Lab 343 Experiment 5 Determination of the rate

Lab. 343

Experiment (5) Determination of the rate constant and energy of activation of the reaction between (K 2 S 2 O 8 + KI)

K 2 S 2 O 8 + 2 KI ü S 2 O 8 -2 + I- ü (S 2 O 8 I) -3 + I- I 2 + 2 Na 2 S 2 O 3 I 2 + 2 K 2 SO 4 (S 2 O 8 I) -3 slow I 2 + 2 SO 4 -2 rapid Na 2 S 4 O 6 + 2 Na. I

K 2 S 2 O 8 + 2 KI 0. 04 0. 4 I 2 + 2 K 2 SO 4 Ø Second order of reaction Ø Pesudo first order reaction log ( a-x) = -K t / 2. 303 + log a
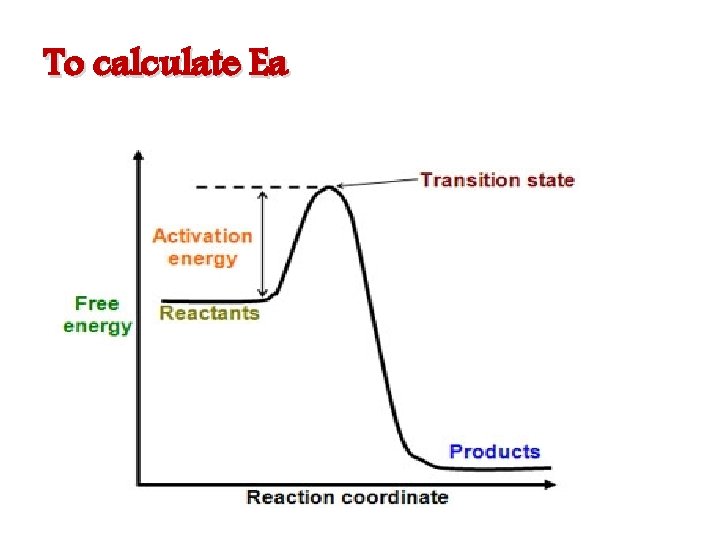
To calculate Ea

Activation energy The minimum energy, which the molecules must absorb before the reaction can take place.

K=Ae –Ea/RT Arrhenius equation Where A = frequency factor R = molar gas constant (8. 314 J/K. mole) Ea = activation energy ( J /mole) T = absolute temp. ( kelvin= C◦ + 273) K = rate constant
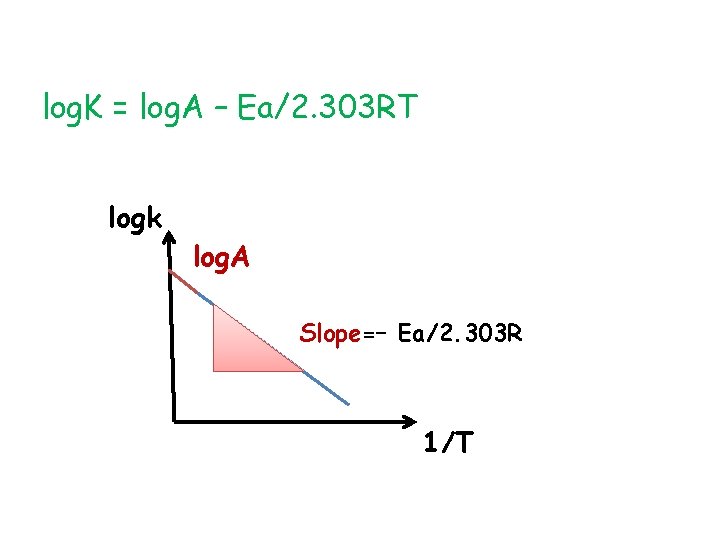
log. K = log. A – Ea/2. 303 RT logk log. A Slope=– Ea/2. 303 R 1/T

