Lab 343 Experiment 3 Determination of the saponification

Lab. 343

Experiment (3) Determination of the saponification rate constant of ethyl acetate in alkaline medium

CH 3 COOC 2 H 5 + Na. OH H+ CH 3 COONa + C 2 H 5 OH t= 0 a b 0 0 t= t a-x b-x x x R = dx/dt = K [CH 3 COOC 2 H 5 ][Na. OH] = K [ a-x] [b-x]
![Secand order reaction equation If • [a] ≠ [b] Or • [a] = [b] Secand order reaction equation If • [a] ≠ [b] Or • [a] = [b]](http://slidetodoc.com/presentation_image_h/b7ac02fa153871f525b5eaf3bae9c2bb/image-4.jpg)
Secand order reaction equation If • [a] ≠ [b] Or • [a] = [b]
![• [a] ≠ [b] dx/dt = K ( a-x) (b-x) dx/ ( a-x) • [a] ≠ [b] dx/dt = K ( a-x) (b-x) dx/ ( a-x)](http://slidetodoc.com/presentation_image_h/b7ac02fa153871f525b5eaf3bae9c2bb/image-5.jpg)
• [a] ≠ [b] dx/dt = K ( a-x) (b-x) dx/ ( a-x) (b-x) = K dt by integration K = [2. 303/ t ( a-b)] [log b(a-x)/a(b-x)]
![• [a] = [b] dx/dt = K ( a-x) (b-x) dx/dt = K • [a] = [b] dx/dt = K ( a-x) (b-x) dx/dt = K](http://slidetodoc.com/presentation_image_h/b7ac02fa153871f525b5eaf3bae9c2bb/image-6.jpg)
• [a] = [b] dx/dt = K ( a-x) (b-x) dx/dt = K ( a-x)2 dx/ ( a-x)2= K dt by integration : 0 ∫ x dx/ ( a-x)2= 0 1/a-x = Kt + constant At t=0 , x=0 ∫ t K dt constant= 1/a-x = Kt + 1/a
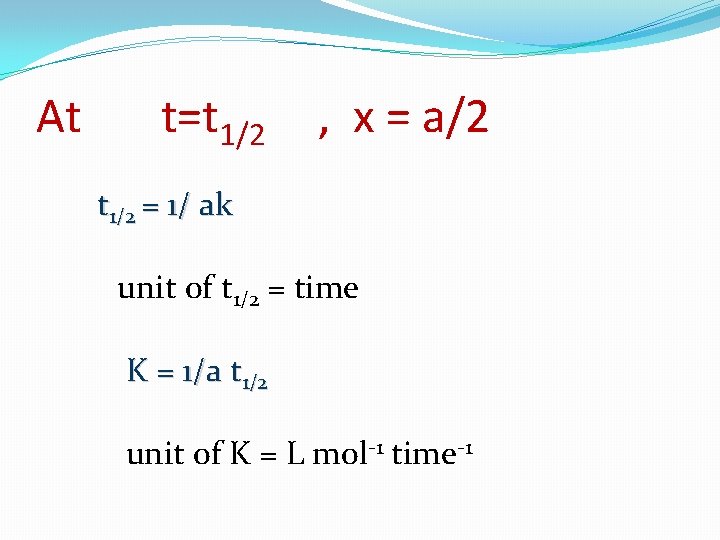
At t=t 1/2 , x = a/2 t 1/2 = 1/ ak unit of t 1/2 = time K = 1/a t 1/2 unit of K = L mol-1 time-1

Prepare: concentration Volume Liquid Solid 0. 05 M 100 ml CH 3 COOC 2 H 5 Na. OH 0. 025 M 250 ml HCl Na. OH

