LAB 3 The Spectrophotometer Measuring Concentration Using Absorbance

LAB 3 The Spectrophotometer: Measuring Concentration Using Absorbance

Measuring Absorbance using a Spec • set-up: – molar mass of KMn. O 4 = 158. 03 g – 1 m. M solution standard – 3 unknowns

Determining Concentration from Absorbance – The Standard Curve • set-up: – – – – multiple test tubes prepared with decreasing concentrations of KMn. O 4 prepare a two-fold serial dilution over 10 cuvettes Cuvette #1 = 1 m. M Cuvette #2 = 2 -fold dilution = 0. 5 m. M Cuvette #3 = 4 -fold dilution (vs. test tube #1) = 0. 25 m. M Cuvette #10 = 512 -fold dilution (vs. test tube #1) each cuvette is read at Abs 545 nm 2 -fold dilution 1. 0 ml 2. 0 ml sample 64 -fold dilution 1 m. M 1. 0 ml water for dilution

Unknown • Unknowns are also read at Abs 545 nm • three unknowns of various concentrations • EXPERIMENTAL APPROACH: measure the absorbance of these three unknowns and compare them to your standards – requires determining the molar absorptivity – this is calculated using your standards and the slope of a Beer-Lambert plot

Beer’s Law • Beer’s Law relates the absorbance of light to the properties of the material through which it is passing • A = ebc – – A = absorbance of the sample at a given wavelength e = molar absorptivity of the sample (no units) b = sample path length of the sample (cm) c = concentration of the sample (same units as your standard) • rewriting the equation gives you – c = A/eb • using a spectrophotometer you can determine A for any unknown • you also know the path length of the sample = 1 cm cuvette – so b= 1 • PROBLEM: unknowns in this equation are e and c