• T 1=25◦C log. K 1 = log. A – Ea/2. 303 RT 1 • T 2=35◦C log. K 2 = log. A – Ea/2. 303 RT 2 log. K 2 - log. K 1 = – Ea/2. 303 RT 2 – (– Ea/2. 303 RT 1 ) log K 2 /K 1 = Ea/2. 303 R (-1/T 2 + 1/T 1 ) log K 2 /K 1 = Ea/2. 303 R (1/T 1 - 1/T 2 ) log K 2 /K 1 = Ea/2. 303 R (T 2 -T 1 / T 1. T 2)
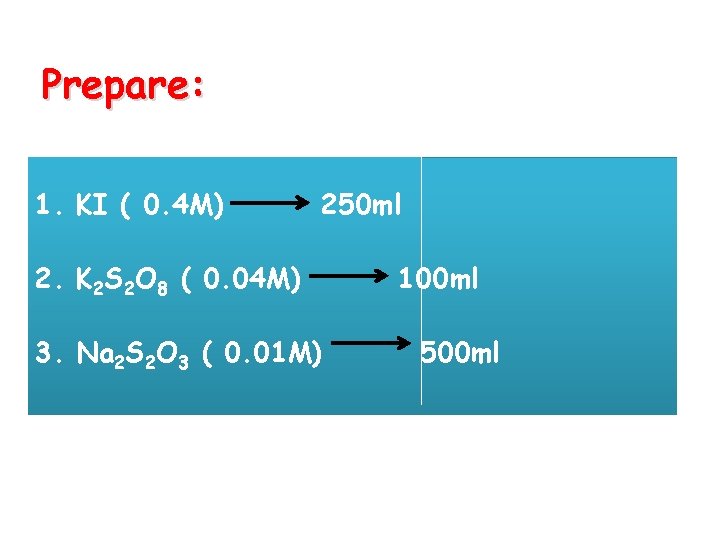
Prepare: 1. KI ( 0. 4 M) 250 ml 2. K 2 S 2 O 8 ( 0. 04 M) 3. Na 2 S 2 O 3 ( 0. 01 M) 100 ml 500 ml
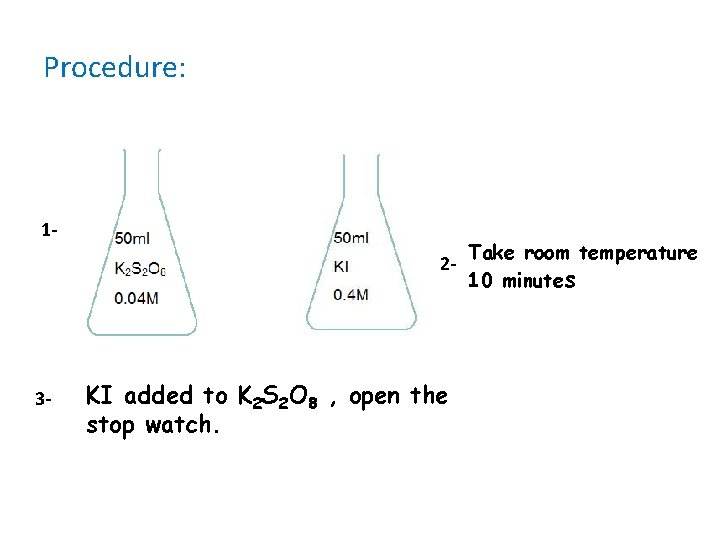
Procedure: 12 - 3 - KI added to K 2 S 2 O 8 , open the stop watch. Take room temperature 10 minutes
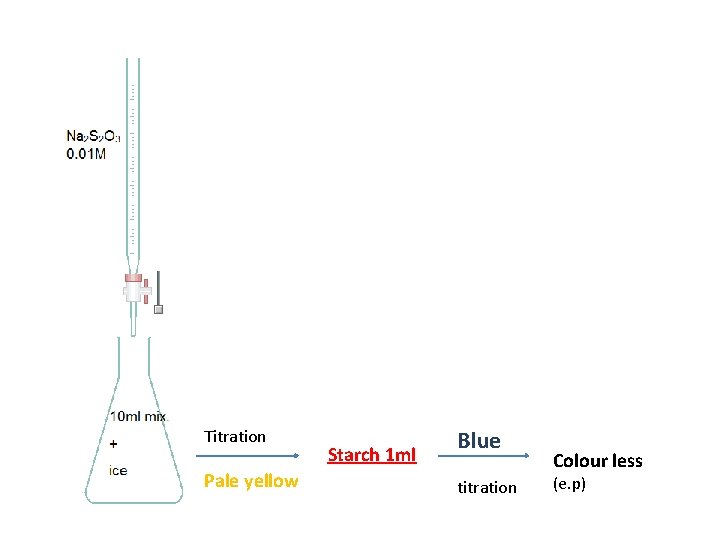
Titration Pale yellow Starch 1 ml Blue titration Colour less (e. p)

