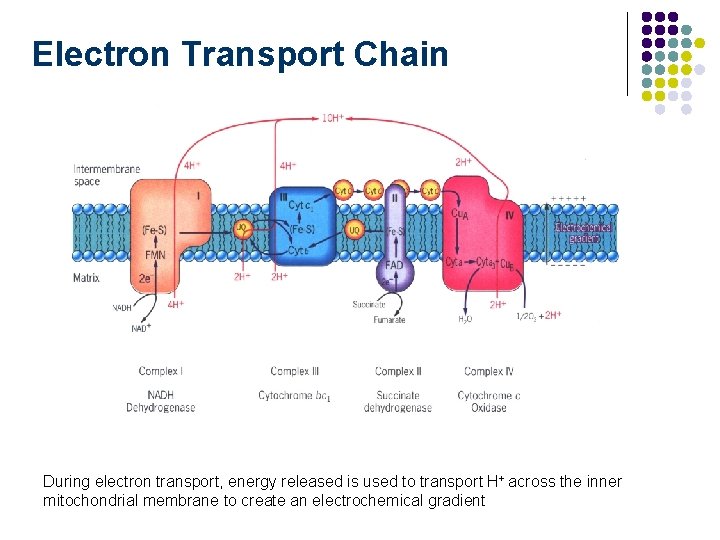
Electron Transport Chain During electron transport, energy released is used to transport H+ across the inner mitochondrial membrane to create an electrochemical gradient Fig. 16 -19
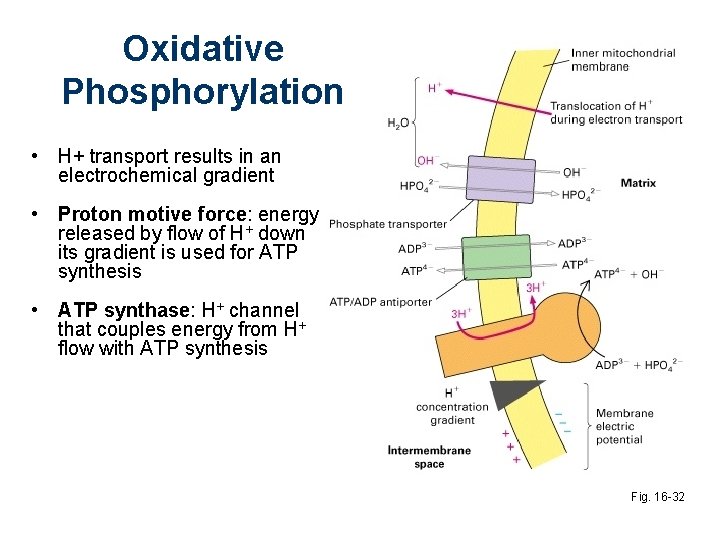
Oxidative Phosphorylation • H+ transport results in an electrochemical gradient • Proton motive force: energy released by flow of H+ down its gradient is used for ATP synthesis • ATP synthase: H+ channel that couples energy from H+ flow with ATP synthesis Fig. 16 -32
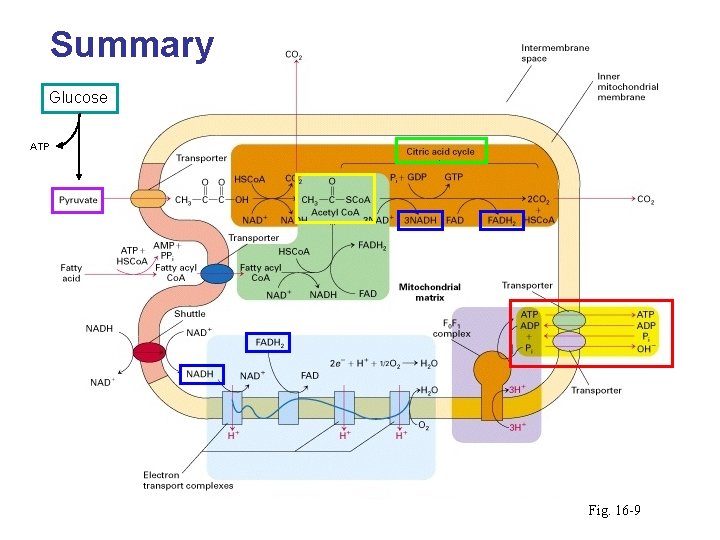
Summary Glucose ATP Fig. 16 -9