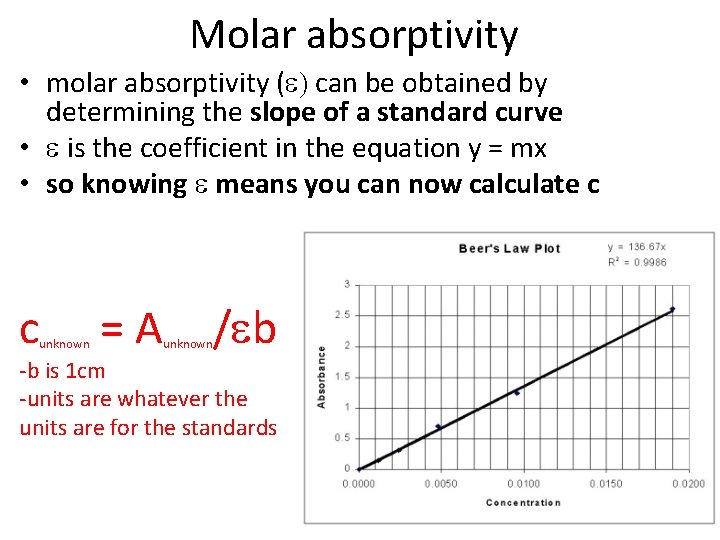
Molar absorptivity • molar absorptivity (e) can be obtained by determining the slope of a standard curve • e is the coefficient in the equation y = mx • so knowing e means you can now calculate c c unknown =A /eb unknown -b is 1 cm -units are whatever the units are for the standards
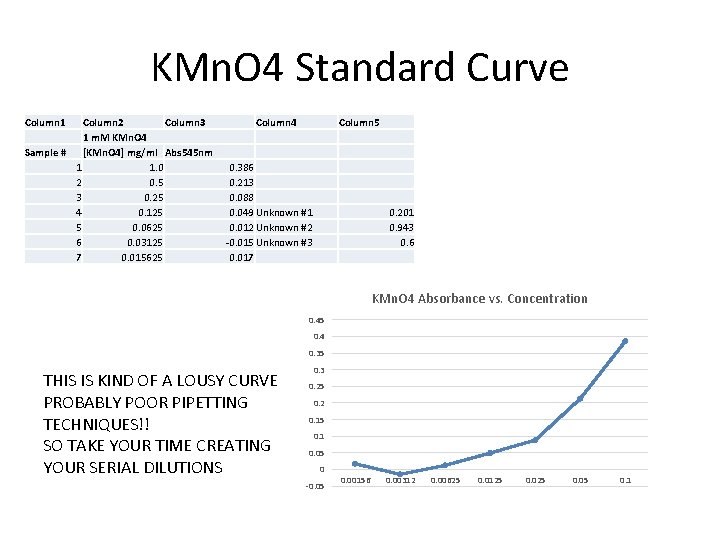
KMn. O 4 Standard Curve Column 1 Column 2 Column 3 1 m. M KMn. O 4 Sample # [KMn. O 4] mg/ml Abs 545 nm 1 1. 0 2 0. 5 3 0. 25 4 0. 125 5 0. 0625 6 0. 03125 7 0. 015625 Column 4 Column 5 0. 386 0. 213 0. 088 0. 049 Unknown #1 0. 012 Unknown #2 -0. 015 Unknown #3 0. 017 0. 201 0. 943 0. 6 KMn. O 4 Absorbance vs. Concentration 0. 45 0. 4 0. 35 THIS IS KIND OF A LOUSY CURVE PROBABLY POOR PIPETTING TECHNIQUES!! SO TAKE YOUR TIME CREATING YOUR SERIAL DILUTIONS 0. 3 0. 25 0. 2 0. 15 0. 1 0. 05 0 -0. 05 0. 00156 0. 00312 0. 00625 0. 0125 0. 05 0. 1
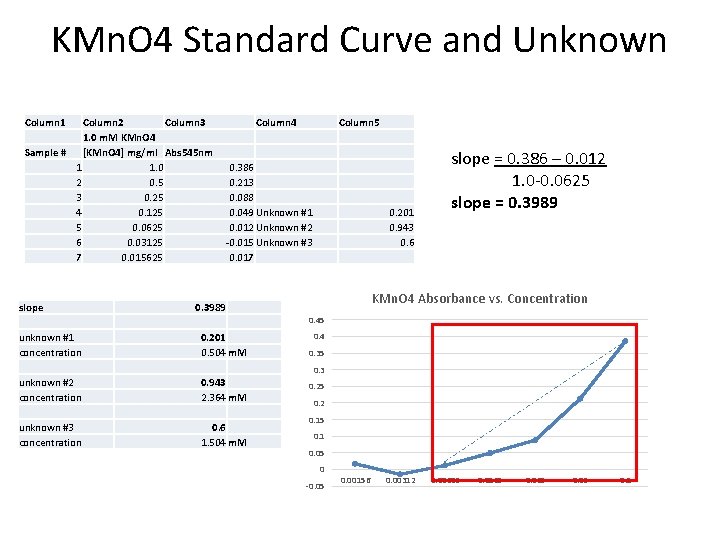
KMn. O 4 Standard Curve and Unknown Column 1 Column 2 Column 3 1. 0 m. M KMn. O 4 Sample # [KMn. O 4] mg/ml Abs 545 nm 1 1. 0 2 0. 5 3 0. 25 4 0. 125 5 0. 0625 6 0. 03125 7 0. 015625 slope Column 4 Column 5 0. 386 0. 213 0. 088 0. 049 Unknown #1 0. 012 Unknown #2 -0. 015 Unknown #3 0. 017 0. 201 0. 943 0. 6 slope = 0. 386 – 0. 012 1. 0 -0. 0625 slope = 0. 3989 KMn. O 4 Absorbance vs. Concentration 0. 3989 0. 45 unknown #1 concentration 0. 201 0. 504 m. M unknown #2 concentration 0. 943 2. 364 m. M unknown #3 concentration 0. 6 1. 504 m. M 0. 4 0. 35 0. 3 0. 25 0. 2 0. 15 0. 1 0. 05 0 -0. 05 0. 00156 0. 00312 0. 00625 0. 0125 0. 05 0. 1
- Slides: 8