Lab 4 Applied Properties of Light Molecules and

Lab 4: Applied Properties of Light Molecules and Light Fall 2018 Chemistry 1331: Chemical Structures and Properties Tues. Oct. 9 - Mon. Oct. 15

Background • Types of Bonds • Covalent • Ionic • Metallic Covalent Bonding. https: //www. s-cool. co. uk/gcse/chemistry/chemical-bonding/revise-it/covalent-bonding (accessed 10/7/2018) Metallic Bonding. https: //www. chemguide. co. uk/atoms/bonding/metallic. html (accessed 10/7/2018) Ionic Bonding. https: //en. wikibooks. org/wiki/Structural_Biochemistry/Chemical_Bonding/Ionic_interaction (accessed 10/7/2018)

Background • • Bond Dipoles Molecular Dipoles

Pre-Lab • Any Questions? • Take 5/10 minutes to take the pre-lab

Procedure 1 • • Solubility • Attempt to dissolve the various solids in water Conductance (1/Resistance) • Measure the Resistance of various solids and solutions using the voltmeter • Rods • Liquid / Solution

• • In chemistry, we need to think about the distribution (or location) of electrons in a bond. The distribution of electrons is called the electron density. This distribution of electrons can be equal between atoms. When the distribution is equal or close to equal, we call this a non-polar bond. When there is relatively large difference in the location of the electrons, we have an uneven distribution of electrons. • • • If there is high electron density (d-) the atom is said to me electron rich. If there is a low of electron density at a particular location (d+) the atom will be more positively charged (electron poor). What affects the uneven sharing of electrons in a bond?

Electronegativity • • Electronegativity is the measure of the tendency of an atom to attract a bonding pair of electrons. The electronegativity difference between two bonded atoms can be used to determine bond polarity. For each of the following bonds, indicate the direction of polarity These bonds can vary in polarity based on their local environment (i. e. other atoms, other polar bonds).
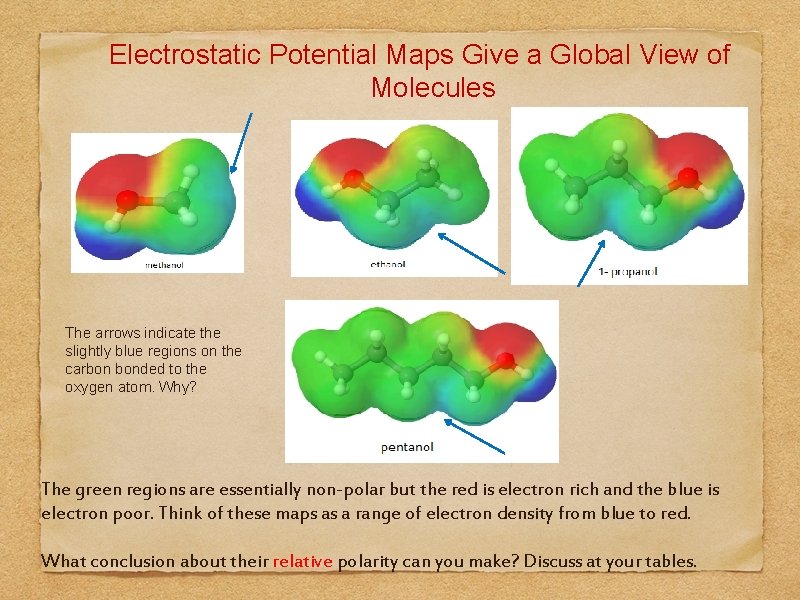
Electrostatic Potential Maps Give a Global View of Molecules The arrows indicate the slightly blue regions on the carbon bonded to the oxygen atom. Why? The green regions are essentially non-polar but the red is electron rich and the blue is electron poor. Think of these maps as a range of electron density from blue to red. What conclusion about their relative polarity can you make? Discuss at your tables.

Procedure 2: • Use the software to build 2 D and 3 D models: http: //chemdata. r. umn. edu/jmol/2 d_3 d. html • • Measure • Bond Distances • Bond Angles • Dipoles • Charge Distributions Observe the EPM

Next Week • For Next week. • Read Lab #7, be prepared to discuss.
- Slides: 10