Procedure: 10. 05 M Take room temperature 10 minutes
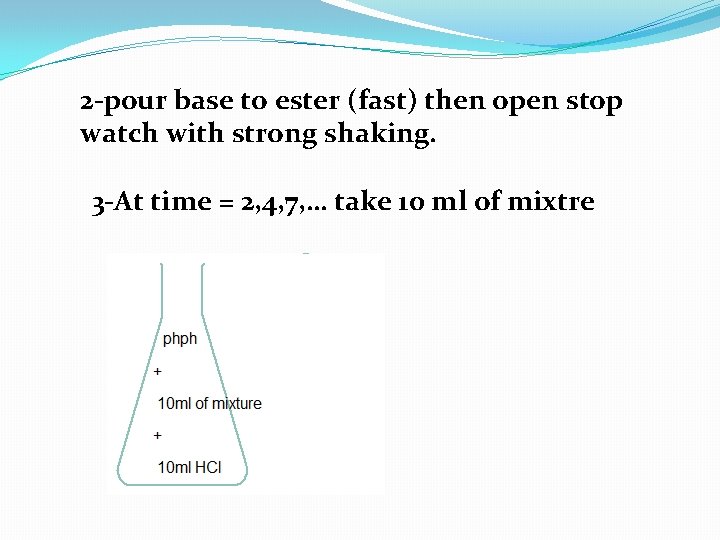
2 -pour base to ester (fast) then open stop watch with strong shaking. 3 -At time = 2, 4, 7, … take 10 ml of mixtre
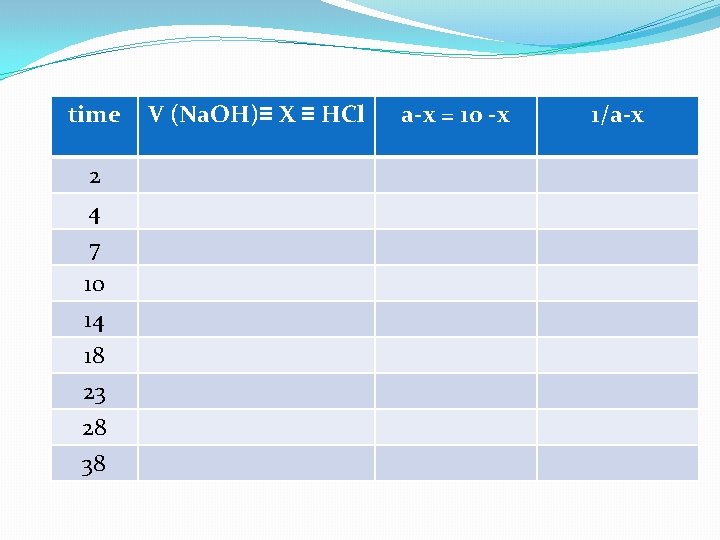
time 2 4 7 10 14 18 23 28 38 V (Na. OH)≡ X ≡ HCl a-x = 10 -x 1/a-x
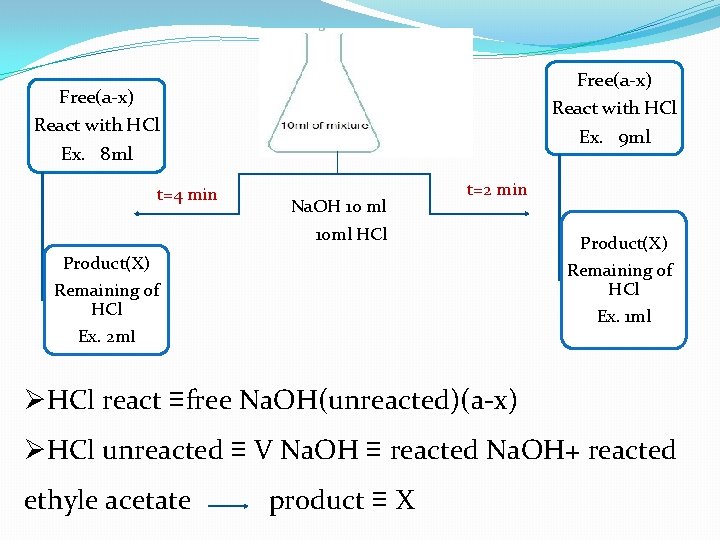
Free(a-x) React with HCl Ex. 9 ml Ex. 8 ml t=4 min Na. OH 10 ml t=2 min 10 ml HCl Product(X) Remaining of HCl Ex. 2 ml Product(X) Remaining of HCl Ex. 1 ml ØHCl react ≡free Na. OH(unreacted)(a-x) ØHCl unreacted ≡ V Na. OH ≡ reacted Na. OH+ reacted ethyle acetate product ≡ X
- Slides: 13