• Starch is added near the end point and not at the beginning of the titration ? ? because starch forms insoluble complex with Iodine(I 2) thus preventing it from reacting. • Excess KI (2 g)is added. . because Iodine I 2 is volatile, however in the presence of KI it forms KI 3 complex which is non-volatile and acts exactly like I 2
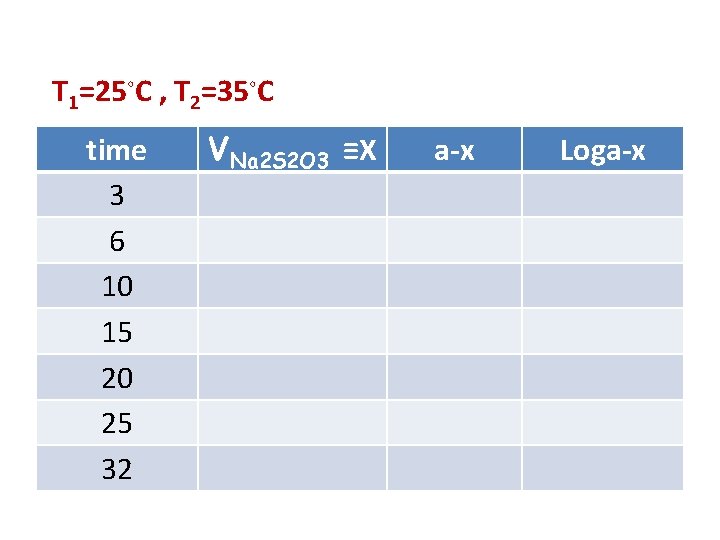
T 1=25◦C , T 2=35◦C time 3 6 10 15 20 25 32 VNa 2 S 2 O 3 ≡X a-x Loga-x
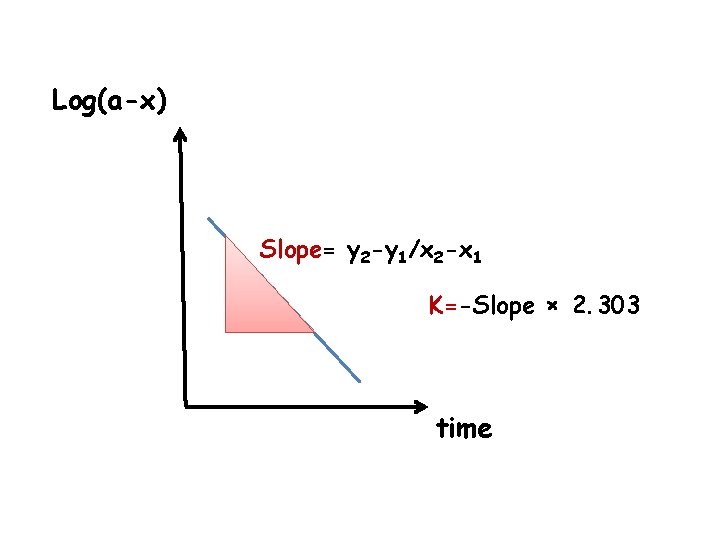
Log(a-x) Slope= y 2 -y 1/x 2 -x 1 K=-Slope × 2. 303 time
- Slides: 15