This week’s lab Day one: Yeast respiration • Goal: learn how to measure O 2 consumption • Compare O 2 consumption by normal and starved yeast Day two: Mitochondria • Examine the effects of various inhibitors and substrates on the rate of respiration • Determine the identity of your unknown (think what substrates you need to add and in what order together with the unknown
![Inhibitors of Glycolysis Hi [ATP] Applicable to yeast respiration, not purified mitochondria—why? N-ethylmaleimide Fig. Inhibitors of Glycolysis Hi [ATP] Applicable to yeast respiration, not purified mitochondria—why? N-ethylmaleimide Fig.](http://slidetodoc.com/presentation_image/24f85a12c85d3b300ac8ecfbc8810f96/image-16.jpg)
Inhibitors of Glycolysis Hi [ATP] Applicable to yeast respiration, not purified mitochondria—why? N-ethylmaleimide Fig. 16 -3
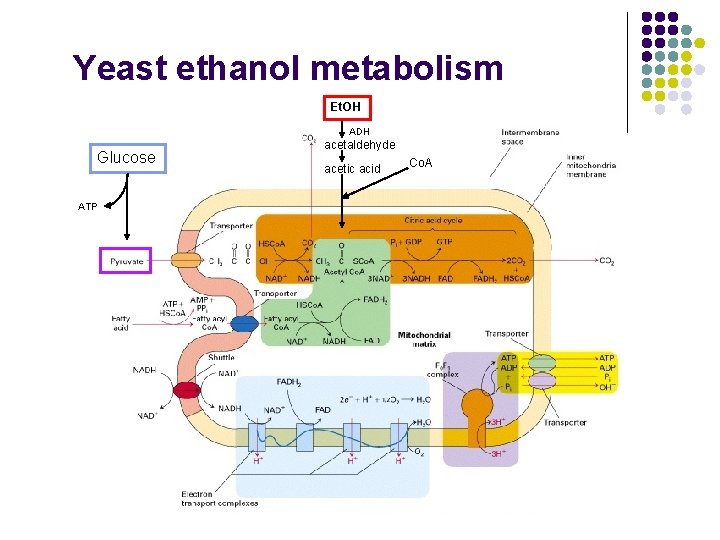
Yeast ethanol metabolism Et. OH ADH Glucose ATP acetaldehyde acetic acid Co. A
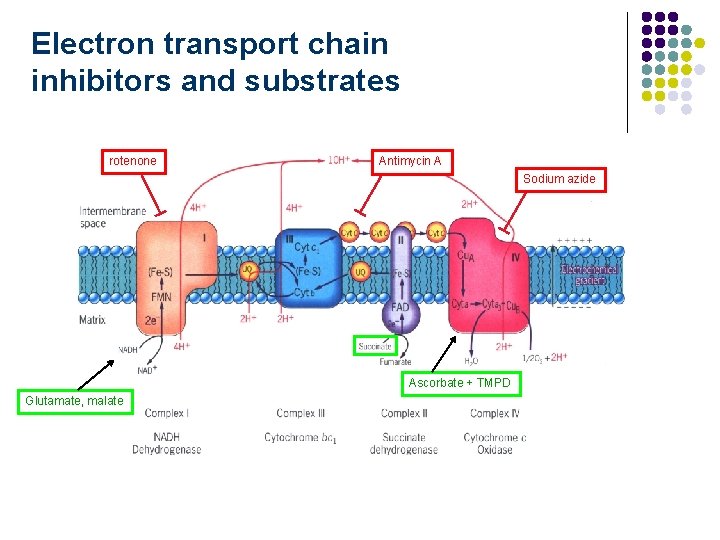
Electron transport chain inhibitors and substrates rotenone Antimycin A Sodium azide Ascorbate + TMPD Glutamate, malate Fig. 16 -19
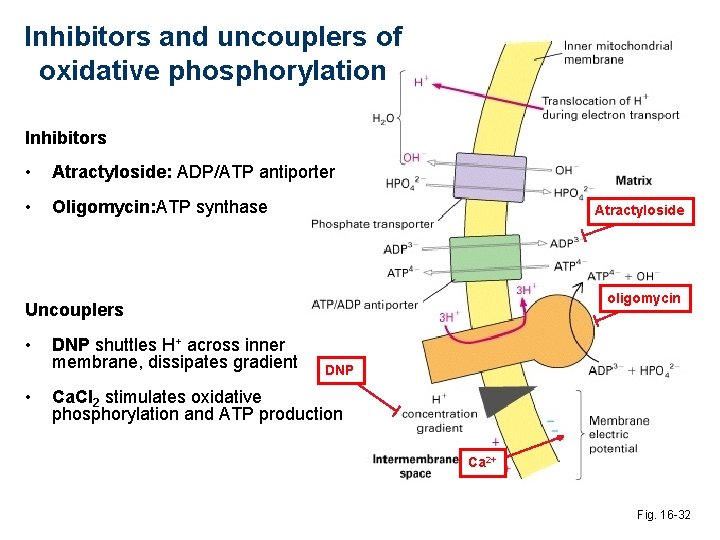
Inhibitors and uncouplers of oxidative phosphorylation Inhibitors • Atractyloside: ADP/ATP antiporter • Oligomycin: ATP synthase Atractyloside oligomycin Uncouplers • • DNP shuttles H+ across inner membrane, dissipates gradient DNP Ca. Cl 2 stimulates oxidative phosphorylation and ATP production Ca 2+ Fig. 16 -32
![Summary of Cellular Energetics Glucose High [ATP] (Pasteur effect) Glycolysis N-ethylmaleimide Pyruvate NADH Malate Summary of Cellular Energetics Glucose High [ATP] (Pasteur effect) Glycolysis N-ethylmaleimide Pyruvate NADH Malate](http://slidetodoc.com/presentation_image/24f85a12c85d3b300ac8ecfbc8810f96/image-20.jpg)
Summary of Cellular Energetics Glucose High [ATP] (Pasteur effect) Glycolysis N-ethylmaleimide Pyruvate NADH Malate FADH 2 Et. OH Acetyl Co. A Citric Acid Cycle Succinate Uncouplers Ca+2, DNP Fig. 16 -2 NADH + FADH 2 Rotenone Antimycin A Ascorbate + TMPD Sodium Azide Electron transport chain Energy released used to pump H+ creating an elecrochemical gradient Flow of protons down the gradient fuels ATP synthase Atractyloside ADP + Pi O 2 Oligomycin ATP H 2 O Oxidative Phosphorylation

Carbon Dioxide Emission Control Authority

Review: Characterization of Cellular Components l Who? l What? l Where? l When? l How? l Why?

Review l Immunofluorescence microscopy l l l Vital staining l l l Microscope Cell staining Colocalization Filters Transfection l l Eukaryotic expression vectors GFP
- Slides: 23